1. Introduction
Due to industrialization and urbanization periods, contaminant such as metals, dyes, suspended solids and organic compounds release into the environment [1,2]. In these pollutants, especially heavy metals and dyes regarded as dangerous contaminants. Chromium (Cr) is a proven carcinogen which extensively applied in leather, painting and many other fields causes serious health issues in humans [3,4]. Other contaminants which are dyes, main components of paint, textile, and tannery industries are released in large amounts to wastewater. In the present day, more than 600,000 tons of dyes are produced annually in the world and more than 50% was used for textile dyeing [5].
Various physical, chemical, physicochemical, and biological methods have been proposed as solutions for the treatment of contaminated effluents [6,7]. Due to limitations and disadvantages of a single treatment such as high equipment maintenance costs, high energy consumption, and excess sludge generation, its application in actual wastewater treatment plants may be difficult. For this reason, to surpass pollutants negative impacts in wastewater, uncomplicated, cost effective, efficient and combined treatment process could optimized [5,8]. For this approach, researchers examine combining physical and biological methods to eliminate contaminants. Compare to traditional treatment methods, biological removal of contaminates like heavy metals and dyes provides benefits [9–11].
Electrospun nanofibrous mats/membranes show small fiber diameters, excellent pore interconnectivity, flexibility in surface functionality and large surface area. Because of their attractive properties, electrospun nanofibrous mats/membranes present a unique opportunity in membrane technology [12]. There are few techniques to produce nanofibers but among them electrospinning is one of the advanced technology to develop nanofibers [13–16]. In bioremediation process, fibrous webs have become a popular carrier matrix for attachment/encapsulation of microorganisms [16,17]. Compared with remediation wastewater with free bacteria, bacteria attached nanofiber mats have the following advantages: 1) the structure of the electrospun nanofibers provide bacteria to attached and produce biofilm formation; 2) nanofibers protect bacteria from environmental extremes 3) lower space necessity 4) and nanofibers can be easily recovered and reused.
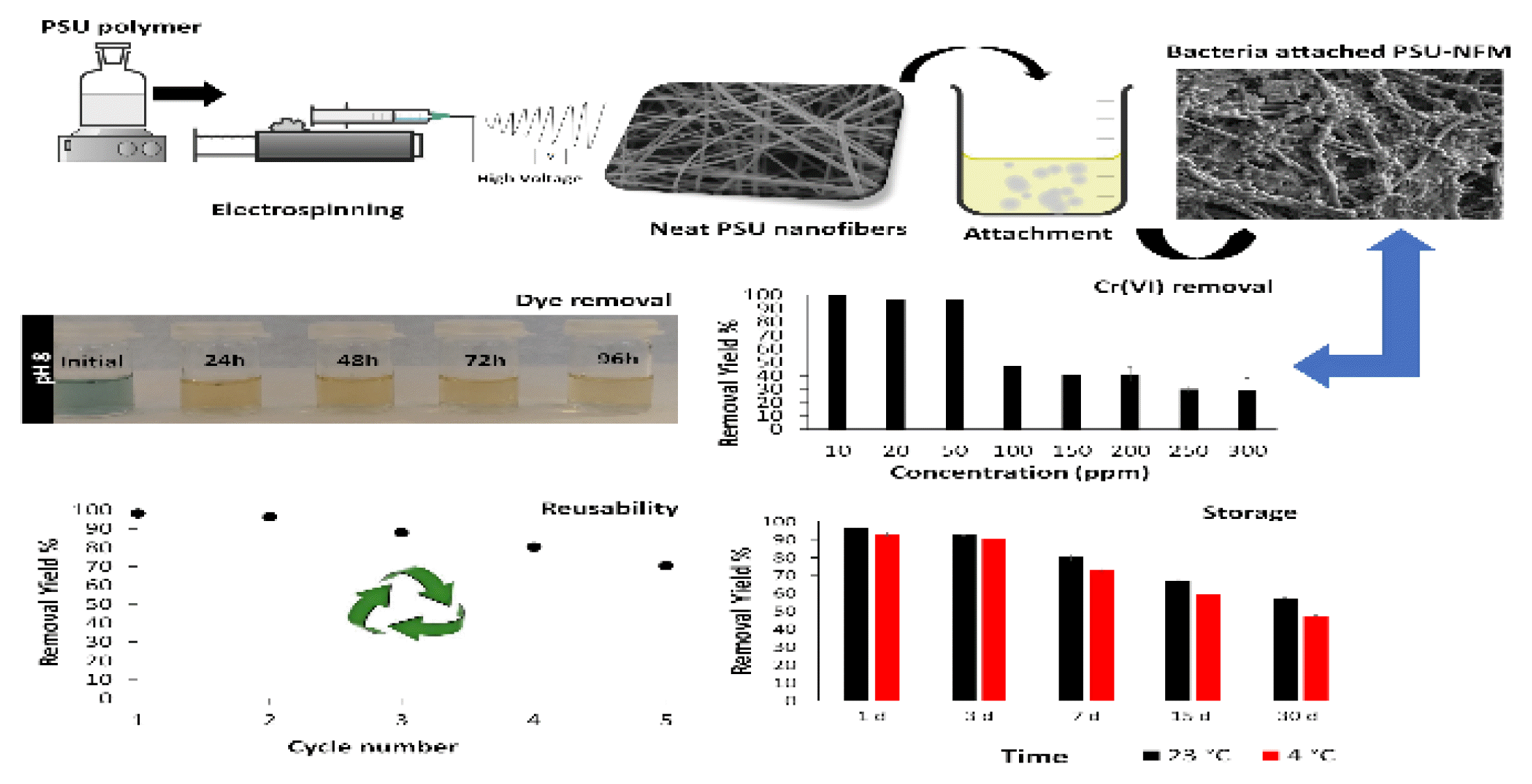
In this study, polysulfone (PSU) which has excellent thermal and mechanical stability was used for production of nanofiber as nanocarrier, followed by attachment of bacteria onto nanofiber mats and then evaluated in the removal of Cr(VI) and reactive dye, finally investigated for reusability properties. Moreover, the study reveling that bacterium attached nanofiber mats could be stored at the room temperature and + 4°C for at least 30 days without losing removal capacity.
