AbstractToxicity assessment using microalgae adopted various endpoint measurements like mortality rate, photosynthetic activity, chlorophyll content, enzymatic activity, cell density, 14C assimilation, and phosphate uptake. These algal toxicity tests usually require 3–4 days of exposure time and laborious work to measure these endpoints parameters. In the present study, we described a simple and rapid toxicity assessment procedure using photosynthetic oxygen evolution as an endpoint measurement to determine heavy metal-induced toxicity. Oxygen evolution in gaseous phase was measured over a 12 h exposure time. The toxicity of six selected heavy metals was assessed. Concentrations of 1.02, 1.91, 0.46, 3.2, 7.5, and 65.6 mg/L were obtained as EC50 values for mercury, silver, cyanide, copper, cadmium, and hexavalent chromium, respectively, for microalgae (Chlorella vulgaris). Our results demonstrated that hexavalent chromium is less sensitive depending on the conditions (low exposure time, pH 6, and high initial biomass). Toxicities in decreasing order of strength were cyanide > mercury > silver > copper > cadmium > hexavalent chromium. Results obtained in the current and earlier studies demonstrate that photosynthetic oxygen evolution offers an alternative endpoint measurement to assess toxicity since it allows sensitive and rapid detection of heavy metal-induced toxicity in water.
1. IntroductionIntensive anthropogenic activities have increased heavy metal-induced toxicity in aquatic environments in the recent past [1]. Due to their high persistence, mobility, and toxicity, heavy metals are classified as one of the most hazardous environmental pollutants [1, 2]. Water contamination by heavy metals poses devastating threats to the ecological balance of the aquatic environment and ultimately extends their impacts to humans and other living organisms through bio-accumulation and bio-magnification [1–3]. The assessment of heavy metal-induced toxicity is a crucial part of environmental monitoring for the protection of water resources and all biota in the ecosystem [4].
The potential for toxicity from heavy metals in aquatic samples has been evaluated using sophisticated instrument techniques like ion chromatography (IC), high performance liquid chromatography (HPLC), and atomic absorption spectroscopy (AAS) [5]. These techniques are highly sensitive and accurate in the quantitative determination of heavy metals in water samples. However, they require skilled personnel, sample pretreatment, and expensive equipment. Moreover, these physiochemical techniques do not yield biochemical effects of heavy metals on living organisms [6]. Bioassays, however, measure changes in specific biochemical endpoints of various organisms when exposed to particular toxicants [7, 8]. Microbial bioassays have been frequently utilized in the eco-toxicological screening of contaminated water due to their ease of culturing and ability to detect a number of environmental pollutants [6–9].
Microalgae bioassays have been used as an essential means to assess the potential toxicity from toxicants as they act as bio-indicators in the aquatic ecosystem [9, 10]. Numerous environmental toxicity assessment studies have utilized microalgae due to their ecological significance, short growth time, rapid response, cost-effectiveness, and high sensitivity towards various environmental toxicants [11]. Conventional toxicity assessment using microalgae employs dry biomass, growth rate, chlorophyll content, mortality, and cell density as response endpoints [12–18]. However, most of the methods mentioned above require 48–96 h of exposure time in order to determine endpoint measurements and also involve a tedious procedure [19, 20]. Therefore, short term microalgae bioassays have been introduced by altering the endpoint parameter from growth rate to photosynthetic responses like carbon uptake, phosphate uptake, oxygen production, and fluorescence induction [21–26]. Although recent studies which employed photosynthetic responses as endpoint measurements have succeeded in the reduction of exposure times, endpoint measurement requires sophisticated instruments and laborious efforts [24, 27]. Therefore, there is a need for a rapid and simple endpoint measurement to eliminate the sophistication and the laboriousness involved in current assessment methods and shorten the time required for toxicity tests.
The present study aims to develop a rapid and straightforward method for the assessment of heavy metal-induced toxicity in water, using oxygen evolution as an endpoint measurement. The photosynthetic activity of microalgae (C. vulgaris) were quantified by measuring the photosynthetic oxygen evolution in the headspace of closed bioassay kits over 12 h of exposure time. Oxygen evolution by the photosynthetic activity of microalgae in the bioassay was measured using an oxygen sensor in gaseous phase. The current study investigated the effects of known concentrations of selected heavy metals (mercury, silver, cyanide, copper, cadmium, and hexavalent chromium) on the photosynthetic oxygen evolution of C. vulgaris. Furthermore, chlorophyll content and micrographs of algal bioassay were also determined for a better understanding in the current study. The concentration causing 50% inhibition of microalgae (EC50) obtained from the current study was compared with existing toxicity guideline values to determine the sensitivity of this protocol.
2. Materials and Methods2.1. Micro-algal Strain and CultivationThe freshwater microalga, Chlorella vulgaris, was acquired from the National Institute of Environmental Research (NIER), South Korea. The microalgae were cultivated in a 2-liter master culture photo bioreactor (MCPBR) prepared in the lab. Modified BG11 media (NaH2PO4 500 mg/L, Na2HPO4 100 mg/L, NaHCO3 500 mg/L, NaNO3 150 mg/L, MgSO4.7H2O 75 mg/L, CaCl2.2H2O 36 mg/L, citric acid 6 mg/L, ammonium ferric citrate green 6 mg/L, EDTANa2 1 mg/L) with minerals (H3BO3 2.86 mg/L, MnCl2.4H2O 1.81 mg/L, ZnSO4.7H2O 0.22 mg/L, Na2MoO4.2H2O 0.39 mg/L, CuSO4.5H2O 0.08 mg/L, and Co(NO3)2.6H2O 0.05 mg/L) were prepared and used for the cultivation of the microalgae. The MCPBR was operated in semi-continuous mode by wasting 1L of culture and feeding with 1L of fresh medium every 10 days. A continuous supply of 2% CO2 and 98% air ensured an adequate carbon source. A photoperiod of 16 h and a dark period of 8 h was maintained with an irradiance of 185 μmol.m−2.s−1 using LED lamps.
2.2 Microalgae Bioassay kit for Photosynthetic Oxygen Evolution MeasurementA schematic diagram (Fig. 1) shows the microalgae bioassay kit used in the present study. The kit consisted of a 25 mL glass vial with a 15 mL algal suspension and 9 mL headspace volume capped with Teflon-lined rubber stoppers and plastic caps. The microalgae cells were harvested in the late exponential phase from the MCPBR, and 1 mL of microalgae containing 0.05 g/L dry biomass (1.68 × 107 cells/mL) was added to 14 mL of medium containing toxic chemicals (Table 1). The medium for the kit test was composed of bicarbonate (NaHCO3 500 mg/L), phosphate buffer (NaH2PO4 500 mg/l + Na2HPO4 100 mg/L), nitrate (NaNO3 750 mg/L), and minerals (H3BO3 2.86 mg/L, MnCl2.4H2O 1.81 mg/L, ZnSO4.7H2O 0.22 mg/L, Na2MoO4.2H2O 0.39 mg/L, CuSO4.5H2O 0.08 mg/L, and Co(NO3)2.6H2O 0.05 mg/L). Nitrogen gas was purged in the headspace of the kit for 1–2 minutes to remove the initial oxygen in the headspace. After purging the nitrogen gas, gas-phase oxygen (%) was measured in the headspace using a needle-type oxygen sensor. The photosynthetic oxygen produced was measured after 12 h of incubation of the test kit which received continuous illumination while resting on an orbital shaker at 75 RPM and 25±1°C. All the toxicity tests were performed in triplicate with negative controls (without algae) to remove the dissolved phase oxygen in the liquid portion, which is subtracted from the final oxygen (%) in the controls and the remaining treatments. A summary of the test conditions is given in Table 1.
