1. Introduction
As a result of human activity, the increases in the accumulation of greenhouse gases in the atmosphere has contributed to severe global warming while industrial engineering developments have accelerated our rapid transition of the modern age [1, 2].
Since the distribution of precipitation is very uneven and the rate of evaporation varies a great deal, depending on the temperature and relative humidity, natural variations caused by climate change are main reason that lakes and rivers over the world are ephemeral features of the landscape. The ecosystem in these freshwater reservoirs are vital resources for aquatic wildlife and human needs and play a significant role in supplying drinking water, fishing, and recreation as well as maintaining a regional ecological balance and sustainable socioeconomic development [3–5]. Undoubtedly, the growing world population is inextricably linked with sustainable freshwater development throughout world which also places considerable pressure on the requirements of fresh water needed for agriculture, energy, and industry. Many countries still face the challenges of eliminating poverty, promoting economic growth, ensuring health and sanitation, preventing curbing land degradation and pollution, and advancing rural and urban development [6]. Therefore, the current generation is required to be responsible for freshwater conservation and leave behind a sustainable inheritance to future generations [7–9].
Mountainous countries (i.e. South Korea) can formulate action plans for water storage, especially a great deal of summer rainfall for freshwater resources through the construction of dams or weirs. Specifically, the four major river restoration projects in South Korea were begun to address the significant environmental challenges faced by the Han, Nakdong, Geum and Yeongsan rivers. However, since then those water bodies have become breeding ground for the cyanobacteria because there was not enough consideration for the aquatic ecosystems. The complex interactions of sufficient sunlight, high levels of nutrients in the rivers, and slower flow speeds are likely responsible for the larger and more frequent algal outbreaks [10–14].
The recent chemical treatment method for acute reduction of cyanobacteria using inorganic coagulants can be classified by several criteria according to: (1) applied site (surface water, water treatment plant, sediment); (2) purpose (decrease of nutrient concentration, inhibition of cyanobacteria toxin and unpleasant odor). In view of the primary role, we focus chemical treatments using inorganic coagulants containing pre-oxidants and algaecides to reduce cell counts of cyanobacteria in the surface freshwater and water treatment plant. For the easy understanding of concept with cyanobacteria control, a scenario of a representative chemical treatment process using metallic coagulants for cyanobacteria control is illustrated in Fig. 1.
2. Algae and Eutrophication in Freshwater
Eutrophication is aquatic ecosystem response to addition of artificial or national nutrients containing mainly phosphorous and nitrogen elements through detergents, fertilizers, and sewage. The trophic status of lakes describes the relationship between the nutrient status of a lake and the growth of organic matter in the lake. Eutrophication is the process of change from one trophic state to a higher trophic state by the addition of nutrients. Agriculture is a major factor in the eutrophication of surface water. Two of the major nutrients in freshwater, nitrogen (N), needed for protein synthesis, and phosphorus (P), needed for DNA, RNA, and energy transfer, are both required to support aquatic plant growth and are the key limiting nutrients in most aquatic and terrestrial ecosystems [10]. Human activity has profoundly altered the global biogeochemical cycle of N. The global production of agricultural fertilizers increased from less than 10 million metric tonnes of N in 1950 to a prediction of more than 135 million metric tonnes of N by 2030. P inputs from fertilizers and manures greatly exceed P outputs in farm produce, and P is, thus, accumulating yearly in the soil. The loading of N and P into the world’s rivers, lakes, and oceans is very strongly influenced by human population densities, the population densities of livestock, and land use [15]. Note that water quality has declined dramatically in the past 50 years, primarily because of the increase of N and P eutrophication from human or animal manure effluents, as well as from municipal and industrial wastewater. The increase in eutrophication is most likely due to an increase in nutrient input from these sources, as a result of increased human activities in the freshwater reservoir surroundings [16–18]. Algal production during the summer can be activated by N and P nutrients in the surface water so appropriate coagulators can be used as process to return to the soil with the algae containing nutrients for plant growth.