2. Materials and Methods
Polysulfone polymer (PSU, Mw 60.000, Acros), Reactive Blue dye (Setazol Blue BRF-X, (Setaş Turkey), N,N-Dimethylacetamide (DMAC, 99%), acetone (≥99%), Tween 80, Nutrient broth (NB), Nutrient agar (NA), LB Broth and LB agar were purchased from Sigma Aldrich. Cr(VI) solutions were prepared by diluting K2Cr2O7 (Riedel-de Haen) stock solution using distilled water.
Residue from corroded pipe system in the water treatment was collected and used for the bacteria isolation. Isolated bacteria characterized using 16S rRNA nucleotide sequences, found Clavibacter michiganensis (C. michiganensis) and submitted to NCBI GenBank under GQ466171 Accession number [18]. Bacterial growth was cultured in N. Broth (NB) medium on a rotary shaker for 24 h at 30 ± 1°C and 100 rpm.
2.1. Preparation of the Electrospun Nanofibrous Membranes
In this study, the nanofibrous mats were produced by electrospinning technology. Aligned and randomly oriented PSU-Nfs were produced by using different collectors. To prepare the PSU polymer solution, 32% (w/v) PSU was dissolved in N,N-dimethylacetamide/acetone (9:1, (v/v)) binary solvent mixture and mixed using magnetic stirrer. PSU solution was filled in syringe fitted with metallic needle within inner diameter of 0.6 mm (23G). Grounded stationary plate metal collector was used for randomly oriented PSU-Nf production. Grounded cylindrical collector with of a rotary motor (3000 rpm) was used for the production of aligned PSU fibers. During the electrospinning process 15 kV was applied to it. The collector was placed at a distance of 11 cm from the spinneret and 0.5 mL/h flow rate was applied. All of the experiments were carried out at ambient temperature (25°C) with 56% relative humidity.
2.2. Bioremoval Studies
Bioremoval experiments were conducted in triplicate. Different parameters include contact time, initial pH of the solution, temperature, shaking conditions (0, 100 rpm, 200 rpm) and initial contaminant concentration were varied to find out the optimum conditions for the removal of Cr(VI) and RB by C. michiganensis.
The effects of initial pH on the RB dye decolorization, initial pH value of NB medium to pH 4–9 were adjusted with acid and sodium hydroxide solutions. Then, 1.0 ml (v/v) of 24 h inoculated bacterium culture was added in 100 ml of NB broth supplemented with 20 mg/L of RB dye followed by incubation at 30 ± 1°C under 100 rpm shaking condition. After different time intervals (24 h to 96 h), samples were taken and centrifuged (10 min at 4000 rpm). Supernatant was utilized to monitor the decolorization of dye at 597 nm using a UV–visible spectrophotometer (Shimadzu UV-1800) [1].
Hexavalent chromium was analyzed at 540 nm with 1,5-diphenylcarbazide in acidic solution. Industrial wastewater contains both Cr(VI) and RB dye. For this reason, to determine the single effect of initial Cr(VI) and RB concentration, C. michiganensis was incubated in NB containing 10–300 mg/L Cr(VI) / RB. Increased concentrations are represented by low, medium, and high concentrations of contaminants.
The% removal yield was calculated following Eq. (1) as below:
where Ci is initial concentration and C0 is the final concentration (mg/L).
2.3. Growth and Bacterial Attachment on PSU Nanofibers
Sterile Nutrient Broth medium for attachment experiments was inoculated with 1 mL of bacterial culture and 20 mg of pristine PSU-NFMs were added to the inoculation flasks and incubated at 30°C in a rotary shaker at 100 rpm for different days with different attachment time: 1 day, 3 days, 7 days, and 10 days to find maximum bacteria attachment time. Maximum bacterial attachment was monitored by attached bacteria counting. For this purpose, after the end of the incubation period, PSU-NFMs were washed with sterile distilled water then NFMs were added into LB Broth medium containing Tween 80 and vortexed at maximum intensity for 30 seconds. Then, a serial dilution of the samples was made and 100 μL of the sample was streak inoculated on LB agar and the petri dishes were incubated at 30°C for 24 hours. After the incubation period, the colonies were counted.