2.3. Chemicals and AnalysisIn the present study, mercury chloride (HgCl2), silver nitrate (AgNO3), potassium cyanide (KCN), copper sulfate (CuSO4), cadmium chloride (CdCl2), and potassium dichromate (K2Cr2O7) were used for the toxicity testing of mercury, silver, cyanide, copper, cadmium and hexavalent chromium, respectively. All the chemicals used in the current study were purchased from Sigma-Aldrich, St. Louis, MO, US and were of analytical grade.
Oxygen (%) in the headspace of the closed bioassay kits was measured using a fixed-needle type oxygen sensor (OXF500PT, Pyro-science sensor technology); the pH in all tests was measured using a portable pH meter (410A, Orion, Boston, MA, US). The relationship between dry algal biomass and optical density was correlated and calibrated for the MCPBR by collecting 10 mL at lag phase (zero time) and after every 24 h of growth for 10 days. Optical density (OD) was determined by measuring algal cultures at 680 nm by spectrophotometer; dry biomass was determined by a gravimetric filtration method [28]. For the kit test, the microalgal cells in the late exponential phase in the MCPBR were centrifuged at 3,000 ×g for 5 min, followed by washing with the fresh medium. The suspension was diluted to attain an initial concentration of 0.05 g/L by measuring optical density as a surrogate of dry biomass.
Light microscopic images of the algal bioassays were taken using a LEICA DM 2500 microscope equipped with a Leica DFC295 digital color camera. Algal cultures exposed to heavy metals for 12 h were examined for chlorophyll content. The chlorophyll content was determined by the solvent extraction method. Five mL of algal sample was centrifuged at 4,000×g for 5 min, 5 mL of 100% methanol was added to the pellet after removal of the supernatant, and the mixture was heated at 63°C for 3 min. The extract was then obtained after repeating centrifugation at 4,000×g for 5 min to remove cell debris. Absorbance was read at 665 and 650 nm on a UV spectrophotometer. The total Chl (Chl a + Chl b) content was calculated by using the following equations [29].
2.4. Statistical AnalysisInhibition % of mercury, silver, cyanide, copper, cadmium and hexavalent chromium for microalgae (C. vulgaris) were calculated by using the following Eq. (4).
Statistical analysis and curves were plotted using a sigma plot 10.0 and graph prism 5. The Hillslope method was used to determine the concentrations inhibiting 50% of the micro-algal population. One-way ANOVA analysis was performed to determine the significant differences between the control and other treatments (oxygen production with respect to the concentration of toxicants). The data has been given in supplementary data with critical values indicating statistical significance (Table S1~S6).
3. Results and Discussion
Fig. 1 illustrates the simple phenomena involved in the current study. We assessed the suitability of the present method and chose oxygen production as an indicator of heavy metals-induced toxicity. Photosynthesis is one of the physiological phenomena which play a vital role in the growth of microalgae. Therefore, the current bioassay, which deals with photosynthetic oxygen evolution, can serve as a tool in evaluating toxicity assessment. Initially, the oxygen evolution experiments were conducted for 12 and 24 h of incubation time. The results obtained at 12 h of exposure were not significantly different compared to 24 h of incubation (Fig. 2(a)). Accordingly, we chose 12 h of incubation time. The effect of silver on photosynthetic oxygen evolution over 24 h incubation is shown in Fig. 2(a). To check the reproducibility of the current method, silver toxicity was evaluated for 10 consecutive days. An average EC50 value of 1.9 mg/L was obtained over 10 days of repetition (Fig. 2(b)). The reproducibility of obtained EC50 values of silver were good with a coefficient of variance (CV) below 22% for silver.
3.1. Toxicity of Mercury
Fig. 3(a) illustrates the effect of mercury on photosynthetic oxygen production. The initial oxygen percentage (%) in all treatments, including the control, was zero. After 12 h incubation, oxygen evolution in the control was observed to be 21.03%, while in the treatments with 0.5, 1, 2.5, 5, 10, and 25 mg/L concentration of mercury, the oxygen produced was 15.3, 8.6, 5.6, 0.7, and 0%, respectively (Fig. 3(a)). It can be deduced from the figures that the photosynthetic activity of C. vulgaris was highly inhibited by mercury. The introduction of 0.5–2.5 mg/L of mercury resulted in 23.3–71.1% inhibition of the photosynthetic activity of C. vulgaris (Fig. 5(a)). The 12 h EC50 of C. vulgaris for mercury was estimated to be 1.02 mg/L, which is comparable to or lower than those in previous studies. Table 2 compares our EC50 values to those in earlier micro-algal toxicity tests based on different species and endpoint measurements. For example, Rosko and Rachlin [30] and Li et al. [31] used C. vulgaris and C. pyreniodosa species for 792 h and 96 h growth inhibition toxicity tests for mercury and reported 1.03 and 4.83 mg/L, respectively, for EC50 values. Although Wu et al. [20] observed a slightly lower EC50 value (0.22 mg/L) for mercury for the C. vulgaris species, the exposure time was very high (96 h) as compared to the current study (12 h). Mercury has been reported to be highly toxic to photosynthesis, including both dark and light reactions. In the light reaction, mercury affects ferredoxin-NADP-oxidoreductase and the photosystem I (PSI) reaction center reduces the activity of PSI [32]. Mercury also affects PSII by strongly binding with thiol groups in proteins in the acceptor and donor side of PSII. In PSII, mercury disturbs chloride binding and damages the oxygen evolution complex [33]. In the dark reaction, mercury inhibits the metabolic fixation of CO2 and the uptake of the carbon source. Singh et al. [34] reported that even at a very low concentrations, mercury inhibits the growth of algae by affecting its nutrient uptake and nitrogen metabolism. It inhibits various enzymatic activities and also induces oxidative stress, which ultimately alters the anti-oxidative defense mechanism.
Chlorophyll content was determined after 12 h of incubation to further examine the effects of mercury (Fig. 4(a)). At 2.5 mg/L concentration of mercury, the chlorophyll content decreased to 63.9% of the control. Mercury inhibited 71.7% of photosynthetic oxygen evolution at a concentration of 2.5 mg/L, which shows a similar trend of inhibition. The EC50 values on the basis of oxygen evolution and chlorophyll content for mercury were 1.02 and 1.15 mg/L, respectively, which are comparable (Fig. 5(a)). Elbaz et al. [35] reported that the chlorophyll content of Chlamydomonas reinhardtii decreased by about 58.6% of control after 96 h of exposure with a mercury concentration of 0.92 mg/L. Fig. 6 shows the optical micrographs of C. vulgaris in the current bioassay. The micrographs of treatment with mercury showed that most of the cells are dead, and agglomeration of cells was dominant due to the accumulation of the extracellular polymeric substance, which led to formation of aggregates when cells were in close proximity (Fig. 6(b)). Mercury significantly affected the morphology of microalgae by shrinking cell size as well as the formation of apoptotic bodies [36, 37]. It can be clearly seen in fig. 6b that due to the tearing of the cell walls of microalgae, apoptosis occurred. The breakage of the cell cytoskeleton and tearing of membranes resulted in the formation of apoptotic bodies and cell shrinkage.