Electrostatic repulsion force make possible to long term stable state with constant distance between suspended algae cells; otherwise, the collision of particles would result in attachment as result of the universal van der Waals forces of attraction. For the growth of the algae, the most important factors include light, temperature, the chemical composition, and the acidity or alkalinity of the freshwater. Shen et al. reported that algae density drops ten times when the temperature of the surface water drops from 34°C to 27°C. This partially explains why outbreaks of algal blooms usually occur in the summer when water temperatures are high, water depth is low and water nutrient concentrations are high. Therefore, in summer when the temperature is relatively high, it is particularly important to remove algae from drinking water sources [19].
3. Cyanobacteria of Major Harmful Algae
3.1. Freshwater Reservoirs
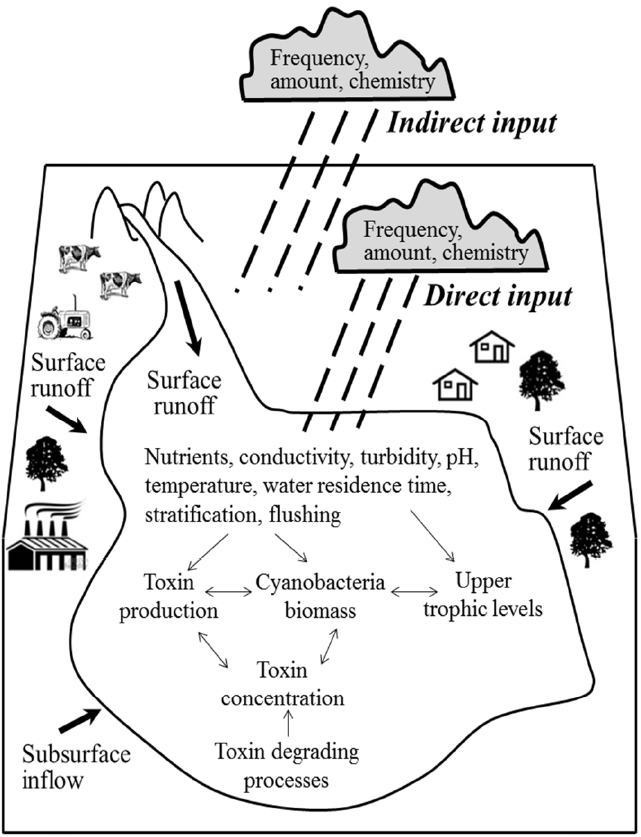
Toxin-producing cyanobacteria (blue-green algae) are the major harmful algal group in freshwater environments and are recognized as a rapidly expanding global problem that threatens human and ecosystem health. Table 1 shows cyanobacterial toxins that produce human health effects when they are taken into the human body over short and long-term periods [20–28]. Blooms of the cyanobacteria are ubiquitous phenomena in eutrophic lakes and reservoirs in many countries around the world from investigations into physicochemical and biological water quality [29, 30]. The thick scums production is a primary factor of water quality deterioration which includes foul odors and tastes because it generate deoxygenation of bottom waters, toxicity dissociation concerns, fish breathing trouble, and aquatic ecosystem destruction. Table 2 lists the cell counts of cyanobacteria species in the Deachung Reservoir which appeared during the summer season of 2003 [31]. In the algal outbreak period, safe drinking water quality is one of the most important issues in public health due to the presence of cyanobacteria in the drinking water supplies poses a hazard to humans [32–39]. A real time monitoring system is one of most critical methodology in water resource management through rapid and accurate recognition and classification of algae and cyanobacteria which cause uncomfortable tastes and odors filter clogging, and deterioration of the drinking water quality [40, 41]. A higher frequency of toxic cyanobacteria blooms in the future is likely to be attributed to complex direct function of water parameters (e.g., temperature, nutrient, light, conductivity) and complex indirect function on their predators and competitors as illustrated in Fig. 2 [42]. Chemical treatment using inorganic coagulants for rapid aggregation of cyanobacteria cell in freshwater reservoirs can be achieved in conjunction with subsequent sedimentation or floatation. While floatation tends to achieve faster rates of separation and more concentrated product stream, it is required further treatment process to move the floating product. On the other hand, coagulation-sedimentation process is often used as the low cost treatment technique in economic analysis.