2.4. Scanning Electron Microscopy (SEM) Analysis
The morphology of the nanofibers and bacteria attached to nanofiber mats was determined using a Scanning Electron Microscope (Tescan GAIA 3 FIB-SEM). Neat nanofibers were coated with ~ 5 nm AuPd using the Precision Surface Etching and Coating System (Leica EM ACE600, Germany). In addition, sample fixation was applied overnight incubation with 2.5% glutaraldehyde solution. The next day, mats were washed with PBS buffer and the samples were dehydrated by immersion in a series of ethanol (EtOH, 30–96%) series. The nanofibers’ diameters were measured using ImageJ.
2.5. Reusability Experiments for Bacteria Attached to PSU-NFMs for Cr(VI) and Dye Removal
Reusability of the bacteria attached to PSU-NFMs were performed five times for decolorization and Cr(VI) removal studies (20 mg/L for RB and 10 mg/L for Cr(VI)). Before each cycle, bacteria attached to NFMs were washed with PBS buffer for the removal of deattached bacteria and nutrients. After reusability cycles, Cr(VI) and dye removal analysis were done. In addition, the morphology of the nanofibrous mat imaged by the SEM.
2.6. Long Term Storage of Bacteria Attached to PSU-NFMs for Cr(VI) and Dye Removal
The storage stability of bacteria attached to PSU-NFM at 4 °C was analyzed. A fixed weight of NFM containing bacteria was collected in sterile plastic flat dishes, placed in sealable plastic self-sealing bags and stored at room temperature (23 ± 2°C) and 4°C. The activity of bacteria attached to PSU-NFM was analyzed at predefined times (after 1, 3, 7, 15, and 30 d).
3. Results and Discussion
3.1. Investigation of Optimum Conditions on the Removal of Cr(VI) by C. michiganensis
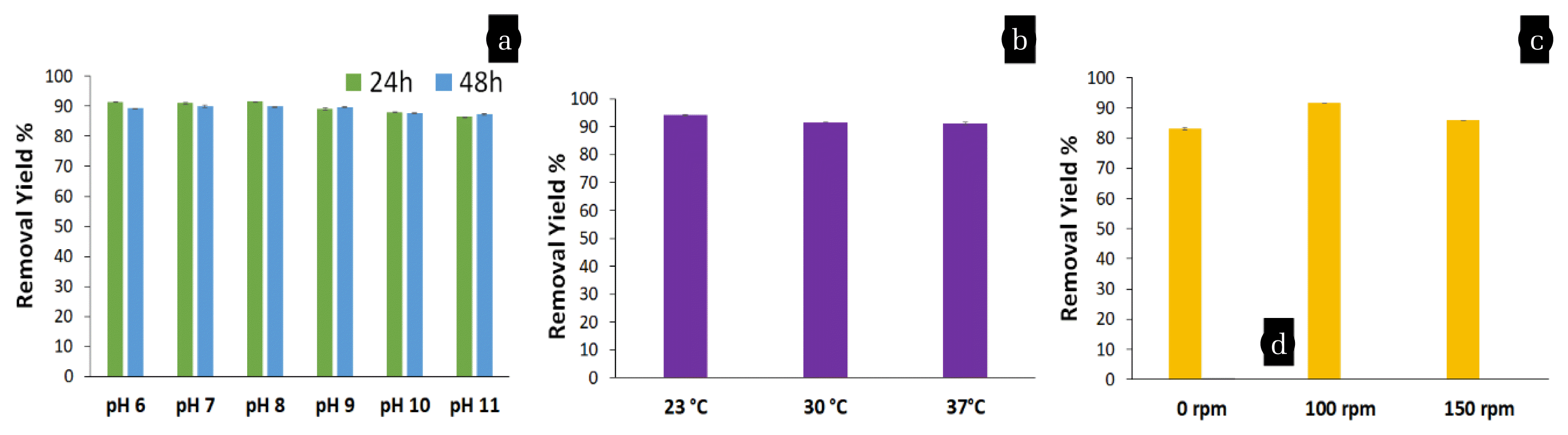
Fig. 1a shows the effect of different pH and contact time on removal of 20 mg/L Cr(VI). At pH 8, the maximum Cr(VI) removal (91.53 ± 0.01%) was achieved followed by pH 6 (91.49 ± 0.01%), pH 7 (91 ± 0.02%), pH 9 (89.09 ± 0.34%), pH 10 (89.98 ± 0.2%) and pH 11 (86.44 ± 0.01%) within 24 h incubation at 30 °C in static conditions. Furthermore, when contact time increased to 48h, Cr(VI) removal rate were 89.3 ± 0.01% (pH 6), 90 ± 0.4% (pH 7), 89.8 ± 0.01% (pH 8), 89.7 ± 0.2% (pH 9), 87.7 ± 0.2% (pH 10) and 87.2 ± 0.2% (pH 11). The results suggested that bacteria are able to remove Cr(VI) in alkaline conditions within 24h. Actual textile wastewater’s pH value is generally neutral or alkaline so that bacteria have better adaptability in neutral or alkaline environments.
