1. Introduction
Bioremediation employs microorganisms to remove hydrocarbon contaminants [1, 2]. The indigenous microbial population plays an important role in biodegradation, but their performance usually declines when the concentration of pollutants is high [3, 4]. Therefore, engineered remediation trails such as biostimulation (BIOS; use of nutrient) and bioaugmentation (BIO; use of exogenous microbes) are sometimes used to meet the remediation time or goal [5–7]. The species of Acinetobacter, Pseudomonas, Rhodococcus, Alcanivorax, Bacillus, Stenotrophomonas, and Arthrobacter have been widely applied for the degradation of aliphatic hydrocarbons [8–10].
The components of petroleum hydrocarbons include volatile and non-volatile aromatic compounds and aliphatic fraction. Diesel oil is mainly composed of aliphatic hydrocarbons (> 90%) and lower levels of aromatic hydrocarbons (< 5%) [10]. Owing to the significant environmental hazard posed by aromatic components, most of the recent studies have focused mainly on these components [8–11]. However, addressing the removal of large quantities of aliphatic hydrocarbons deposited in the soil is important to achieve remediation goals. Furthermore, the mid-length aliphatic hydrocarbons of diesel oil are less biodegradable due to non-polar, non-volatile, water insoluble, and limited bioavailability properties [10, 11]. Recent studies have shown that the mid-length C18-C22 hydrocarbons of diesel oil in the unsaturated soil conditions are not readily degraded as compared to other aliphatic hydrocarbons [12].
This study focused on the biodegradation of diesel oil and selective n-alkanes (octadecane, C18; icosane, C20; and docosane, C22) in unsaturated soil. The three carbon fractions were monitored during the BIO and BIOS + BIO processes to ascertain how the carbon fractions change during the biodegradation. To achieve this, a newly isolated bacterium Acinetobacter sp. K-6 and inorganic nutrients were delivered into soil contaminated with diesel oil and C18, C20, and C22 hydrocarbons.
There have been no previous reports on the potential of Acinetobacter species in bioremediation of unsaturated soils contaminated with the specific and major residual fractions of C18, C20, and C22 hydrocarbons. The main objectives of the present work were (1) to isolate, identify, and characterize the petroleum-hydrocarbon degrading strain Acinetobacter sp. K-6, (2) to evaluate the biodegradability pattern and identify the most bio-recalcitrant aliphatic residual fraction of diesel oil, and (3) to evaluate the degradation of C18, C20, and C22 hydrocarbons using the newly isolated bacterial strain Acinetobacter sp. K-6.
2. Materials and Methods
2.1. Enrichment and Isolation of Bacteria
For isolation of the petroleum-hydrocarbon-degrading bacteria, oil-contaminated soil samples were collected from the US army area, Yongsan, South Korea (GPS coordinates: 37°31′59.99″N 126°58′59.99″E). The n-alkanes (C18, C20, and C22) were obtained from Sigma-Aldrich, USA. The composition of the mineral salt medium (MSM) used for the enrichment of bacterial cultures was similar to that described previously [13]. The soil sample was air-dried and then sieved using a mesh sieve (2 mm pore size). The sieved soils were used to prepare inoculums for the enrichment and isolation of bacterial strains.
For the enrichment cultivation, a modified culture technique using six-well polycarbonate Transwell plates was employed [14]. Five grams of oil-contaminated soil was added to each well of the Transwell plate and the inserts containing 1 mL soil inoculums, 3 mL MSM, and 2,400 ppm n-alkanes (800 ppm each of C18, C20, and C22 hydrocarbons) were placed on the soil surface. The soil inoculums were added in the insert for enrichment cultivation and isolation of effective bacterial strains. The plate was capped, sealed with tape, and incubated in a shaking incubator (100 rpm) at 28°C for four weeks. The MSM liquid media containing 1,200 ppm hydrocarbons was added periodically in the inserts to compensate for the loss due to evaporation. After four weeks of enrichment, 1 mL of enriched liquid culture from each insert was taken and serially diluted 1,000-fold. Each dilution of the enriched culture suspension (100 μL) was spread on MSM agar plates containing the above-mentioned hydrocarbons. The culture plates were incubated aerobically at 28°C for two weeks. The unique pure colonies were selected and subcultured in R2A agar (MB Cell, KisanBio, South Korea) [13]. All the pure bacterial isolates were assessed for hydrocarbon-degrading ability as described previously [15].