3.2. Water Treatment Plant
World Health Organization (WHO) has published a provisional guideline value of 1.0 μg Microcystin (MC)-LR/l as the toxicity of cyanobacteria toxins in drinking water [43]. In practice, due to the small dimension and the low specific gravity of cyanobacteria, a single conventional process for cyanobacteria control is extremely difficult, so it is necessary to strengthen the use of combined conventional treatment processes. A most classical process in US water plants for the removal of suspended solids containing cyanobacteria residue in drinking water consists of five separate steps: 1) Coagulation, 2) Flocculation, 3) Sedimentation, 4) Filtration, and 5) Disinfection [44]. Coagulation is the process of decreasing or neutralizing the electric charge of suspended particles containing cyanobacteria with negative zeta potential. Similar electric charges on small particles in water cause the particles to naturally repel each other and hold the small, colloidal particles apart and keep them in suspension. The coagulation/flocculation process neutralizes or reduces the negative charge on the particles. This allows the van der Waals force of attraction to encourage the initial aggregation of colloidal and finely suspended materials to form microfloc. Flocculation is the process of bringing together the microfloc particles to form large agglomerations by either physically mixing them or through the binding action of flocculants. Suitable chemicals in the coagulation process are added to the initial influent wastewater stream, which is stirred and intensively mixed at a high speed. The influent wastewater in the flocculation process is only moderately stirred in order to form large flocs, which are easily settled out. The following factors should be taken into consideration for selecting the most appropriate coagulant/flocculant aid: 1) The effectiveness of removing phosphorus, 2) Cost and the reliability of supply, 3) Sludge considerations, both volume and characteristics, 4) Compatibility with other upstream or downstream treatment processes, 5) Environmental effects, 6) Labor and equipment requirements for storage, feeding, and handling [44].
In drinking water treatment, conventional coagulation is still the main treatment process for algal removal which results in the repeated collision and attachment of cells, forming progressively larger agglomerates that quickly settle. The inorganic particles usually originate from natural or anthropogenic sources and inhibit algal growth by attenuating light and limiting photosynthesis. Besides scattering and adsorbing solar radiation, suspended solids can also act as an adhesion surface.
4. Conventional Chemical Treatment
4.1. Preoxidants
Peroxidation is deemed effective in promoting the coagulation of algal cells and is widely used in drinking water treatment. Peroxidation chemical includes ozone, chlorine, chlorine dioxide, permanganate, or ferrate. The effects of disinfection and inactivation on the algae cells cause the changes of the zeta potential due to promote their aggregation. Peroxidation by permanganate also generates manganese dioxides in situ, which can adhere to the cells and promote their settling. However, peroxidation by chlorine (NaOCl) or chlorine dioxide (ClO2) forms undesirable disinfection byproducts such as chlorinated organics, chlorate, and chlorite. Ozone, chlorine, or ferrate can induce release of cyanobacteria toxins. Especially, ozone and permanganate are the most effective oxidants in promoting coagulation, so they are widely used in water treatment. Peroxidation can improve cyanobacteria coagulation to reduce the these cell counts but some of them result in taste- and odor occurrences due to higher oxidation strength [45–50].
4.2. Algaecide
To control the massive growth of algae in lakes and reservoirs, copper sulfate (CuSO4·5H2O) has been the most widely used as substance for the control of undesirable algal growth for more than 110 years [51–54]. Single additions of copper sulfate do not effect a permanent reduction in algal abundances and repeated additions are needed to keep populations under control. Eventually copper is trace element that is an essential component of the biochemical functions of photosynthetic organisms. However, it can affect electron transport through photosystem and modify or inhibit fundamental enzymatic activities. Especially, in excessive concentrations of copper sulfate can promote the release of toxic Microcystin compounds because they affect physiological stress and cell membrane damage of cyanobacteria by great algaecide strength. Also, copper precipitates out of the water column or sorbs to suspended solids and accumulates in bottom sediments. Copper toxicity has also been the subject of many studies on freshwater algae and cyanobacteria species and copper bioavailability is related to its speciation, which is most likely relating to pH, redox potential, alkalinity, and organic and inorganic ligand concentrations. Therefore, for the potential impacts of copper sulfate additions on lake ecosystems might need to anticipate future environmental issues [55–57].