The effect of temperature on the removal of Cr(VI) at 23°C, 30°C and 37°C is shown in Fig. 1b. At 23°C, a maximum of 94.15 ± 0.2% Cr (VI) removal was recorded followed by 91.53 ± 0.1% (30 °C) and 91.21 ± 0.4% (37 °C) within 24 h incubation. Fig. 1b depicts that those bacteria are able to remove the Cr (VI) over a wide range of temperatures (23–37°C).
Different agitation rates such as 0, 100 rpm and 150 rpm were applied while keeping other parameters constant. Fig. 1c indicate that the Cr(VI) removal efficiency increased from 83.04% to 91.53% when agitation rate increased from 0 to 100 rpm. As a result, removal efficiency generally increased with increasing agitation rate.
3.2. SEM of PSU Nanofibers
Fig. 2a and Fig. 2b shows the SEM micrographs with photographs of the inset of PSU nanofibers obtained with aligned and randomly oriented morphologies. To find the suitable morphology of nanofiber mat for Cr(VI) removal, aligned and randomly oriented nanofiber mats were used for removal process. Removal experiments yield were very close as 89.66 ± 0.1% and 87.93 ± 0.1% for randomly oriented and aligned nanofiber respectively. Because of the removal yields, further studies were performed with randomly oriented PSU nanofiber as carrier matrix for bacterial integration and the filter.
3.3. Bacterial Attachment on PSU Nanofibers and Cr(VI) Removal Capabilities
Equivalent PSU nanofibers were used for bacterial attachment experiments. Firstly, nanofiber mats were cut and added into the active culture of C. michiganensis for 1, 3, 4, and 7-days inoculation at 100 rpm, 30°C for. On inoculation days, samples were taken, and bacteria count were evaluated by colony count analysis. In addition, bacteria attached mats were used for Cr(VI) removal analysis. According to colony count analysis (Fig. 3), the number of viable cell counts were increased with increased inoculation time. Colony count was 2.5 × 105 (CFU/mL) after 1-day attachment and the Cr(VI) removal was 90.06 ± 0.2%. The viable counts after 3 and 4-day incubation could significantly increase to 18 × 105 and 23 × 105 (CFU/mL) with the higher removal yields (96.4 ± 0.2%, 96.79 ± 0.2%). However, after 7-day