AbstractGeneration of food waste is a serious issue that needs to be addressed worldwide. Developing suitable treatment methods while generating energy (methane) is a common practice for sustainable treatment of waste. In this study, methane generation by food waste was investigated in mesophilic and thermophilic regimes at various hydraulic retention times (HRTs) and organic loading rates (OLR). In temperature regimes, influent concentrations and HRTs ranged from 30 to 110 g COD/L and 18 to 30 days, respectively, which corresponding to an OLR of 1.0 to 6.1 kg COD/m3-d. Better methane production and organic removal was observed under thermophilic conditions because of the enhanced hydrolysis of complex polymers and microbial activity at higher temperature. The peak methane productivities attained in thermophilic and mesophilic regimes were 1.30 and 0.99 m3/m3-d, respectively. The maximum methane yields were achieved at 50 g COD/L and HRT of 24 d in both cases, and the values were 264 and 221 m3/ton COD, respectively. The results of this study will facilitate the development of sustainable methane production technologies using food waste as a feedstock.
1. IntroductionThe 3R (Reduce, Reuse and Recycle) concept of waste management has recently received a great deal of attention because of the generation of enormous amounts of solid/agricultural/municipal wastes [1]. Global food waste (FW) generation is immense because of the increasing population and consumption by humans. According to the Food and Agriculture Organization (FAO), every year about one third of food created for consumption is wasted worldwide. However, the FW in developing countries and developed countries clearly differs [2]. For developing countries, over 40% of FW occurs after harvest and during processing, while in developed countries, over 40% occurs as a result of retail and consumption behavior. FW is officially defined as material composed of raw or cooked food that is thrown out during the entire process, from manufacturing to disposal. FW can be classified into groups depending on the original source, chain of food consumption, and physical-chemical characteristics [3].
Anaerobic digestion has long been applied to degrade organic matter efficiently and produce methane as a product. This process is a well established technology that transforms the organic fraction into renewable fuels such as methane, especially for slaughterhouse waste, municipal waste and agricultural wastes [4, 5]. Methane can be used for various purposes such as direct combustion, generation of electricity, fuel for vehicles and other applications [6–8]. In general, the conversion of FW to methane generation via anaerobic digestion is a complex process, and many reports have indicated that hydrolysis and methanogenesis are the rate-limiting step in anaerobic digestion because of the accumulation of undesirable volatile fatty acids [9–11]. Nevertheless, controlling the major reactor operational parameters such as organic loading rate (OLR), temperature, and hydraulic retention time (HRT) can improve the process efficiency and stable methane generation. Previous studies reported that FW can be effectively converted to methane under mesophilic and thermophilic regimes [12, 13]. However, limited studies have focused on long term operations to assess bioreactor stability. Thus, in this study, the long-term anaerobic digestion of food waste (nearly 380 days) was evaluated under various (OLRs), (HRTs) and temperature regimes (mesophilic and thermophilic).
2. Materials and Methods2.1. Inoculum and FeedstockSeed sludge was collected from a mesophilic anaerobic sludge digester in a local wastewater treatment plant. The pH, total sludge solids (TS), and volatile sludge solids (VS) concentration of the sludge were 7.5, 26.7 g/L, and 18.9 g/L, respectively. The sludge was stored at 4°C in a refrigerator until being used in the experiments. The same seed sludge was used for both mesophilic and thermophilic experiments.
The food waste was collected form a cafeteria located on the campus of Daegu University. The collected food waste was ground using a blender to obtain a homogenous mixture, then stored at 4°C in a refrigerator. The characteristics of the food waste were as follows: total solids (TS) 158±39 g/L, volatile solids (VS) 151±37 g/L, total chemical oxygen demand (COD) 157±61 g/L, soluble COD 47±17 g/L, total carbohydrate 99±28 g/L as glucose, soluble carbohydrate 54±19 g/L as glucose, total suspended solids (TSS) 68±48 g/L, volatile suspended solids (VSS) 67±50 g/L, pH 4.5±1.0, g carbohydrate/g VS 0.656, g carbohydrat/g COD 0.631, g VS/g TS 0.956.
2.2. Continuous Methane Production from Food WasteMethane fermentation was conducted in continuously stirred tank reactors with a working volume of 7 L (thermophilic) and 3.6 L (mesophilic), respectively. Four different HRTs were evaluated in regimes, 18, 20, 24 and 30 d. The feed and draw was once a day with continuous agitation of 100 rpm. The feedstock concentration was adjusted to 30–110 g COD/L by dilution of the food waste with distilled water, which corresponds to an OLR of 1–5.5 g COD/L/d. No additional nutrients were supplied throughout the operation period.