4.3. Inorganic Coagulants
4.3.1. Clay
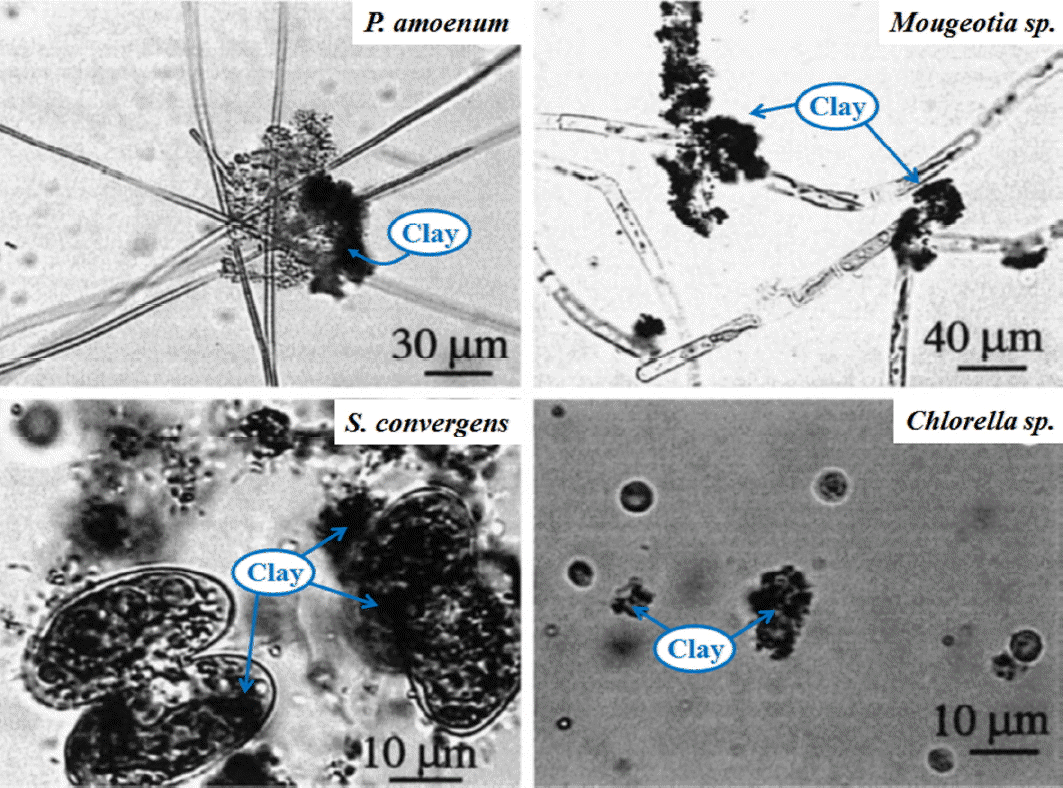
Many countries (US, Australia, Sweden, Japan, South Korea) have been known to use clay coagulants to remove algae [58–61]. In a 1996 report, workers in South Korea dispersed approximately 60,000 tons of dry yellow loess (a kaolinite-bearing sediment) that removal rates of specific algal blooms were calculated at 90% to 99% [61]. Fig. 3 shows the appearance of various kinds of algal cell surface that clay is adhered on algae cells by coagulation process [60]. Most representative clay has been Ca-montmorillonite, kaolinite, sericite, and illite as shown in Table 3 [62–64]. Clay minerals form sheet-like particles or platelets with a wide range of particle sizes from tens of angstroms to millimeters and their inherent properties also make them chemically active and adsorptive, thereby leading to a variety of uses as absorbent and catalyst products. Fig. 4 illustrates that in clay minerals, the active sites may arise from (1) broken edge sites and exposed surface aluminol and silanol groups, (2) isomorphic substitutions, (3) exchangeable cations, (4) hydrophobic silanol surfaces, (5) hydration shells of exchangeable cations, and (6) hydrophobic sites on adsorbed organic molecules [65]. The elements of silica, iron, aluminum, and magnesium can bridge linking algal cells, and coagulate them into flocs. Other heavy metals with positive charges such as Cr3+, Cu2+, Pb2+, Zn2+, Cd2+ have a high affinity for other negatively charged algae cells. However, this adsorption capacity varies among clay types which may influence the extent of the aggregation process. The initial zeta potential of an algal solution without cells was −39.7, but the clay (i.e. vermiculite) treated solution was fixed at 36 [66]. The suspended particles may also increase the algal cells sinking through their mutual aggregation. The formation of algae-clay aggregates depends on the number of contacts and the subsequent adhesion between algal cells and clay particles. The degree of contact will then be a function of both algae and clay concentrations, as well as of the algal morphology. The probability of this contact will depend on the clay concentration and the cell size (i.e., length, width, and surface area). The extent of the adhesion will be a function of the ionic interaction between the algae and clay as well as of the algal cell surface properties. Small sized algal cells with high surface area would require a proportionally larger amount of adhered clay particles to accelerate their sinking [60]. During the last several decades, for instance, South Korea has mainly used clay because of its cost effective advantages but clay may cause structural and functional damage in cases of long-term exposure into the human bodies because quartz chemicals, dissolving from clay, is especially related to silicosis and lung cancer [67–69].