2.2. Bacterial Identification
Genomic DNA from the bacterial isolates was extracted using the DNA extraction kit (InstaGene Matrix kit, Bio-Rad, USA) following the manufacture’s protocol. PCR amplification of the 16S rRNA gene was performed using the primer set 27F and 1492R [13]. Purification of the PCR product was performed using MultiScreen PCR 96-well Filtration System (Millipore Corp., Bedford, MA, USA). The purified product was then sequenced using the 3730xL DNA Analyzer and the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems Inc., CA, USA). The nearly complete 16S rRNA gene sequence was then assembled with the SeqMan software (DNASTAR Inc., USA). The 16S rRNA gene sequences of the bacterial isolates were compared with the available 16S rRNA genes in the GenBank. The closest member was identified by uploading the sequences to the EzBioCloud server [16]. For phylogenetic study, the maximum-likelihood tree was generated using the MEGA6 program [17]. The strain Acinetobacter sp. K-6 was further characterised using the sequences of two housekeeping genes (rpoB and gyrB) and a catabolic gene (alkB). The primers and PCR cycling conditions for amplifying the rpoB, gyrB, and alkB genes were as described previously (Table S1) [15, 18].
The chemotaxonomic characterisation of Acinetobacter sp. K-6 was performed by analysing the fatty acid profile of the bacteria. The fatty acids were extracted using the standard MIDI protocol (Sherlock Microbial Identification System, version 6.0B), analysed by GC system (HP 6890 Series; Hewlett Packard), and identified utilizing the TSBA6 database [19]. The morphological, physiological, and biochemical studies of Acinetobacter sp. K-6 were performed as described previously [15] and by using the commercial test kits (API ZYM, API 20NE, and API ID 32GN; BioMérieux) according to the manufacturer’s instructions.
2.3. Screening for Degradation Ability and Growth-Tolerance
A qualitative analysis was conducted to determine the degradation potentiality and growth-tolerance of Acinetobacter sp. K-6 on various petroleum-derived hydrocarbons. The degradation ability was assessed in 50-mL capped bottles containing MSM liquid media (10 mL, pH 7.0) and Acinetobacter sp. K-6 culture (0.1 g L−1). These bottles were supplemented with 2,400 ppm of n-alkanes (800 ppm each of C18, C20, and C22 hydrocarbons) and 2,400 ppm of diesel oil (purchased from GS Caltex, Suwon, South Korea), and incubated at 28°C in a shaking incubator (120 rpm) for 14 d. After incubation, the residual hydrocarbons and diesel oil were extracted with dichloromethane (DCM; Wako Chemical Co., Osaka, Japan), and their concentrations were determined with GC-FID (HP 6890, Agilent, USA) as described previously [13].
The growth ability of Acinetobacter sp. K-6, in the presence of different hydrocarbons, was evaluated in 10 mL MSM liquid media containing 10 g L−1 each of glucose, dextrose, and mannitol, 0.5 g L−1 yeast extract, and 50 ppm each of benzene, toluene, and xylene. A single colony of Acinetobacter sp. K-6 was inoculated into each of the culture tubes and incubated at 28°C for 1–3 d. All the experiments were conducted in triplicate. The absorbance of the growing cells at 600 nm was measured at 12 h intervals for each culture using a UV-visible spectrophotometer (Biochrome Libra S4).
2.4. Bioremediation of Petroleum-Hydrocarbons in Soil
2.4.1. Preparation of contaminated soil
The soil used for the bioremediation experiment was prepared by mixing 80% (w/w) sand, 10% (w/w) kaolin, and 10% (w/w) peat moss. The physicochemical characteristics of the experimental soil are presented in Table S2. The soil texture was determined through the particle size distribution (sieve and sedimentation/ pipette method). The soil pH was measured using a pH meter (ThermoFisher Scientific, B30631). The loss-on-ignition technique was used to estimate the amount of organic matter present in the soil. The total iron content was estimated by atomic absorption spectroscopy (PerkinElmer Analyst 200, USA). The number of indigenous microflora was determined by serial dilution method and expressed as colony forming unit (CFU g−1). To prepare the aliphatic hydrocarbon-contaminated soil and diesel-contaminated soil, the selected n-alkanes (C18, C20, and C22 dissolved in DCM) and diesel oil at 5,500 mg kg−1 and 6,500 mg kg−1, respectively, were mixed thoroughly. Both the diesel and aliphatic hydrocarbon-contaminated soils were allowed to stabilize for three months before conducting the bioremediation study.