3.2. Toxicity of SilverThe treatment with 0.5, 1, 2.5, 5, 10, and 25 mg/L of silver showed 14.8, 12.1, 8.5, 3.6, 2.4 and 0% of oxygen evolution, respectively, over 12 h of exposure time (Fig. 3(b)). Exposure of 0.5–2.5 mg/L of silver for 12 h resulted in 23.1–57.6% inhibition of photosynthetic activity of C. vulgaris (Fig. 5(b)). The 12 h EC50 value of C. vulgaris for silver in the current study was observed to be 1.9 mg/L, which is comparable or slightly higher than in previously reported studies (Table 2). For example, Ksiazyk et al. [38] and Ribeiro et al. [39] reported EC50 values of 1.6 and 0.03 mg/L for silver for Pseudokirchneriella subcapitata species over 72 h exposure time. Johari et al. [40] estimated the EC50 value to be 0.2–2.1 of the Dunaliella salina species for silver over 72 h time which is comparable to the present study. However, the earlier studies require long exposure times and a growth inhibition test was mostly employed to detect silver toxicity. Exposure of algal cells to silver might decrease its accessibility to light, resulting in inhibition of algal growth and reduction of nutrient uptake [41, 42]. Silver interacts directly with the surface of algal cells, resulting in the formation of cell aggregates, reactive oxygen species (ROS) and lipids peroxidation [40, 43]. The 12 h exposure of 2.5 mg/L silver led to inhibition of 57.3 and 57.6% chlorophyll content (Fig. 4(b)) and photosynthetic oxygen evolution, respectively, demonstrating a strong correlation between both endpoints (Fig 5b). Fig. 6(c) shows the effects of exposure of silver on C. vulgaris, which resulted in the aggregation of dead and injured cells. The cells in the control treatments are spherical shaped, intact, and relatively larger in size as compared to those exposed to silver (Fig. 6(a)). Silver breached the cell wall which damaged the cell membrane and caused loss of membrane integrity and cell lysis [44]. Abnormal morphology, especially shrinkage of cells and cytoskeleton rupture, was observed in silver treated cultures (Fig. 6(c)).
3.3. Toxicity of CyanideCyanide exhibited high toxicity among all tested heavy metals. Oxygen production of 8.6, 7.2, 1.4, and 0.8% were noted at 0.5, 1, 2.5, and 5 mg/L concentrations of cyanide, respectively (Fig. 3(c)). At 10 and 25 mg/L concentrations of cyanide, photosynthetic oxygen production was zero, which indicated 100% inhibition of photosynthesis. The impact of cyanide on the photosynthetic activity of C. vulgaris was significantly higher than for all other tested heavy metals used in the current study (Fig. 5(c)). Introduction of 0.5–2.5 mg/L of cyanide for 12 h resulted in approximately 57.1–93% inhibition of photosynthetic activity of C. vulgaris (Fig. 5(c)). A study by Manar et al. [45] investigating the inhibition of the P. subcapitata species of microalgae by cyanide found an EC50 value of 0.28 mg/L of at 72 h exposure time. Choi et al. [46] observed a high EC50 value (14.19 mg/L) with the same species (P. subcapitata) but the endpoint measurement was suppression of Fv/Fm over 1.2 h exposure time. Although the toxicity detection time was low, the observed EC50 value was higher than in the present study. Prior research has reported that cyanide may directly inhibit the cytochrome oxidase enzyme in electron transport, thereby affecting PSII; thus, chlorophyll content is inhibited [47]. Cyanide may directly impact chloroplasts via inhibition of the photosynthesis enzyme Rubisco, as Rubisco is crucial in photosynthesis [46]. Exposure to 2.5 mg/L cyanide resulted in inhibition of 66.3% of chlorophyll content and 93% photosynthetic activity (Fig. 5(c)). A low EC50 value indicates that the photosynthetic activity of C. vulgaris was more readily inhibited by cyanide, suggesting a higher sensitivity for the toxicity detection endpoint. The micrograph of C. vulgaris after 12 h of exposure to cyanide demonstrated that the aggregation of cells occurred due to the rupture of cellular membranes (Fig. 6(d)). Liu et al. [48] reported that cyanide may destroy the integrity of the cell membrane resulting in the formation of clusters of dead cells. Fig. 6d illustrates the formation of clumps of cells due to the tearing of membranes. Shrinking of cells led to deformation of cellular shape and reduction of cell size compared to the control (Fig. 6(d)).
3.4. Toxicity of CopperMonitoring copper toxicity in the current study proved to be least sensitive compared to that of mercury, silver and cyanide. Exposure to 2.5 mg/L copper for 12 h showed 44% inhibition of photosynthetic activity of C. vulgaris (Fig. 5(d)). The 12 h EC50 value of C. vulgaris for copper in the current study was estimated to be 3.2 mg/L which is substantially higher than in previous studies. Table 2 compares our EC50 values to those obtained from earlier studies with varying endpoints and exposure times. Lukavsky et al. [19] found EC50 values of 1.86, 8.54, 1.24, 3.10, and 0.62 mg/L of copper on the basis of growth inhibition of Scenedesmus quadricauda, Chlorella kessleri, Scenedesmus subspicatus, and Raphidocelis subcapitata species of microalgae, respectively, for 322–658 h exposure time. The sensitivity of earlier studies varied according to the species. A few studies have lower EC50 values compared to our study, but the exposure times for these earlier toxicity tests were considerably longer (96 h or more). Juneau et al. [49] reported a lower EC50 of 0.034 mg/L copper for C. reinhardtii species by measuring pulse-amplitude modulation (PAM); however, a long exposure time of 96 h was used. All algae require copper as a micronutrient for growth and reproduction; however, high concentrations of copper are very toxic to algae [50]. Copper affects photosynthesis by exerting its toxicity on subcellular organelles like chloroplasts [51]. In the current study, 44.1 and 44.4% inhibition of chlorophyll content and photosynthetic activity by 2.5 mg/L copper was observed, respectively. The EC50 values based on oxygen evolution and chlorophyll contents were 3.21 and 3.64 mg/L, respectively, which depict a strong correlation (Fig. 5(d)). Although earlier studies estimated low EC50 values for copper toxicity depending on various microalgae species and endpoints, the current method is more suitable due to its short detection time. The micrographs (Fig. 6(e)) show C. vulgaris exposed to copper, which resulted in the agglomeration of dead cells. Copper reduced the cell size of microalgae by 2.1 times as compared to the control, altered the cell shape due to the breaching of the cytoskeleton, and aggregated cells into clumps [52]. The breaching of the cytoskeleton could make the live and dead cells stick together when in close proximity. Reduction of cell size and formation of dead cells aggregates are obvious in copper treated cultures (Fig. 6(e)).