2.3. Specific Methanogenic ActivityThe specific methanogenic activity (SMA) of the biomass from both mesophilic and thermophilic reactors was measured as previously described [12] using batch serum vials. The biomass (methanogenic microflora) was collected from each operation condition and utilized for evaluation of SMA. Biomass of 0.6–0.8 g VSS was checked for a specific substrate (2 g COD/L) of glucose, acetate and propionate.
2.4. AnalysisBiogas production was measured using water displacement equipment connected to each reactor, and then corrected to the standard temperature and pressure (0°C and 1 atm). CH4, N2, and CO2 contents in the biogas were analyzed by gas chromatography (GC, SRI 310, SRI Instrument) using a thermal conductivity detector and a 1.8 m × 3.2 mm stainless-steel column packed with mole sieve 5A (SRI Instrument) and a 0.9 m × 3.2 mm stainless-steel column with Porapak Q (80/100 mesh, SRI Instrument). Organic acids were analyzed by high performance liquid chromatography (HPLC, YL9100 series) using an ultraviolet detector (210 nm) and a refractive index detector with a 300 mm × 7.8 mm Aminex HPX-87H ion exclusion column with 0.005 M H2SO4 as eluent. Solids, COD, and alkalinity were measured according to the Standard Methods [14]. All other analyses were conducted as described in our earlier studies [15–17].
3. Results and Discussion3.1. Biogas Production at Various HRTs and OLRsThe estimated generation of biogas production and its methane content is an important parameter to consider when designing pilot scale or industrial level anaerobic digesters [18, 19]. Various types of organic feedstock have been used such as cattle manure, swine manure, food waste and empty palm fruit bunches, and their co–digestion has been studied for the generation of methane as an energy source. The methane production performance at various HRTs and OLRs is shown in Fig. 1 and Table 1.
The influent concentration was examined at 30, 50, 80 and 110 g COD/L, while the OLRs were 1, 1.66, 2.08, 3.33, 4.58, 5.5 and 6.1 kg COD/m3-d based on the HRTs, which were set at 30, 24, 20 and 18 d under both mesophilic and thermophilic conditions. As shown in the table, production performance gradually increased as the OLR increased. This likely occurred because of adaptation of the methanogens to the reactor environment and the substrate. The production performances revealed that the peak methane productivity attained was 1.30 m3/m3-d, while the HRT was 20 and 18 days and the OLR was 5.5 and 6.1 kg COD/m3-d (influent concentration of 110 g COD/L) under thermophilic conditions, respectively. However, the corresponding high yield of 264 m3/ton COD was achieved at HRT of 24 d with an OLR of 2.08 kg COD/m3-d. This could be attributed to the better COD removal of 78% and lower influent COD of 50 g COD/L. The production rates ranged from 0.20 to 1.30 m3/m3-d and the yields ranged from 156 to 264 m3/ton COD, respectively.
3.2. Thermophilic vs Mesophilic Regime-Process PerformanceThermophilic digestion of the organic fractions to methane has recently received a great deal of attention owing to its better hydrolysis rates and improved sanitary conditions [20–23]. In this study, the performance of the reactor was enhanced under thermophilic conditions (Table 1 and Fig. 1). The MPR ranged from 0.20 to 0.99 m3/m3-d under mesophilic conditions and 0.22 to 1.30 m3/m3-d under thermophilic conditions, respectively. A peak MY of 264 m3/ton COD was attained under thermophilic conditions and an OLR of 2.1 kg COD/m3-d, influent concentration of 50 g COD/L and HRT of 24 d. Under mesophilic conditions, a peak MY of 221 m3/ton COD was achieved at the same influent concentration (50 g COD/L), OLR and HRT, respectively. The COD removal range was 54–85% for mesophilic and 61–86% for thermophilic conditions. Based on these results, reducing the HRT significantly enhanced the production performance, as mentioned in previous studies [22, 23]. However, stable or similar production performance was observed at a HRT of 24 d, although the yield decreased slightly (from 172 to 162 m3/ton COD under mesophilic conditions; 213 from 236 m3/ton COD under thermophilic conditions). This could mainly be attributed to volatile fatty acid generation and lower COD removal efficiencies.