2.4.2. Experimental design for bioremediation of petroleum-hydrocarbons
The petroleum-hydrocarbon degradation was investigated separately in both the diesel and n-alkanes (C18, C20, and C22) contaminated soils. The experiment was set-up in a reactor container (size: 24 cm × 17 cm × 16 cm) each containing 2.3 kg of the prepared soil (Fig. S1). The soil was adjusted to a height of 5 cm. Hydrocarbon degradation was evaluated using four different microcosm conditions (Table 1). Due to the negligible number (102 CFU g−1) of indigenous microflora in the soil prepared in the laboratory, this study did not evaluate the effect of nutrients alone. But, this study had designed BIOS + BIO experiment to assess the effect of nutrient amendment combined with the BIO approach. A natural attenuation (NA) experiment was conducted to monitor the natural remediation of petroleum-hydrocarbons without the addition of the treatment reagents. The control (Ctrl) experiment was conducted with the addition of sterile water. The volume of water poured in the experimental container was same as the aqueous phase volume in the BIO experiment.
For the BIO experiment, the contaminated soil was treated with the bacterial strain Acinetobacter sp. K-6 (cell concentration: 8 × 106 CFU g−1). For the BIOS + BIO experiment, the contaminated soil was treated with two nutrients (NH4NO3 and KH2PO4), and the bacteria. The nutrients were prepared while maintaining the carbon, nitrogen, and phosphorous at the ratio of 100:10:1. In both of these experiments, all the treatment reagents (bacteria and nutrients) were delivered into the soil through the foam-spray technique. Here, the foam was generated by bubbling the microbe-surfactant mixture containing bacteria, nutrients, 0.1% alpha olefin sulfonate (AOS; AK Chemical Inc., Korea), and 0.7% gelatin (Der Grune Punkt, Germany) by air through a capillary tube at a flow rate of 250 cc/min [20]. All the experimental containers were incubated at 20°C for 30 d.
2.4.3. Analysis of residual hydrocarbons
To determine the concentration of residual hydrocarbons, the soil was sampled at 0, 5, 15, and 30 d from four different sites of each reactor container, and mixed to make a composite sample of 10 g. TPH analysis was performed by the Korean Soil Analysis Method, ES 07552.1a [20, 21]. The residual hydrocarbons were extracted by mixing 50 mL dichloromethane (DCM; Wako Chemical Co., Osaka, Japan) in 10 g of soil sample followed by sonication of the mixture in a sonicator (Sonics Vibra Cell, USA) for 10 min. The sonicated mixture was then centrifuged (3,000 rpm) for 15 min, and the resulting supernatant was filtered and collected. The pellet was again mixed with 25 mL of DCM and the process was repeated for a higher yield of residual hydrocarbons. The combined filtered supernatant was then evaporated at 60°C and re-extracted with DCM in 2-mL GC vial for GC analysis. The residual hydrocarbon concentration was analysed by injecting 2 μL sample in GC-FID (HP 6890, Agilent, USA), using an Ultra 2 capillary column [12, 13].
2.4.4. Bacterial enumeration in different soil microcosms
The number of viable bacteria in the soil of different microcosmic conditions was determined by the serial dilution technique. From each experimental condition, the soil suspension was prepared by mixing 1 g of soil, sampled at a 5 d interval, in 9 mL of R2A broth (KisanBio, Korea), and incubated at 20°C for 1 h in a shaking incubator (100 rpm). Then, this 1 mL soil suspension was used for serial dilution. One hundred microliters of each diluted sample was spread on R2A agar and incubated at 20°C for 48–72 h. The bacterial count was enumerated as CFU g−1 of dry soil.
2.5. Statistical Analyses
The data were subjected to statistical analyses to calculate the mean, standard deviation (SD), and standard error. The significant differences (p < 0.05) between the various treatments were determined using two-way ANOVA. All the statistical analyses were conducted using Microsoft Office Excel 2007 and OriginPro 8.5 software.