3.5. Toxicity of CadmiumThe photosynthetic activity of C. vulgaris was minimally affected by the introduction of low amounts of cadmium. Twelve h exposure to 2.5 mg/L of cadmium led to only 17% inhibition of C. vulgaris (Fig. 5(e)). An EC50 value of 7.5 mg/L of cadmium was estimated for C. vulgaris in the current study which is comparatively higher than in previous studies. EC50 values of 0.31, 1.10, 1.24, 0.46, and 0.31 mg/L for cadmium were estimated for the growth inhibition of Scenedesmus quadricauda, Chlorella Kessleri, Scenedesmus subspicatus, and Raphidocelis subcapitata species, respectively [19]. Juneau et al. [49] and Wu et al. [20] reported EC50 values of 0.05 and 0.13 mg/L for copper for the inhibition of PAM and growth of S. capricornutum and C. vulgaris species, respectively, over 96 h exposure time. A 96 h EC50 value of 10.5 mg/L for cadmium for Selenastrum capricornutum was stated by Bozeman et al. [53] using relative fluorescence as an endpoint measurement. Torres et al. [16] found an EC50 value of 22.39 mg/L for Phaeodactylum tricornutum over 96 h exposure time; Bascik-Remisiewicz et al. [54] obtained an EC50 value of 16.8 mg/L for Desmodesmus armatus after 24 h exposure time. Cadmium affects the photosynthesis and growth of microalgae by affecting the chloroplasts and vacuole compartments [55]. In the present study, exposure to 2.5 mg/L of cadmium caused 37.9% inhibition of the chlorophyll content of C. vulgaris which is slightly higher than the inhibition of photosynthetic activity at the same concentration (Fig. 5(e) Although there is a large variation in EC50 values for cadmium depending on the species, endpoint measurements and detection times, our method is suitable for detecting cadmium toxicity in water. Microscopic analyses of cadmium treated cultures showed morphological changes in terms of cell size (cell shrinkage) and aggregation of cells after 12 h exposure to C. vulgaris (Fig. 6(f)). Cadmium deformed the shape of microalgae by disrupting the cytoskeleton [56]. The primary target for cadmium-associated cytotoxicity is mitochondria which results in structural abnormalities [57]. Cadmium treated cultures demonstrated clustering of cells due to the accumulation of cellular polymeric substances released by cells through cell membrane lysis (Fig. 6(f)).
3.6. Toxicity of Hexavalent ChromiumThe impact of hexavalent chromium on the photosynthetic activity of C. vulgaris was insignificant as compared to all other tested heavy metals in the current study. The presence of 75 mg/L of hexavalent chromium resulted in the 12 h inhibition of only 59.3% of oxygen evolution. An EC50 value of 65.67 mg/L was estimated for hexavalent chromium, which is very high compared to other heavy metals tested in the present study (Fig. 6(f)). Earlier studies have reported 1.55, 8.95, 1.55, 1.70, and 1.55 mg/L of hexavalent chromium EC50 values for Scenedesmus quadricauda, Chlorella kessleri, Scenedesmus subspicatus, and Raphidocelis subcapitata species, respectively, by measuring growth inhibition as an endpoint over 322–658 h exposure time [19]. Kviderova et al. [17], Wu et al. [20], and Blinova [58] reported 0.64, 0.11, and 19.5 mg/L EC50 values for hexavalent chromium for C. kessleri, C. vulgaris, and Selenastrum capricornutum species, respectively, over 96 h exposure by measuring florescence and growth inhibition as endpoints. The 12 h chlorophyll content inhibition was observed to be 8.4% at 2.5 mg/L of hexavalent chromium (Fig. 4(f)). Although photosynthetic oxygen evolution was not decreased significantly, chlorophyll content showed significant inhibition (50% inhibition at 5.01 mg/L concentration of hexavalent chromium). The micrograph illustrates that hexavalent chromium induced morphological changes by formation of aggregates due to membrane breaching (Fig. 6(g)). Rocchetta et al. [59] reported that significant disorganization of chloroplast thylakoids and the presence of several vacuoles were observed in cultures treated with high concentrations of hexavalent chromium.
The EC50 values for hexavalent chromium obtained in the present study for photosynthetic oxygen evolution are high compared to those measured based on various other endpoints reported in the literature, but the exposure times in those studies exceeded 24 h. We conclude that 12 h exposure of hexavalent chromium is insufficient to inhibit the oxygen evolution of C. vulgaris. Significant inhibition of photosynthesis may require a long exposure to hexavalent chromium. Zemeri et al. [60] reported that by measuring rapid kinetics and multiphase (OJIP), there were no significant effects observed on pigment-protein complexes associated with PSII over 24 h of exposure time with 10 mg/l concentration of hexavalent chromium, while algae with 48 h of exposure time showed significant inhibition of PSII. Another study revealed that 96 h of exposure of L. gibba (duckweed) to 58.8 mg/L of hexavalent chromium resulted in 90 % growth inhibition, while 29% and 17% inhibition of oxygen-evolving complex proteins (OEC 33 kDa and OCE 24 kDa, respectively) were observed [61]. The absorption spectra of P. tricornutum at less than 24 h of exposure did not show any change for chlorophyll a and c fluorescence; only high concentrations of hexavalent chromium (above 20 mg/L) slightly decreased chlorophyll c [62]. The OJIP parameters of fluorescence kinetics of induction curves of P. tricornutum exposed to 25 mg/l of hexavalent chromium after 24 h of incubation showed 13%, 28%, and 26% inhibition of maximum photochemical yield of PSII, activity of PSII, and φ Eo (quantum yield of electron transport of PSII), respectively [60]. Besides the exposure time, there are several other possible explanations for the low toxicity of hexavalent chromium. Kováčik et al. [63] reported that low pH (less than 7) may induce reduction of Cr (VI) to Cr (III), which is less toxic to microalgae [64]. The initial biomass may also affect the sensitivity of toxicity tests by binding chromium to exudates [63, 65, 66]. The effects of prolonged exposure, pH, and initial biomass will be investigated in future research to better understand the inhibition of photosynthetic oxygen evolution by hexavalent chromium toxicity.
4. ConclusionsIn the present study, photosynthetic oxygen evolution by microalgae (C. vulgaris) as an endpoint measurement was tested for the rapid detection of heavy metal-induced toxicity. The data obtained demonstrated that photosynthetic oxygen evolution was considerably inhibited by the tested heavy metals. Correlation of EC50 values based on oxygen evolution and chlorophyll content confirmed that oxygen evolution may represent a promising method for the determination of selected heavy metals toxicity. Our results revealed that oxygen evolution is less sensitive to hexavalent chromium. We conclude from both the published literature and current study that the exposure times in toxicity testing for hexavalent chromium have been insufficient. The initial biomass of algae and pH of medium may also reduce the toxicity of hexavalent chromium. Toxicities in decreasing order of strength were cyanide > mercury > silver > copper > cadmium > hexavalent chromium. Results obtained in the current and earlier studies demonstrate that photosynthetic oxygen evolution is sensitive and offers an alternative endpoint measurement to assess toxicity, since it allows rapid detection of heavy metal-induced toxicity in water. Furthermore, the present method eliminates the sophistication and complexity of earlier toxicity determination methods especially the standard algal toxicity test, by reducing the exposure times and making it easy to identify the endpoint measurement. In standard algal toxicity tests, the initial and final biomass concentrations to determine toxicity are the result of methods which are quite onerous and require 3–4 days of incubation. In contrast, in the present study, initial and final oxygen percentages in the closed bioassay kit are measured to determine toxicity using a highly sensitive oxygen sensor. Our future research will focus on increasing the sensitivity of the current method by optimizing its conditions. Detailed studies of the effects of pH, initial biomass, and increased exposure times will be investigated for hexavalent chromium.
AcknowledgmentsThis work was supported by the Korea Ministry of Environment (MOE: 20190002000022) and by 2017 Research Grant from Kangwon National University(No. 520170217).
References1. Singh J, Kalamdhad AS. Effect of heavy metals on soil, plants human health and aquatic life. Int J Res Chem Environ. 2011;2:15–21.
2. Raza M, Hussain F, Lee JY, Shakoor MB, Kwon KD. Groundwater status in Pakistan: A review of contamination, health risks, and potential needs. Crit Rev Environ Sci Technol. 2017;47:1713–1762.