incubation, the attached bacteria number was decreased to 2.2 × 105 CFU/mL and removal yields began to decrease (93.86 ± 0.2%) due to depletion of an essential nutrient, and/or the formation of an inhibitory product. After 4 days of incubation, which were chosen optimum bacterial attachment time, the biofilm formation was examined by SEM imaging. As seen in Fig. 3b–e, after 4 days, bacteria were attached and formed thick biofilm layers on the NFW.
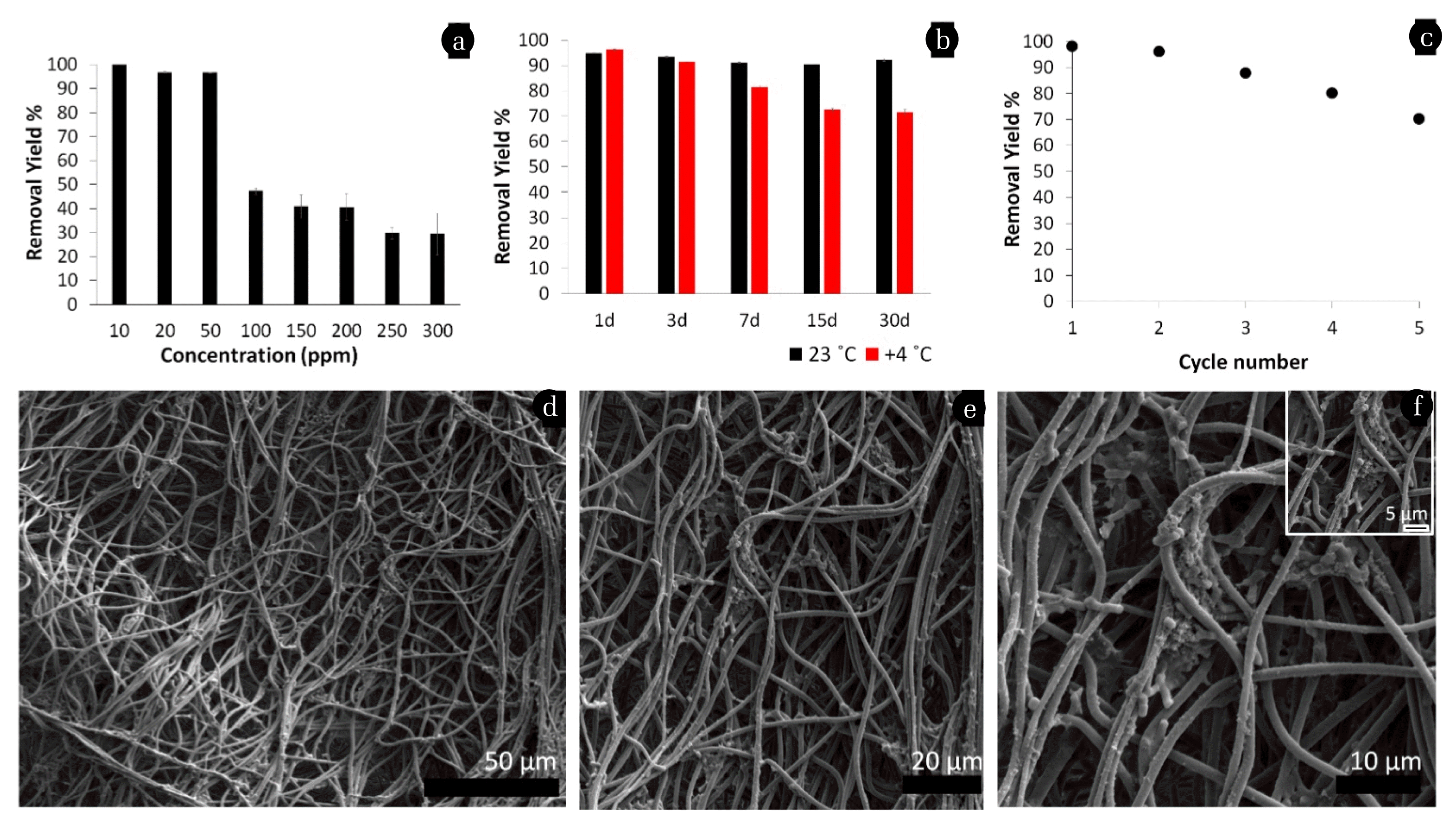
The effect of initial Cr(VI) concentration on the removal by bacteria attached to PSU-NFMs were investigated by using various contaminant concentrations. As given in Fig. 4a, the removal efficiency of Cr(VI) by bacteria attached to PSU-NFMs were 100%, 96.79%, 96.69%, 47.33%, 41%, 40.71%, 29.78%, and 29.38% for the initial Cr(VI) concentration of 10 mg/L, 20 mg/L, 50 mg/L, 100 mg/L, 150 mg/L, 200 mg/L, 250 mg/L, and 300 mg/L, respectively. As regards the removal efficiency, it decreased from 100% to 29.38% as the initial Cr(VI) concentration increased from 10 to 300 mg/L.
3.4. Long-term Storage and Reusability Results of PSU-NFMs for Cr(VI) Removal
Stability during long-term storage and operation is essential for the practical application of attached/immobilized cell systems. In this respect, bacteria attached to PSU-NFMs showed good storage stability at both 23°C and 4°C (Fig. 4b). Bacteria attached to PSU-NFMs did not lose their removal potential after room temperature and cold storage up to 30 days with a high removal efficiency with 92.12 ± 0.5% and 71.52 ± 1.1% for 23°C and 4°C respectively. Finally, the reusability performance of the bacteria attached to PSU-NFMs were checked by applying a slight washing step to the nanofibrous web after each removal step, and then the nano mats were investigated up to five cycles. As shown in Fig. 4c, bacteria attached to PSU-NFMs provides high reusability performance (at 3rd reuse 75 ± 1.7%). In each new cycle, due to loss of bacterial attachment that proved with SEM micrographs (Fig. 4d–f) Cr(VI) removal was decreased. However, SEM images of the reused bacteria attached to PSU-NFMs do not show any notable change for nanofiber structure.