3. Results and Discussion
3.1. Isolation and Characterisation of Bacteria
With the enrichment cultivation technique, 21 bacterial strains were isolated from the oil-contaminated field soil which could utilise the petroleum-hydrocarbons as a sole carbon source for growth. The enrichment technique with the desired contaminants facilitates the selective isolation of the bacteria with biodegradation ability. This enrichment strategy simulates natural environments that provide greater adaptation, greater resistance, and excellent degradation potentiality to native microorganisms [14]. For successful bioremediation, it is essential to isolate effective strains that can survive and adapt in the contaminant environments. Therefore, the oil-contaminated soils are important source of petroleum-hydrocarbon degrading bacteria [2, 7]. The closest relative member and the hydrocarbon degradation efficiency of all the 21 isolates are provided in Table S3. The preliminary assessment of their remediation ability on hydrocarbon mixture showed the lowest (5.1%) degradation by the strain D-26 and the highest (85.4%) degradation by the strain K-6 (Table S3). Therefore, strain K-6 was further used in this study and has been deposited in the Korea Environmental Microorganisms Bank (culture collection number: KEMB 9005–643).
The 16S rRNA gene sequence (GenBank accession number: KY907005) and phylogenetic tree analysis showed that the isolated bacterial strain K-6 belonged to the genus Acinetobacter and is closely related to ‘Acinetobacter oleivorans DR1’ (99.86% sequence identity). The closest neighbours of the strain K-6 are depicted in the maximum-likelihood tree (Fig. S2). The rpoB and gyrB gene sequence analyses (GenBank accession numbers: MF405441 and MF405442, respectively) also revealed that the strain K-6 is closely clustered with the members of the genus Acinetobacter. The strain K-6 grew well on a wide range of commercial media namely, tryptone soya agar (Oxoid), R2A agar (MB Cell), nutrient agar (Oxoid), veal infusion agar (Difco), and Luria-Bertani agar (Oxoid). The growth was also observed in MSM containing kerosene, diesel, gasoline, benzene, toluene, xylene, and petroleum-hydrocarbons (C18, C20, and C22). Other morphological, biochemical, physiological, and chemotaxonomic properties of Acinetobacter sp. strain K-6 are summarised in Table 2.