3. Shakoor MB, Nawaz R, Hussain F, et al. Human health implications, risk assessment and remediation of As-contaminated water: a critical review. Sci Total Environ. 2017;601:756–769.
4. Fu F, Wang Q. Removal of heavy metal ions from wastewaters: A review. J Environ Manage. 2011;92:407–418.
5. Van WA, Mons M, Delft WV. New methods to monitor emerging chemicals in the drinking water production chain. J Environ Monit. 2010;12:80–89.
6. Ahmed N, Oh SE. Toxicity assessment of selected heavy metals in water using a seven-chambered sulfur-oxidizing bacterial (SOB) bioassay reactor. Sens Actuators B Chem. 2018;258:1008–1014.
7. Oh SE, Hassan SH, Van Ginkel SW. A novel biosensor for detecting toxicity in water using sulfur-oxidizing bacteria. Sens Actuators B Chem. 2011;154:17–21.
8. Hassan SH, Van Ginke SW, Hussein Mohamed AM, Abskharond R, Oh SE. Toxicity assessment using different bioassays and microbial biosensors. Environ Int. 2016;92:106–118.
9. Rioboo C, Rrado R, Herrero C, Cid A. Population growth study of the rotifer Brachionus sp. fed with triazine-exposed microalgae. Aquat Toxicol. 2007;83:247–253.
10. Cho CW, Phame TPT, Jeon YC, et al. Microalgal photosynthetic activity measurement system for rapid toxicity assessment. Ecotoxicology. 2008;17:455–463.
11. Prado R, Garcia R, Rioboo C, et al. Comparison of the sensitivity of different toxicity test endpoints in a microalga exposed to the herbicide paraquat. Environ Int. 2009;35:240–247.
12. Ahmed H, Häder DP. Rapid ecotoxicological bioassay of nickel and cadmium using motility and photosynthetic parameters of Euglena gracilis. Environ Exp Bot. 2010;69:68–75.
13. Wells AS, Coombe VT. On the freshwater ecotoxicity and biodegradation properties of some common ionic liquids. Org Process Res Dev. 2006;10:794–798.
14. Chen CY, Lin JH. Toxicity of chlorophenols to Pseudokirchneriella subcapitata under air-tight test environment. Chemosphere. 2006;62:503–509.
15. Li X, Ping X, Xiumei S, Zhenbin W, liqiang X. Toxicity of cypermethrin on growth, pigments, and superoxide dismutase of Scenedesmus obliquus. Ecotox Environ safe. 2005;60:188–192.
16. Torres E, Cid A, Herrero C, Abalde J. Effect of cadmium on growth, ATP content, carbon fixation and ultrastructure in the marine diatom Phaeodactylum tricornutum Bohlin. Water Air Soil Pollut. 2000;117:1–14.
17. Kvíderová J. Rapid algal toxicity assay using variable chlorophyll fluorescence for Chlorella kessleri (Chlorophyta). Environ Toxicol. 2010;25:554–563.
18. OECD. Algal growth inhibition test. OECD guideline fortesting of chemicals 201. Paris: OECD; 1984.
19. Lukavský J, Furnadjieva S, Cepák V. Toxicity of metals, Al, Cd, Co, Cr, Cu, Fe, Ni, Pb and Zn on microalgae, using microplate bioassay 1: Chlorella kessleri, Scenedesmus quadricauda, Sc. subspicatus and Raphidocelis subcapitata (Selenastrum capricornutum). Algol Stud. 2003;110:127–141.
20. Wu MN, Wang XC, Ma XY. Phytotoxicity comparison of organic contaminants and heavy metals using Chlorella vulgaris. Desalin Water Treat. 2016;57:20809–20816.
21. Franklin NM, stauber JL, Apte SC, Lim RP. Effect of initial cell density on the bioavailability and toxicity of copper in microalgal bioassays. Environ Toxicol Chemistry. 2002;21:742–751.
22. Katsumata M, Ikushima Y, Bennett K, et al. Validation of rapid algal bioassay using delayed fluorescence in an interlaboratory ring study. Sci Total Environ. 2017;605:842–851.
23. Kaneko H, Shimada A, Hirayama K. Short-term algal toxicity test based on phosphate uptake. Water Res. 2004;38:2173–2177.
24. Latała A, Stepnowski P, Nedzi M, Mrozik W. Marine toxicity assessment of imidazolium ionic liquids: Acute effects on the Baltic algae Oocystis submarina and Cyclotella meneghiniana. Aquat Toxicol. 2005;73:91–98.
25. Ma J, Chen J. How to accurately assay the algal toxicity of pesticides with low water solubility. Environ Pollut. 2005;136:267–273.
26. Durrieu C, Tran-Minh C. Optical Algal Biosensor using Alkaline Phosphatase for Determination of Heavy Metals. Ecotoxicol Environ Safe. 2002;51:206–209.
27. Lin JH, Kao WC, Tsai KP, Chen CY. A novel algal toxicity testing technique for assessing the toxicity of both metallic and organic toxicants. Water Res. 2005;39:1869–1877.
28. Griffiths MJ, Garcin C, Hille RPV, Harrison STL. Interference by pigment in the estimation of microalgal biomass concentration by optical density. J Microbiol Methods. 2011;85:119–123.
29. Lee YK, Chen W, Shen H, et al. Basic culturing and analytical measurement techniques, Handbook of microalgal culture: applied phycology and biotechnology. John Wiley & Sons, Ltd GBR. 2013;12:37–68.
30. Rosko JJ, Rachlin JW. The effect of cadium, copper, mercury, zinc and lead on cell division, growth, and chlorophyll a content of the chlorophyte Chlorella vulgaris. Bull Torrey Bot Club. 1977;226–233.
31. Li M, Chengyan W, Xiaojie P, et al. Acute toxic effects of zinc, cadmium, and mercury on the growths of three unicellular green microalgae with relatively high initial densities. Fresenius Environ Bull. 2012;21:1349–1356.
32. Lu CM, Chau CW, Zhang JH. Acute toxicity of excess mercury on the photosynthetic performance of cyanobacterium, S. platensis – assessment by chlorophyll fluorescence analysis. Chemosphere. 2000;41:191–196.
33. Deng C, Zhang D, Pan X, Chang F, Wang S. Toxic effects of mercury on PSI and PSII activities, membrane potential and transthylakoid proton gradient in Microsorium pteropus. J Photoch Photobio B. 2013;127:1–7.
34. Singh R, Dubey G, Singh VP, et al. High light intensity augments mercury toxicity in cyanobacterium Nostoc muscorum. Biol Trace Elem Res. 2012;149:262–272.
35. Elbaz A, Wei YY, Meng Q, Zheng Q, Yang ZM. Mercury-induced oxidative stress and impact on antioxidant enzymes in Chlamydomonas reinhardtii. Ecotoxicology. 2010;19:1285–1293.
36. Hazlina AZ, Devanthiran L, Fatimah H. Morphological changes and DNA damage in Chlorella vulgaris (UMT-M1) induced by Hg2+
. Malays Appl Biol. 2019;48:27–33.
37. Krammer PH, Arnold R, Lavrik LN. Life and death in peripheral T cells. Nature Reviews Immunology. 2007;7:532–542.
38. Książyk M, Asztemborska M, Steborowski R, Bystrzejewska-piotrowska G. Toxic Effect of Silver and Platinum Nanoparticles Toward the Freshwater Microalga Pseudokirchneriella subcapitata. Bull Environ Contam Toxicol. 2015;94:554–558.