3.5. Investigation of Optimum Conditions on the Removal of RB by C. michiganensis
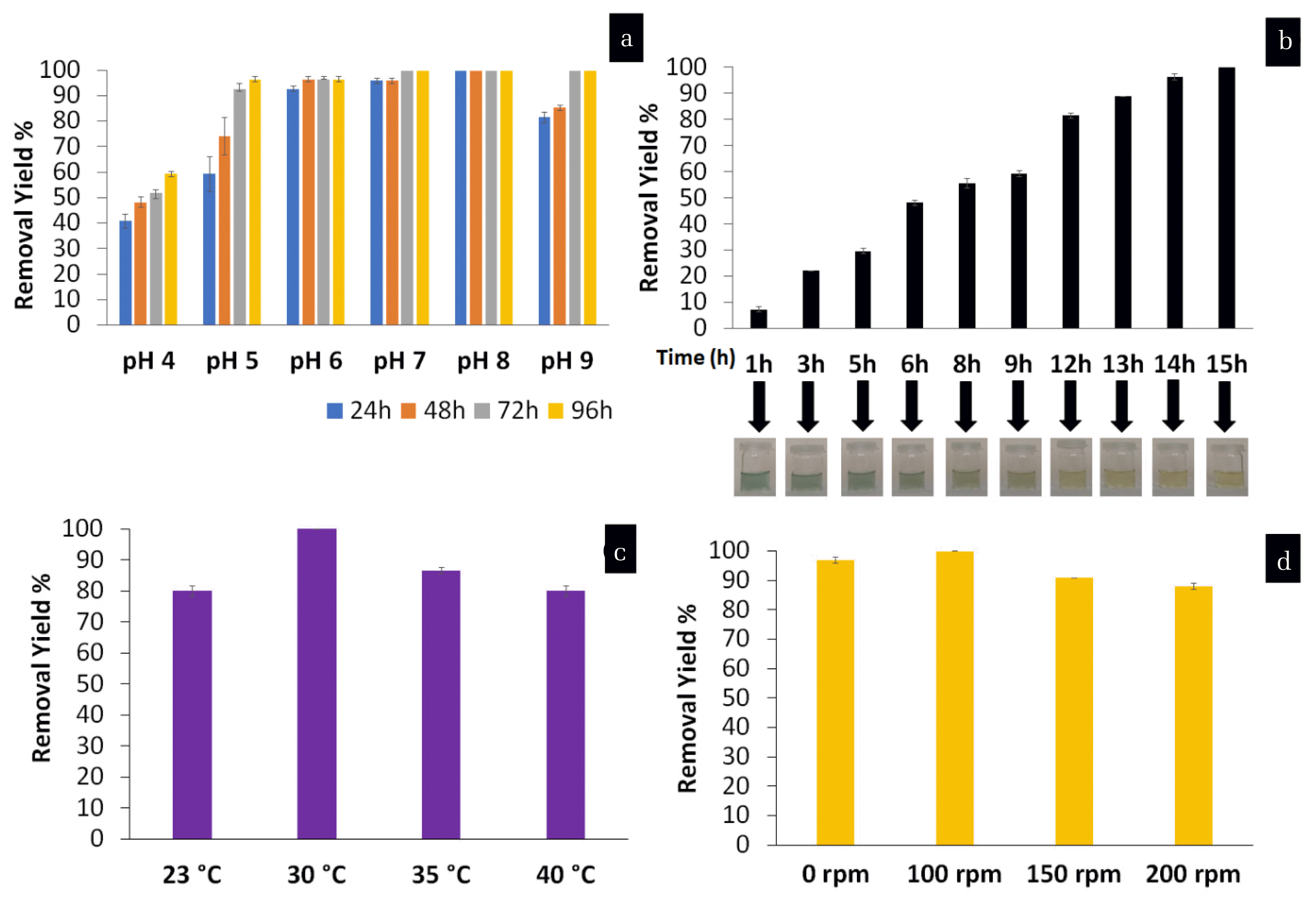
Like heavy metals, dyes are also exhibit carcinogenic, immunogenic and, mutagenic characteristics [5]. Especially, due to water-soluble properties, reactive dyes pass unaffected through conventional treatment systems and create problems. The efficiency of dye removal was first tested as a function of the pH and contact time and results were given in Fig. 5. As shown in Fig. 5a, the RB removal efficiency increased from 40.7% to 100% in 24 h, when pH value increased from 4 to 8. However, the removal efficiency showed a downtrend when pH value further increased, decreasing to 81.4% at pH value of 9. As a result, the dye was completely removed at pH 8 in 24 h. After that, the optimum contact time was investigated by measuring the percentage of dye removal at different periods of time from 1 h to 15 h in pH 8 (Fig. 5b). The results showed that dye removal increased with the trial time and reached 100% removal yield for 15 h. Color changes between initial to 15 h contact time also support the removal results. As depicted in Fig. 5c, the RB can be completely removed at 30°C and the removal of RB was inhibited at temperature of 25°C (80%), 35°C (86.7%) and 40°C (80%). At low temperature, fluidity and transportation ability of the cell membrane decreased. For this reason, the substrates cannot rapidly enter the cell to support cell growth. Furthermore, the structure of membrane will change due to high temperature and protein structures damaged [19]. Finally, after shaking at 100 rpm, a decolorization of 100% was recorded, whereas it achieved 90% and 87% for 150 rpm and 200 rpm, respectively (Fig. 5d). In the literatures, for instance, Zamel et al. [20] studied the immobilization of Bacillus paramycoides bacterial strain in cellulose acetate-poly(ethylene oxide) nanofibers for decolorization ad removal of basic methylene blue dye from wastewater. In our study, a complex reactive dye which is very difficult to remove was studied.