3.3. Specific Methanogenic Activity of the Inoculum
Fig. 2 (a and b) compares the specific methanogenic activity (SMA) of the biomass at each OLR. Analyses were conducted using the main precursors of the anaerobic digestion, glucose, acetate and propionate to identify the methanogenic activity of the acclimated anaerobic microflora. Thermophilic anaerobic microflora clearly exhibited better performance than mesophilic microflora. The SMA decreased as the OLR increased. These findings were similar to those of our previous study [12]. Moreover, the SMA of acetate was relatively higher at OLRs of 1.7 and 2.1 kg COD/m3-d during the mesophilic digestion, resulting in 48.2 and 98.6 mL CH4/g VSS-d, respectively. However, in cases of OLRs of 3.3 and 5.5 kg COD/m3-d, propionate provided better results (17.4 and 27.1 mL CH4/g VSS-d, respectively). Thermophilic digestion of food waste showed enhanced activity of SMA and more affinity towards acetate than propionate, which was accompanied by propionate accumulation during the operation. However, the activity was quite similar to that of the mesophilic microflora, whereas increasing OLR resulted in decreased SMA. The peak SMA of 185.2 mL CH4/g VSS-d was attained during the OLR of 2.1 kg COD/m3-d using acetate as the precursor. In contrast to the mesophilic digestion, during the high OLRs the SMA showed more affinity towards glucose precursor, with peak SMA values of 63.7 and 32.8 mL CH4/g VSS-d being observed for OLRs of 3.3 and 5.5 kg COD/m3-d, respectively. Thermophilic digestion improved the SMA and production performance when food waste was used as substrate.
3.4. Volatile Fatty Acids (VFA) ProductionThe volatile fatty acid production profile is shown in Fig. 3 (a) and (b). The production of propionic acid was higher under mesophilic conditions, whereas acetate was dominant under thermophilic conditions at OLRs of 4.6–6.1 kg COD/m3-d in both regimes, and acetate and propionate were the major organic acids detected. Moreover, higher amounts of total organic acid (482–918 mg COD/L) were observed at OLRs of 4.6–6.1 kg COD/m3-d under mesophilic conditions, while total organic acid concentrations of 256–726 mg COD/L were observed at the same OLR range under thermophilic conditions. Thus, the accumulation of propionate led to failure of the mesophilic reactor since the conversion of acetate from propionate is a limiting step in the anaerobic digestion process [24, 25]. The appearance of formic acid and lactic acid was observed at a higher OLR of 6.1 kg COD/m3-d. In contrast, n-butyric acid was not detected throughout the operation. These findings indicated that increasing the organic loading rate resulted in acetogenesis rather than methanogenesis and led to lower production performances. Moreover, these findings indicate that accumulation of acids should be avoided to enable better production performances.
4. ConclusionsIn this study, the long term evaluation of conversion of food waste to methane was demonstrated at various HRTs and OLRs under thermophilic and mesophilic conditions. The results revealed that increasing the OLR increased the methane production performance under both mesophilic and thermophilic conditions. Better productivities were attained under thermophilic conditions because of the efficient hydrolysis of food waste at elevated temperatures. The results of this study demonstrated that food waste could act as a feasible feedstock for the bioenergy production.
AcknowledgmentsThis work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIP) (No. 2014R1A2A2A04005475).
References1. Sakai S, Yoshida H, Hirai Y, et al. International comparative study of 3R and waste management policy developments. J Mater Cycles Waste Manag. 2011;13:86–102.
2. Lin CY, Sen B. Conference report: kitchen waste-based bioenergy: a report of the international workshop on kitchen waste-based bioenergy. Biofuels. 2013;4:155–157.
3. Bao Dung TN, Sen B, Chen CC, Kumar G, Lin CY. Food waste to bioenergy via anaerobic process. Energ Proc. 2014;61:307–312.
4. Holm-Nielsen JB, Al Seadi T, Oleskowicz-Popiel P. The future of anaerobic digestion and biogas utilization. Bioresour Technol. 2009;100:5478–5484.
5. Abbasi T, Tauseef SM, Abbasi SA. Anaerobic digestion for global warming control and energy generation-An overview. Renew Sustain Energy Rev. 2014;16:3228–3242.
6. Bayr S, Rantenen M, Kaparaju P, Rintala J. Mesophilic and thermophilic anaerobic co-digestion of rendering plant and slaughter house wastes. Bioresour Technol. 2012;104:28–36.
7. Kim SH, Han SK, Shin HS. Kinetics of LCFA inhibition on acetoclastic methanogenesis, propionate degradation and β-oxidation. J Environ Sci Heal Part A. 2004;39:1025–37.
8. Palatsi J, Vinas M, Guivernau M, Fernandez B, Flotats X. Anaerobic digestion of slaughterhouse waste:main process limitations and microbial community interactions. Bioresour Technol. 2011;102:2219–2227.
9. Kondusamy D, Kalamdhad AS. Pre-treatment and anaerobic digestion of food waste for high rate methane production – A review. J Env Chem Eng. 2014;2:1821–1830.
10. Escudero A, Lacalle A, Blanco F, Pinto M, Díaz I, Domínguez A. Semi-continuous anaerobic digestion of solid slaughterhouse waste. J Env Chem Eng. 2014;2:819–825.
11. Takashimaa M, Tanaka Y. Acidic thermal post-treatment for enhancing anaerobic digestion of sewage sludge. J Env Chem Eng. 2014;2:773–779.