39. Ribeiro F, Gallego-Urrea JA, Jurkschat K, et al. Silver nanoparticles and silver nitrate induce high toxicity to Pseudokirchneriella subcapitata, Daphnia magna and Danio rerio. Sci Total Environ. 2014;466–467:232–241.
40. Johari SA, Sarkheil M, Tayemeh MB, Veisi S. Influence of salinity on the toxicity of silver nanoparticles (AgNPs) and silver nitrate (AgNO3) in halophilic microalgae, Dunaliella salina. Chemosphere. 2018;209:156–162.
41. Wei C, Zhang Y, Guo J, et al. Effects of silica nanoparticles on growth and photosynthetic pigment contents of Scenedesmus obliquus. J Environ Sci. 2010;22:155–160.
42. Perreault F, Bogdan N, Morin M, Claverie J, Popovic R. Interaction of gold nanoglycodendrimers with algal cells (Chlamydomonas reinhardtii) and their effect on physiological processes. Nanotoxicology. 2012;6:109–120.
43. Oukarroum A, Bras S, Rerreault F, Popovic R. Inhibitory effects of silver nanoparticles in two green algae, Chlorella vulgaris and Dunaliella tertiolecta. Ecotoxicol Environ safe. 2012;78:80–85.
44. Taylor C, Matzke M, Kroll A, Read DS, Svendsen C, Crossley A. Toxic interactions of different silver forms with freshwater green algae and cyanobacteria and their effects on mechanistic endpoints and the production of extracellular polymeric substances. Environ Sci: Nano. 2016;3:396–408.
45. Manar R, Bonnard M, Rast C, Veber AM, Vasseur P. Ecotoxicity of cyanide complexes in industrially contaminated soils. J Hazard Mater. 2011;197:369–377.
46. Choi CJ, Berges JA, Young EB. Rapid effects of diverse toxic water pollutants on chlorophyll a fluorescence: Variable responses among freshwater microalgae. Water Res. 2012;46:2615–2626.
47. Holmes JJ, Weger HG, Turpin DH. Chlorophyll a fluorescence predicts total photosynthetic electron flow to CO2 or NO3−/NO2− under transient conditions. Plant Physiol. 1989;91:331–337.
48. Liu Q, Zhang G, Ding J, Zou H, Shi H, Huang C. Evaluation of the removal of potassium cyanide and its toxicity in green algae (Chlorella vulgaris). Bull Environ Contam toxicol. 2018;100:228–233.
49. Juneau P, Berdey AE, Popovic R. PAM fluorometry in the determination of the sensitivity of Chlorella vulgaris, Selenastrum capricornutum, and Chlamydomonas reinhardtii to copper. Arch Environ Contam Toxicol. 2002;42:155–164.
50. Guanzon NG, Nakahara H, Yoshida Y. Inhibitory effects of heavy metals on growth and photosynthesis of three freshwater microalgae. Fish Sci. 1994;60:379–384.
51. Cid A, Herrero C, Torres E, Abalde J. Copper toxicity on the marine microalga Phaeodactylum tricornutum: effects on photosynthesis and related parameters. Aquat Toxicol. 1995;31:165–174.
52. Markina ZV, Aizdaicher NA. Content of photosynthetic pigments, growth, and cell size of microalga Phaeodactylum tricornutum in the copper-polluted environment. Russ J Plant Physl. 2006;53:305–309.
53. Bozeman J, Koopman B, Bitton G. Toxicity testing using immobilized algae. Aquat Toxicol. 1989;14:345–352.
54. Báscik-Remisiewicz A, Tomaszewska E, Labuda K, Tukaj Z. The Effect of Zn and Mn on the Toxicity of Cd to the Green Microalga Desmodesmus armatus Cultured at Ambient and Elevated (2%) CO 2 Concentrations. Pol J Environ Stud. 2009;18:
55. Tukaj Z, Bascik-Remisiewicz A, Skowronski T, Tukaj C. Cadmium effect on the growth, photosynthesis, ultrastructure and phytochelatin content of green microalga Scenedesmus armatus: A study at low and elevated CO2 concentration. Environ Exp Bot. 2007;60:291–299.
56. Přibyl P, Cepák V, Zachleder V. Cytoskeletal alterations in interphase cells of the green alga Spirogyra decimina in response to heavy metals exposure: I. The effect of cadmium. Protoplasma. 2005;226:231–240.
57. Silverberg BA. Cadmium-induced ultrastructural changes in mitochondria of freshwater green algae. Phycologia. 1976;15:155–159.
58. Blinova I. Use of freshwater algae and duckweeds for phytotoxicity testing. Environ Toxicol. 2004;19:425–428.
59. Rocchetta I, Mazzuca M, Conforti V, Ruiz L, Balzaretti V, de Molina MD. Effect of chromium on the fatty acid composition of two strains of Euglena gracilis. Environ Pollut. 2006;141:353–358.
60. Khalida Z, Youcef A, Zitouni B, Mohammed Z, Radovan P. Use of chlorophyll fluorescence to evaluate the effect of chromium on activity photosystem II at the alga Scenedesmus obliquus. Int J Res Rev Appl Sci. 2012;12:304–314.
61. Ali NA, Dewez D, Didur O, Popovic R. Inhibition of photosystem II photochemistry by Cr is caused by the alteration of both D1 protein and oxygen evolving complex. Photosynth Res. 2006;89:81–87.
62. Gabbasova DT, Matorin DN, Konyukhov IV, Seifullina NK, Zayadan BK. Effect of chromate ions on marine microalgae Phaeodactylum tricornutum. Microbiology. 2017;86:64–72.
63. Kováčik J, Babula P, Hedbavny J, Kryštofová O, Provaznik I. Physiology and methodology of chromium toxicity using alga Scenedesmus quadricauda as model object. Chemosphere. 2015;120:23–30.
64. Pakshirajan K, Worku AN, Acheampong MA, Lubberding HJ, Lens PN. Cr (III) and Cr (VI) removal from aqueous solutions by cheaply available fruit waste and algal biomass. App Biochem Biotech. 2013;170:498–513.
65. Nacorda JO, Martinez-Goss MR, Torreta NK. Bioremoval and bioreduction of chromium (VI) by the green microalga, Chlorella vulgaris Beij., isolated from Laguna de Bay, Philippines. Philip J Sci. 2010;139:181–188.