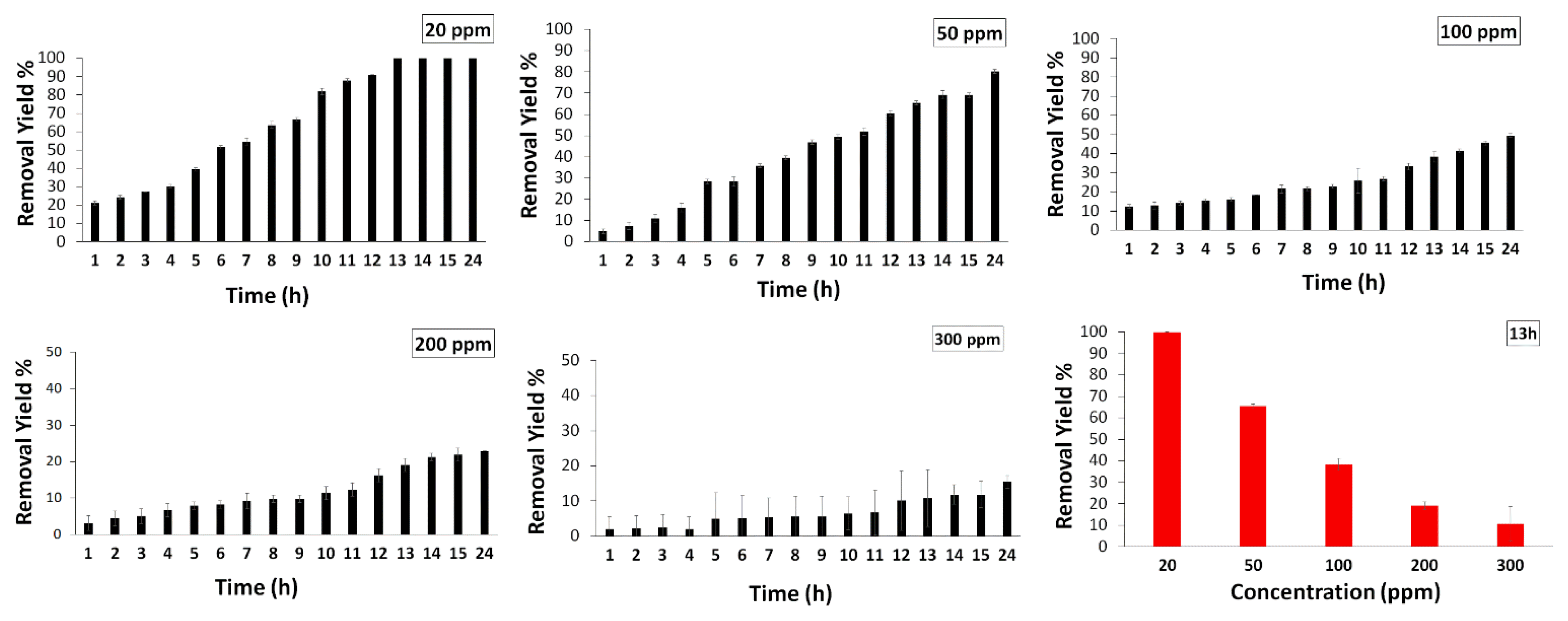
3.6. Dye Removal Capabilities
After determination of the optimum conditions, the effect of RB concentration on the removal by bacteria attached to PSU-NFM was investigated by using various contaminant concentrations. The RB removal is greatly influenced by the initial concentration (Fig. 6). At high concentrations of the dye such as 300 ppm, dye is toxic to bacteria and limits the dye removal activity, so the yield of dye removal is low (10.7%). When the RB concentration was less than 100 mg/L, the decolorization rate was found to be up to 65% at 13 h and 80% (50 ppm) and 100% (20 ppm) at 24 h. The highest removal efficiency 100% of RB was recorded at 20 ppm after 13 hours. The removal efficiency was 65.4%, 38.2%, 19.05%, and 10.75%, for the initial RB concentration of 50 mg/L, 100 mg/L, 200 mg/L, and 300 mg/L after 13 hours, respectively.
3.7. Long-term Storage and Reusability Results of PSU-NFMs for Dye Removal
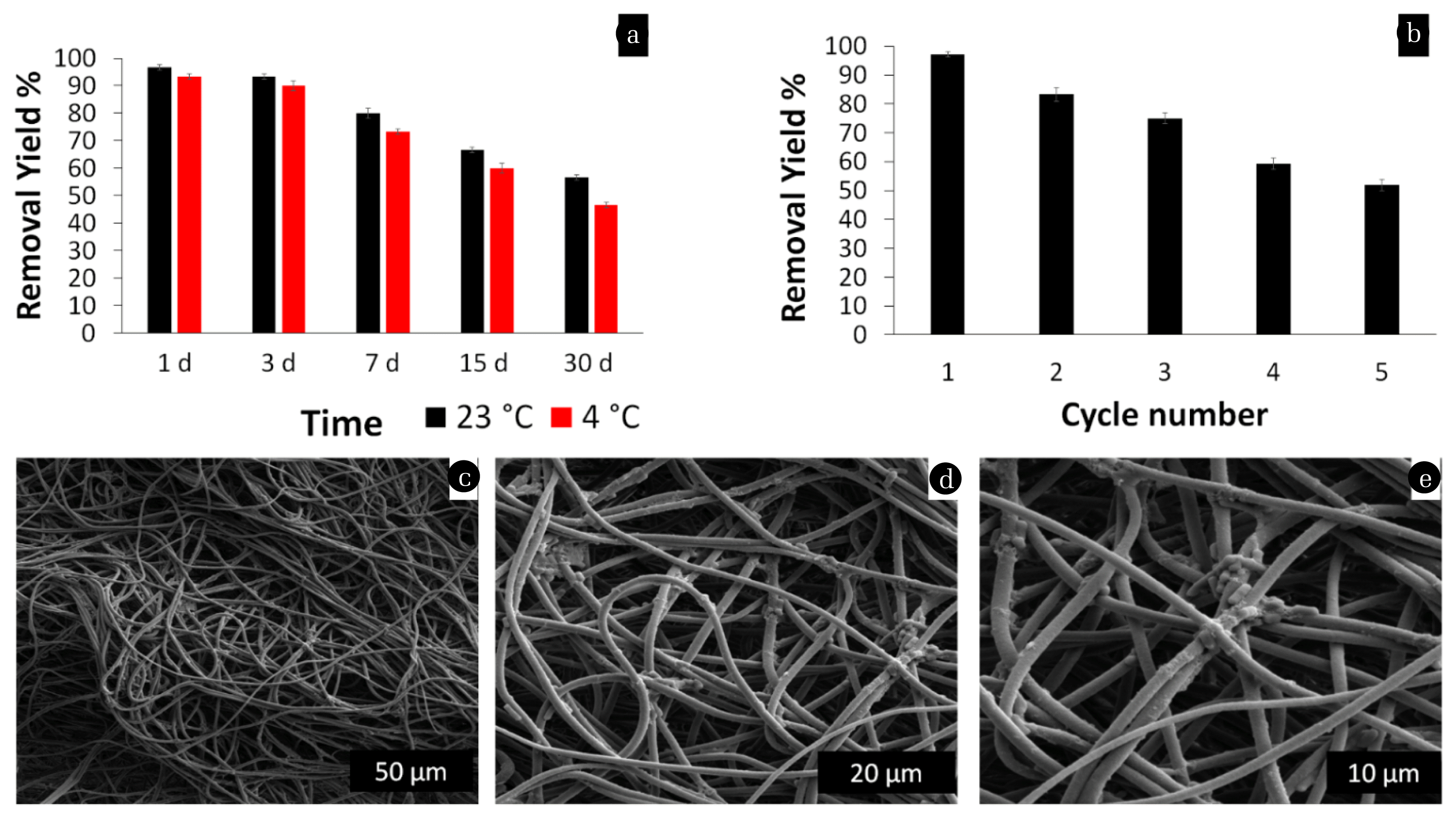
Another one of the superiorities of the attachment strategy is storage stability, which is necessary to check its reusability important for real applications. To determine the storage stability of bacteria attached to PSU-NFMs at 23°C and + 4°C, removal measurements have been performed for 30 days. Fig. 7a showed that after one week, bacteria attached to PSU-NFM retained more than 80% and 70% removal yield at 23°C and + 4°C respectively. Furthermore, bacteria attached to PSU-NFM retained more than 55% and 46.6% of removal activity after storage for 30 days at 23°C and +4°C respectively. The high removal yield after storage shows that attachment of bacteria on nanofiber mat is a useful strategy to increase their operational performance in large-scale wastewater treatment applications. The reusability of the materials as well as its removal capacity is very important. The performance for seven successive cycles is shown in Fig. 7b. It was clearly apparent that as the