There are several strains of the genus Acinetobacter isolated from various types of soil environments which have been reported as effective petroleum-hydrocarbon-degraders [15, 22, 23]. In the study conducted by Kang et al. [22], Acinetobacter oleivorans DR1, which was isolated from rice paddy soil, was able to grow in diesel-oil. Dahal et al. [15] isolated Acinetobacter halotolerans R160T from reclaimed grassland soil and was able to degrade a mixture of hydrocarbon and diesel oil. Therefore, isolating the strains of Acinetobacter from soils and utilizing them in bioremediation study has great significance.
3.2. Growth and Degradation Ability of Acinetobacter sp. Strain K-6
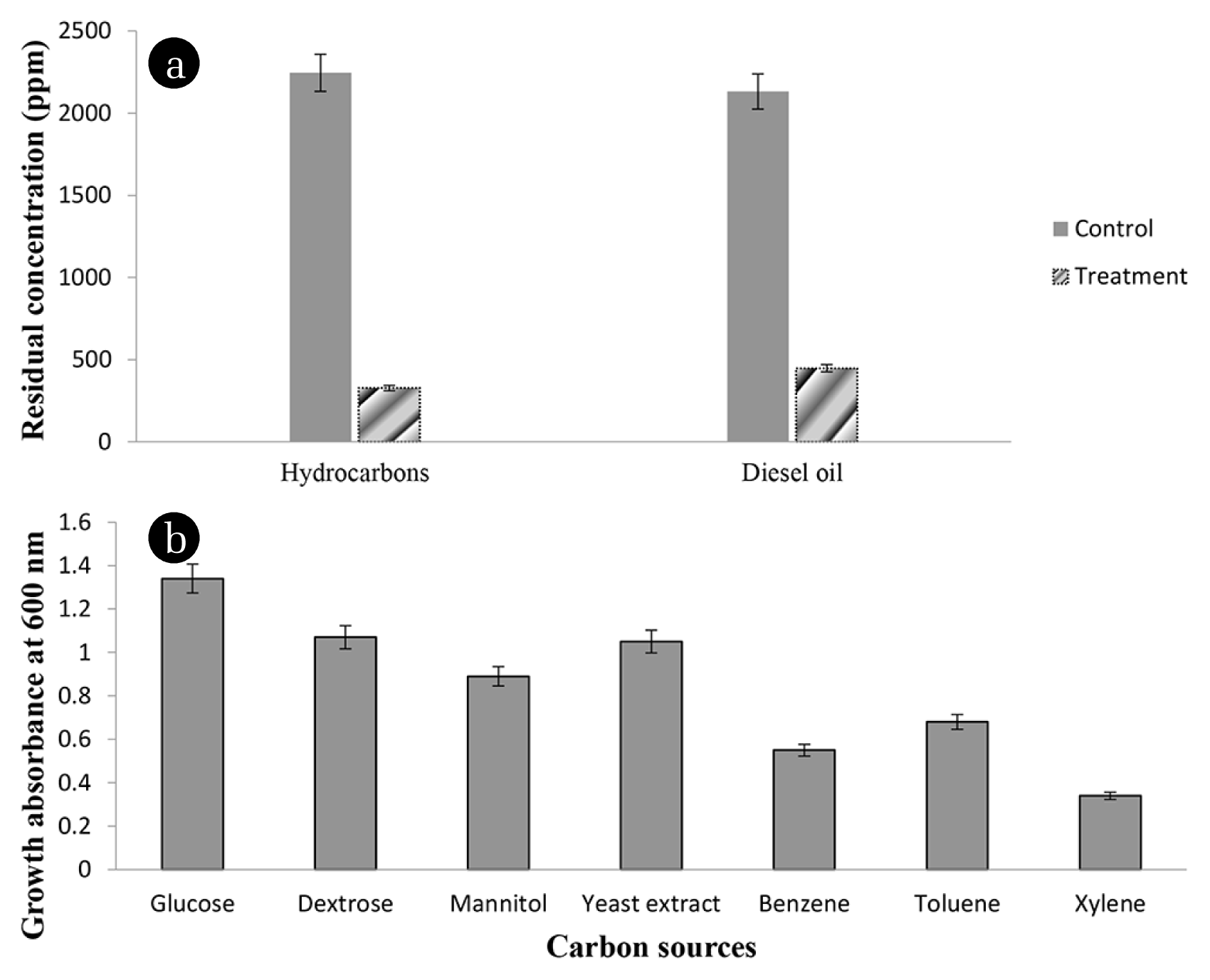
The Acinetobacter sp. strain K-6 showed the highest hydrocarbon-degradation ability at 79.0% and 85.4% for diesel oil and hydrocarbons (C18, C20, and C22), respectively, at 28°C with a cell density of 0.1 g L−1 in MSM liquid media (Fig. 1(a); and Table S3). Statistical analyses showed that the rates of degradation of diesel oil and hydrocarbons by Acinetobacter sp. strain K-6 were not significantly different (p > 0.05). Previous studies also revealed that different strains of Acinetobacter isolated from various source have high petroleum-hydrocarbon-degradation ability [15, 23].
Strains of Acinetobacter are well known to survive in media containing, benzene, toluene, diesel, kerosene, gasoline, and a wide-range of aliphatic hydrocarbons [15, 24, 25]. The assessment of the ability of the bacterium to survive on various carbon sources in this study also showed that Acinetobacter sp. strain K-6 can grow well in media containing glucose, dextrose, mannitol, yeast extract, benzene, toluene, and xylene (Fig. 1(b)). Since diesel oil contains C8-C40 aliphatic hydrocarbons [8, 12], the results of the current study suggest that Acinetobacter sp. strain K-6 can effectively degrade a broad range of hydrocarbon components present in petroleum oil. Their ability to degrade petroleum-hydrocarbons was further confirmed by the presence of a catabolic gene (alkB) in the genome of Acinetobacter sp. strain K-6 (GenBank accession number: MF196316). The catabolic gene alkB encodes the enzyme alkane hydroxylase which is responsible for the degradation of aliphatic hydrocarbons under aerobic conditions [26].