Fig. 2Inhibition of photosynthetic oxygen evolution over 24 h of incubation in closed bioassay kit with varying concentrations of silver (a). The vertical bars represent standard deviation (n = 3). The 12 h effective concentration (EC50) based on photosynthetic oxygen for silver in a closed bioassay kit over 10 days of repetition (b). The average EC50 with mean ± 1.5SD and mean ± 2SD are shown in the figure. 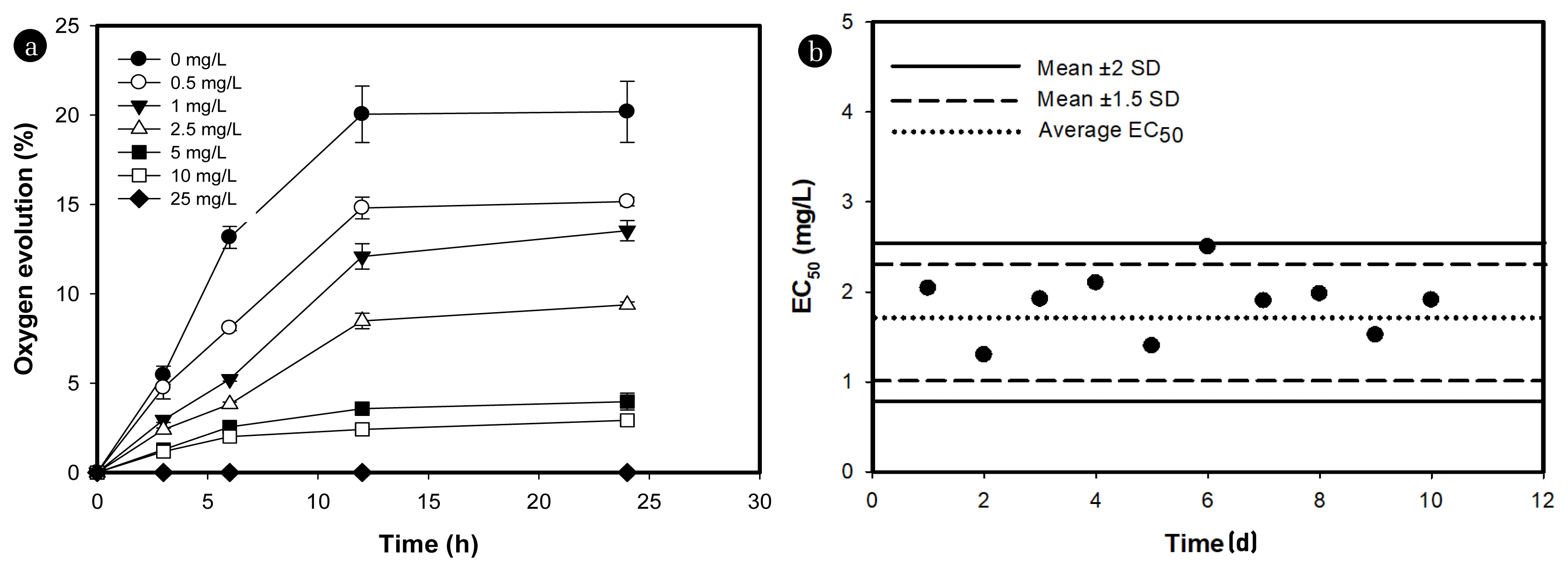
Fig. 3Inhibition of photosynthetic oxygen evolution in closed bioassay kits with varying concentrations of mercury (a), silver (b), cyanide (c), copper (d), cadmium (e), and hexavalent chromium (f) over 12 h incubation. The vertical bars represent standard deviation (n = 3). 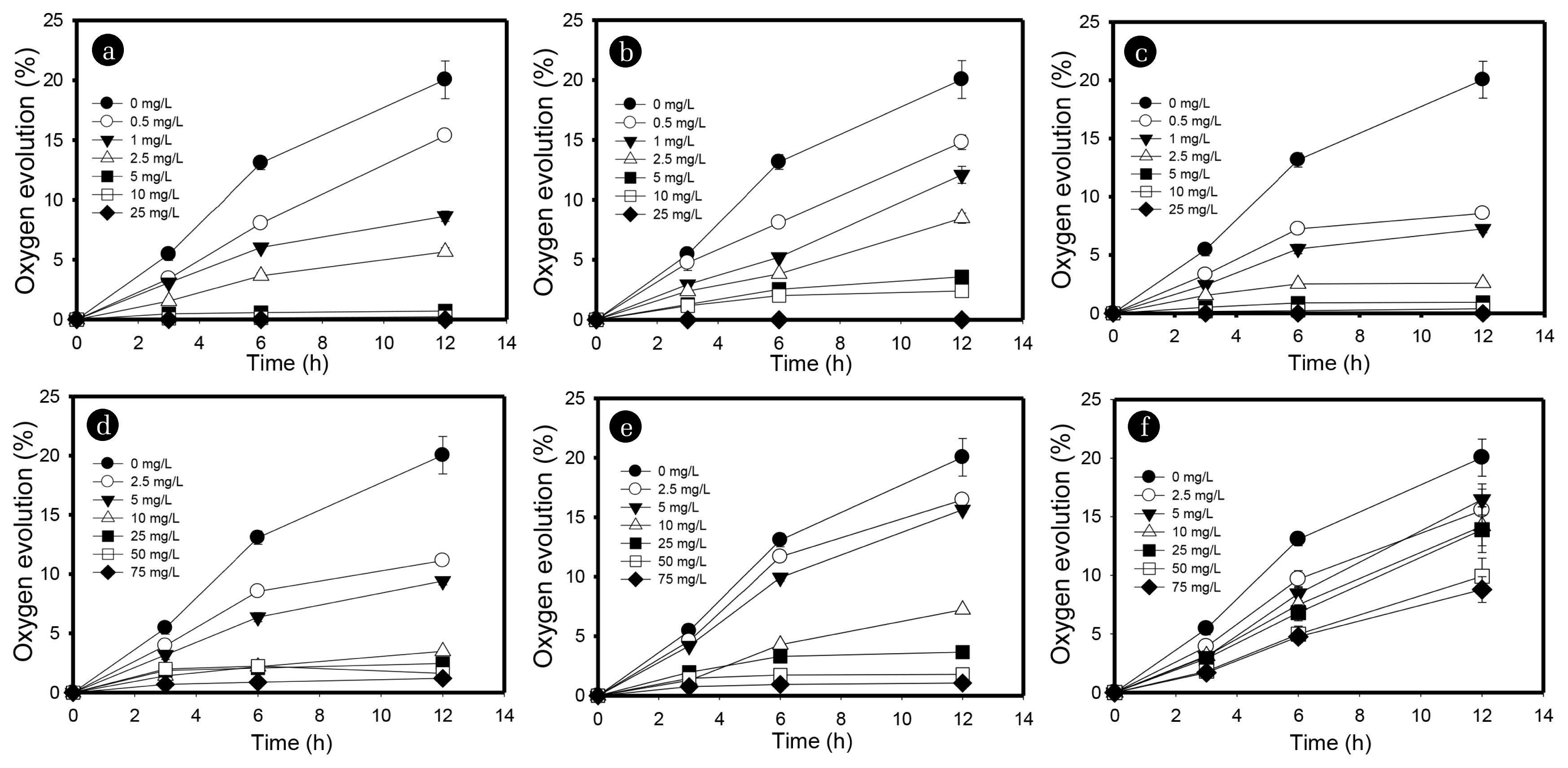
Fig. 4Effects of mercury (a), silver (b), cyanide (c), copper (d), cadmium (e), and hexavalent chromium (f) on the inhibition of chlorophyll content of C. vulgaris over 12 h incubation. The vertical bars represent standard deviation (n = 3). 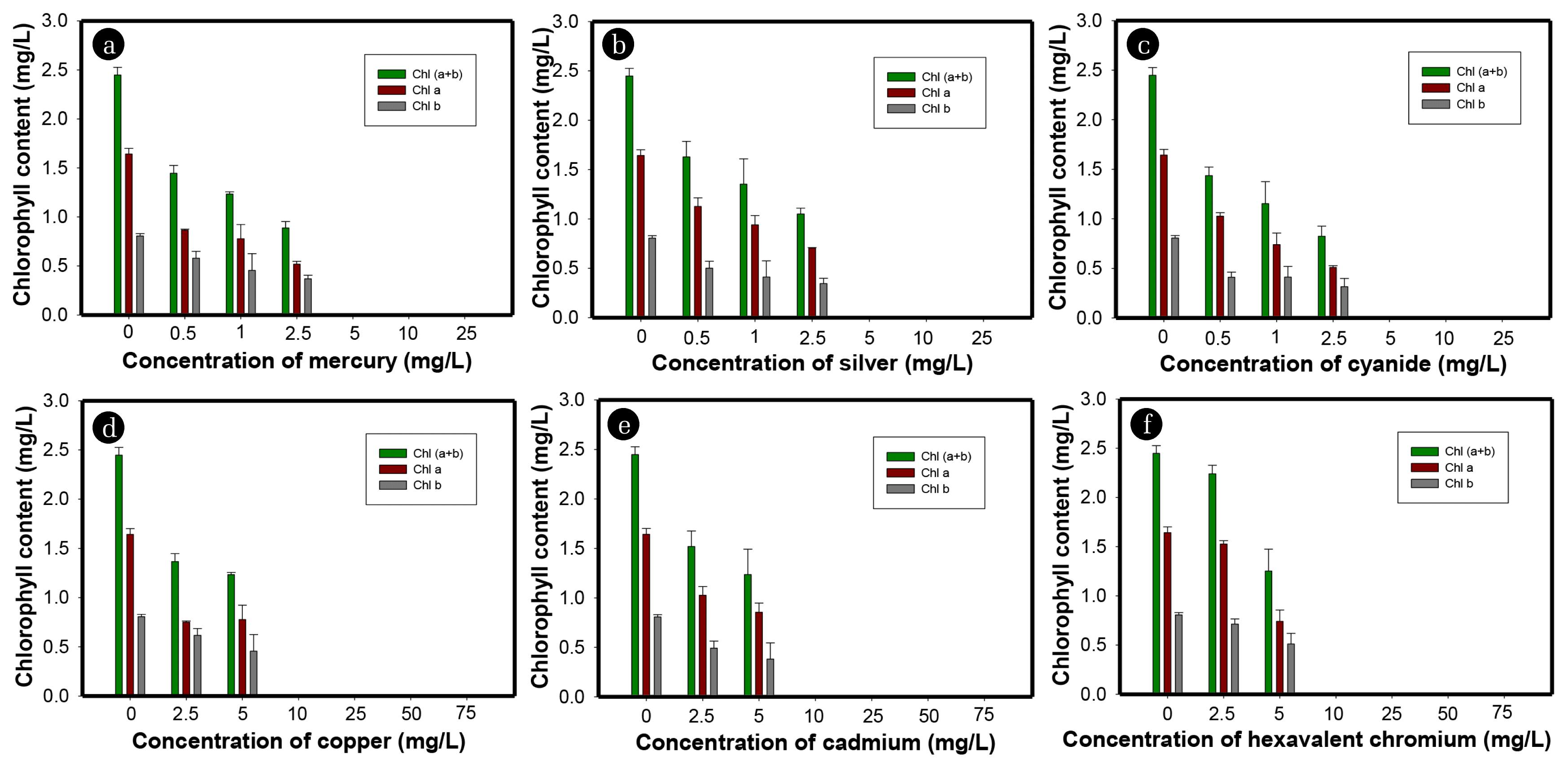