4.3.2. Aluminum sulfate or ferric chloride
The coagulation process of algal blooms from polluted freshwater has used routinely Al and Fe salts (i.e., aluminum sulfate, ferric chloride) since early in the 20th century and played a significant role in the removal of a broad range of impurities, including suspended algal cells, colloidal particles, and dissolved organic substances [70–74]. The essential mechanisms for algae removal using these salts is accomplished by charging the neutralization of negatively-charged colloids by dissolved cations and incorporating the impurities into an amorphous precipitate of metal hydroxide. A simple mechanism for the destabilization of negatively-charged particles is the specific adsorption of cationic species from solutions. The cationic Al and Fe ions of hydrolysis products neutralize or reduce the negative charge on the suspended solids containing anionic algae cells in coagulation/flocculation process. In the case of Al3+ and Fe3+, there is a primary hydration shell with six octahedrally coordinated water molecules (e.g., Al(H2O)63+). The hydrolysis of such ions is thought of as progressive deprotonation of water molecules in the primary hydration shell as following Eq. (1):
Increasing the pH causes the equilibria to be shifted to the right since each step involves the loss of a proton. In the case of Al, the dominant species in the solution changes from Al3+ to Al(OH)4− over little more than 1 pH unit. In contrast, the corresponding change for Fe occurs over a range of more than 8 pH units and intermediate species, such as Fe(OH)2 + and Fe(OH)3, can represent more than 90% of the soluble forms at intermediate pH values. At pH values around 5–6, the suspended algae cells are negatively charged and amorphous Al and Fe hydroxides are positively charged, which gives strong adsorption and some charge neutralization [70].
However, concerns have been raised that the use of such coagulants may increase Al concentrations in treated water. In Japan, the drinking water quality guideline for Al is 0.1 mg/L, but water treatment plants usually set stricter values for finished water (e.g., < 0.05 mg/L). Orally ingested Al is acutely toxic to humans and a risk factor for the onset of Alzhemer’s disease in the case of long-term exposure (WHO 1998). Since the ingestion of Al from both food and drinking water is the most common form of human exposure, the main rout of Al adsorption is through the gastrointestinal tract [75–77]. In particular, maintenance of Fe and Al residuals of less than 100 μg/L was recommended, which was more formally accepted in the Water Quality Regulations of 1989 which mandated less than 200 μg/L Fe or Al in tap water. On the other hand, concerning the water supply, coexistent chloride anions with these metal cations generate corrosion products in non-ferrous or ferrous metal which consist of storage tanks, pumps, piping, valves, and concrete. The corrosion products deteriorate water quality as well as provide habitats for microbial growth, react with disinfectant residuals [78–82].
4.3.3. Lime
Usually, single or multiple treatments of slake lime (Ca(OH)2) and calcite (CaCO3) are applied for removal on algal blooms and nutrients in eutrophic freshwaters. Whereas single lime treatments resulted in variable and mostly temporary changes in chlorophyll a and P concentrations, multiple treatments were effective in reducing both chlorophyll a and P concentrations over longer periods [83–85]. Lime application seems to be an economical and non-toxic tool for the restoration of surface waters because lime (calcium hydroxide) treatment does not cause any toxin release of the microcystin LR (a species of cyanobacteria). However, copper sulfate should not be used to treat toxic algal blooms in waters to be consumed by humans or animals within several weeks following treatment [86]. Cyanobacteria have been recognized as a key player in the precipitation of calcium carbonate in freshwater systems. They increase pH as a result of photosynthetic activity and also produce extracellular polysaccharides, which act as binding sites for Ca2+ and CO32− [87].