12. Kim SH, Choi SM, Ju HJ, Jung JY. Mesophilic co-digestion of palm oil mill effluent and empty fruit bunches. Environ Technol. 2013;34:2163–70.
13. Shin JD, Han SS, Eom KC, Sung S, Park SW, Kim H. Predicting methane production potential of anaerobic co-digestion of swine manure and food waste. Environ Eng Res. 2008;13:93–97.
14. APHA/AWWA/WEF. Standard methods for the examination of water and wastewater. 21st edWashington, D.C: American Public Health Association; 2005. p. 1368.
15. Kim SH, Cheon HC, Lee CY. Enhancement of hydrogen production by recycling of methanogenic effluent in two-phase fermentation of food waste. Int J Hydrogen Energ. 2012;37:13777–13782.
16. Kim SH, Shin HS. Effects of base treatment on continuous enriched culture for hydrogen production from food waste. Int J Hydrogen Energ. 2008;33:5266–74.
17. Park JH, Yoon JJ, Park HD, Kim YJ, Lim DJ, Kim SH. Feasibility of biohydrogen production from Gelidium amansii. Int J Hydrogen Energ. 2011;36:13997–14003.
18. Cho HS, Moon HS, Lim JY, Kim JY. Effect of long chain fatty acids removal as a pretreatment on the anaerobic digestion of food waste. J Mater Cycles Waste Manag. 2013;15:82–89.
19. Kim IS, Kim DH, Hyun SH. Effect of particle size and sodium ion concentration on anaerobic thermophilic food waste digestion. Wat Sci Technol. 2000;41:67–73.
20. Angelidaki I, Ahring BK. Effects of free long chain fatty acids on thermophilic anaerobic digestion. Appl Microbiol Biotech. 1992;37:808–812.
21. Forster-Carneiro T, Perez M, Romero LI. Thermophilic anaerobic digestion of source sorted organic fraction of municipal solid waste. Bioresour Technol. 2008;99:6763–6770.
22. Cooney C, Ackerman RA. Thermophilic anaerobic digestion of cellulose waste. Appl Microbiol Biotech. 1975;2:65–72.
23. Kim HW, Nam JY, Shin HS. A comparision study on the high rate co-digestion of sewage sludge and food waste using a temperature phased anaerobic sequencing batch reactor system. Bioresour Technol. 2011;102:7272–7279.
Fig. 1Daily variation of methane production performance under mesophilic (a) and thermophilic (b) conditions. 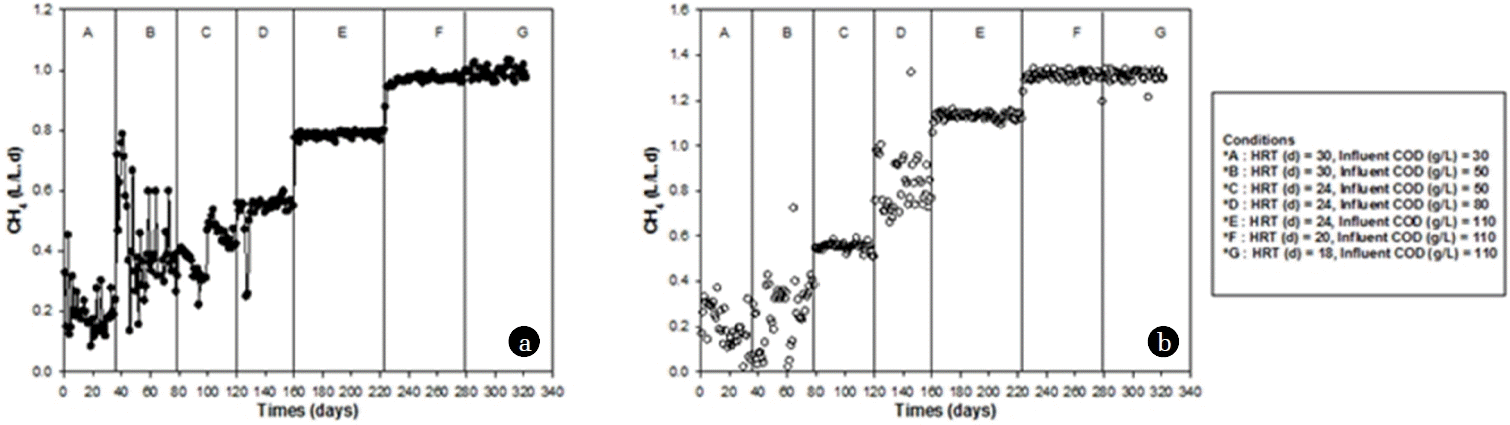
Table 1Production Performances at Various HRTs and OLRs
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||