reused cycles were increased the removal yield tended to be decreased from 97.2% to 51.8% for 5 cycles. Thus, for the practical purpose, the nano bio-composite could be a good adsorbent for the wastewater treatment containing reactive dyes. At the end, bacteria attached to PSU-NFM were characterized by SEM in order to observe morphological changes due to reusability process. After reusability, the fiber mat retained its shape and there were seen few numbers of attached bacteria on the nanofiber. Compare the results in the similar studies, Sarioglu et al. [21] prepared a bio-composite material by encapsulation of Pseudomonas aeruginosa within polyethylene oxide (PEO) and polyvinyl alcohol (PVA) electrospun nanofibrous membrane which has been applied in methylene blue (MB) dye removal. Due to water soluble properties of PEO and PVA, nanofibers were not reused.
4. Conclusions
In summary, we have developed PSU nanofiber mats via electrospinning and Cr(IV) and RB remediating Clavibacter michiganensis bacteria were attached on nanofibrous mats. Electrospun PSU-Nf was chosen as non-toxic, and biocompatible support for attachment of bacteria. The number of attached live bacteria were checked by counting and SEM imaging. The efficient Cr(VI) removal with 100% yield was achieved within 24 hours (10 ppm) with nano bio-composite mats. Furthermore, the highest removal efficiency 100% of RB was recorded at 20 ppm after 13 hours. The prepared bacteria attached PSU-Nf mats showed storage stability at 23°C and 4°C with efficient removal yields. The reusability of bacteria attached to PSU-Nf mats were determined after five cycles for both pollutant, and 70.3% and 51.8% of the Cr(VI) and dye removal capacities were obtained, respectively. Results taken from this study are promising and therefore suggest that bacteria attached to PSU-NFMs could be applicable for the removal of different heavy metals and dyes because of their versatility, reusability and long-term storability.