Oyster shells have a high potential for reuse due to their low levels of heavy metals and high level of calcium (35%). The shell residue can potentially produce added economic value if it is transformed into raw material for other production processes because the shells contain high amounts of calcium carbonate. The calcium carbonate content of oyster shells can range from 80% to 95% [88]. After the addition of shell powder into algal freshwater, the color was dramatically changed by physicochemical reactions according to the four-step process of addition, agitation & carbonation, coagulation, and settlement. Visible transparency of algal freshwater was also gradually converted from dark green to colorless according to the alteration of added amounts of calcined powders as shown in Fig. 5 [89]. For the total pollutant load of the algal solution, the mean removal (or reduction) efficiency of total phosphorus (T-P), total nitrogen (T-N), and chemical oxygen demand (COD) was recorded at 96.5%, 91.4%, and 33.8%, respectively [89]. The calcium hydroxide and carbonation combination in algae-containing water significantly contributed to improve water quality which is very dependent on the addition amount of calcined oyster shell powder [90].
Table 4 shows the charge transfer reaction and 25°C logK alteration between protonic and metallic charge groups of dolomite coagulator surfaces and carboxylate, phosphate, and hydroxyl charge groups of algae surfaces. In natural water, anionic algal surface charge arises from carboxylate (pH 4–6), phosphate (pH 7–8), and hydroxyl groups (pH 9–10) which become deprotonated, lose their H+. However, dolomite coagulator dissolves the oppositely-charged divalent cations such as Ca2+ and Mg2+ and cation bridges with adjacent algae coagulation. The solution reaches a maximum at pH ~11 because of Ca(OH)2, Mg(OH)2 formation. Mg2+ and Ca2+ adsorption to algae might induce prompt coagulation to charge neutralization by forming bridges between individual, negatively-charged algae because the mutual repulsion of the negatively-charged algal surface helps keep algae in suspension in most natural waters. When the pH is raised from ~8 to 9–11, algae coagulation is also caused due to electrostatic interactions between anionic algae and divalent cations such as Ca2+ and Mg2+. Their precipitates with hydroxide or/and calcite formation are shown in Fig. 6 [91].
4.3.4. Future outlook of coagulants
The primary key of the successful treatment is to select available metals or chemical compounds as reasonable coagulators because every coagulant has both advantages and disadvantages as summarized in Table 5 [92, 93]. Still, the usage of coagulants is the cheapest and fastest-working method and, thus, is the most widespread across the world. However, toxic algicide (e.g., copper sulfate) can be replaced by more environmentally-friendly coagulants (e.g., aluminum sulfate, ferric chloride, and lime) although their price is higher relatively. The weak point of some coagulants is that it is still too expensive to produce them with high quality and purity. To apply industrial algae coagulants to water in reservoirs or dams, it is probably necessary to employ cost effective materials such as recycled oyster shell waste or unused dolomite kiln dust. Additional efforts will be needed to contribute to establishment of emergent solutions to algal blooms and to preserve valuable freshwater resources.
5. Conclusions
For sustainable freshwater resources development, conventional chemical treatment containing preoxidants, algaecide, metallic coagulators have been demonstrated extensively in the literature to efficiently remove cyanobacteria which release the toxins in eutrophic freshwater and water treatment plant. The proliferation of algal blooms in eutrophic freshwater and water treatment plant is a worldwide problem. Although the only long-term solution for this problem is the restriction of nutrient inputs into water bodies, this method seems to be unavailable for most areas across the world because of the large financial costs. Thus, a reasonable application of techniques for cyanobacteria removal can be well founded in some cases with regards to health risks and the commercial occupancy of water bodies. This paper deals with potential coagulants to immediately improve water quality of reservoirs and plants in summer seasons through emergent chemical treatments for excessive cyanobacteria control and its residue removal. Future chemical treatment for cyanobacteria control should additionally be considered nutrient reduction of its external inputs and reduction of internal loading nutrient from released from sediments. Therefore, to successful management of the freshwater, chemical treatment performance with nutrient fixation and oxygen production ability should be additionally required to sustainable freshwater resources development and healthy aquatic animal and plants preservation.