Fig. 5Dose-response curve showing effective concentration at which 50% of the population is inhibited on the basis of oxygen evolution and chlorophyll content in closed bioassay kits with varying concentrations of mercury (a), silver (b), cyanide (c), copper (d), cadmium (e), and hexavalent chromium (f) over 12 h incubation. 
Fig. 6Optical micrographs of C. vulgaris treated with heavy metals for 12 h. Control (a), mercury (b), silver (c), cyanide (d), copper (e), cadmium (f), and hexavalent chromium (g). Arrows indicate agglomerates of metals clumped with algal cells of C. vulgaris. Note aggregates in C. vulgaris cells. Bars, 10 μm; magnification, 100X. 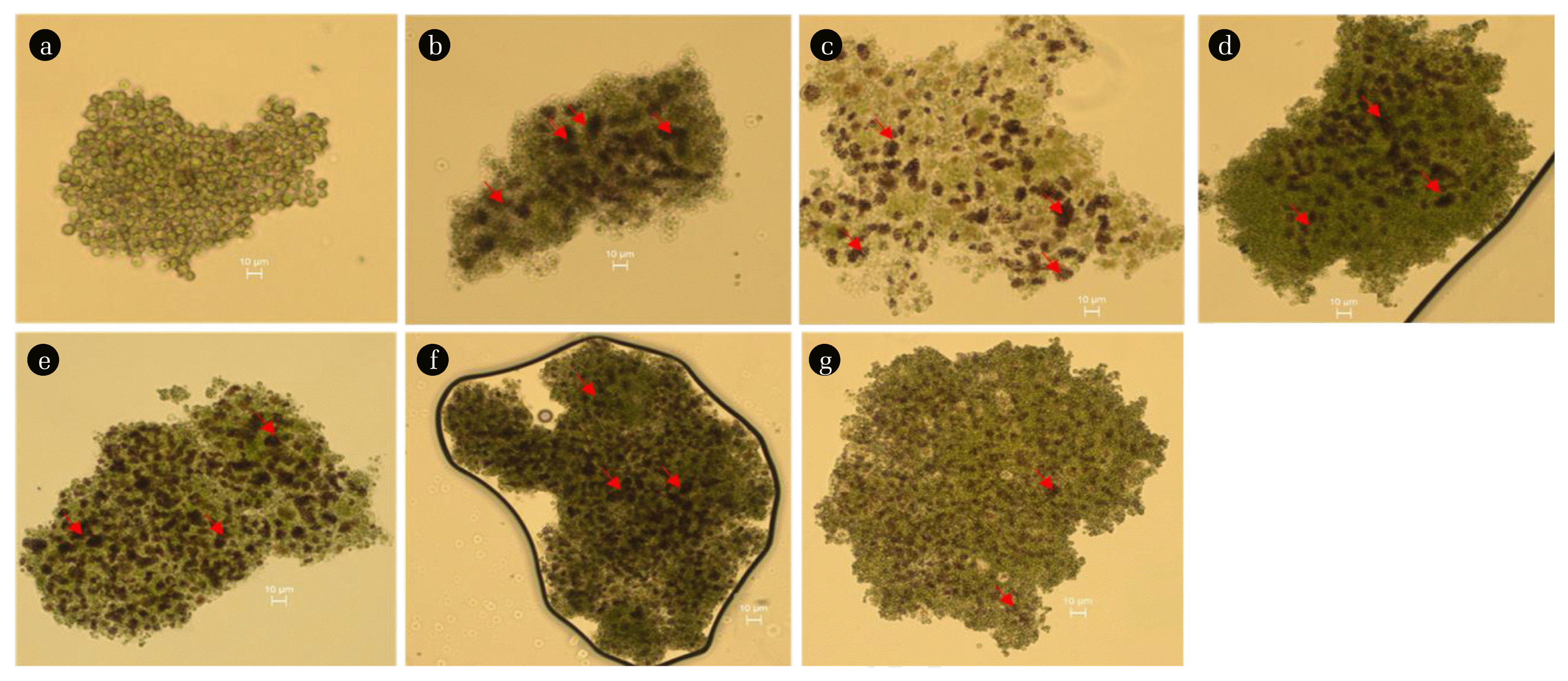
Table 1Summary of Test Conditions |
|
||||||||||||||||||||||||||||||||||||||||