3.3. Biodegradability Patterns and Fractional Analysis of Diesel Oil
3.3.1. Biodegradation of diesel oil
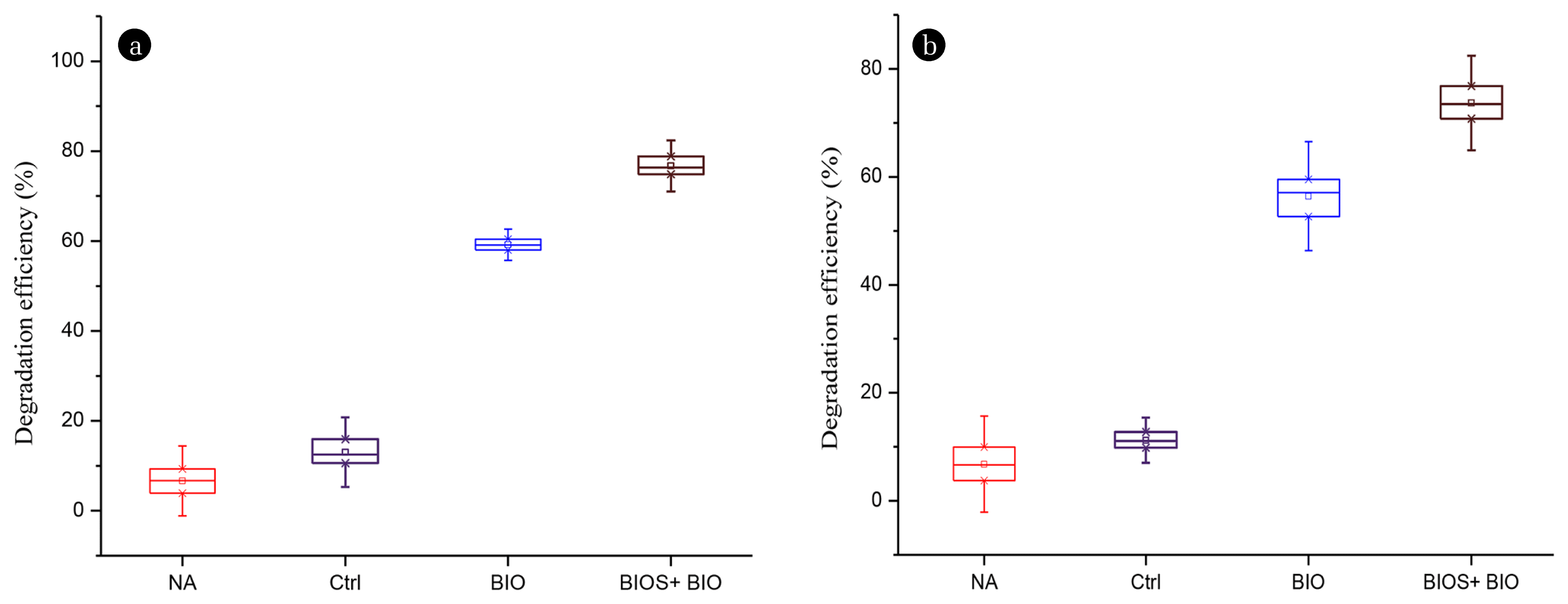
This study evaluated the biodegradability patterns of aliphatic hydrocarbon components of diesel oil using different treatment approaches. Fig. 2(a) indicates the diesel removal efficiencies after 30 d treatment with Acinetobacter sp. strain K-6. The diesel removal efficiency in the BIO microcosms was 59.2% which was significantly higher compared to the NA and Ctrl conditions (p < 0.05). In a similar laboratory scale study, Chang et al. [27] showed lower (only 15%) degradation rate with Acinetobacter baumannii T30C in a crude oil-contaminated soil after 35 d experiment. The BIO strategy is designed to enhance the microbial activity to remediate environmental contaminants, and it is also helpful in boosting degradation during the early phase of treatment [28]. However, several studies have reported that BIO alone cannot lower the contaminants to an acceptable level [12, 27, 29]. This study also confirmed that BIO alone cannot remove sufficient amount of diesel oil. Therefore, the biodegradability pattern of diesel oil was further investigated by implementing the combined approach of BIOS and BIO.
The BIOS + BIO treatment showed enhanced removal of 76.7% of diesel oil as shown in Fig. 2(a), a significantly higher degradation rate compared to the BIO alone treatment (p < 0.05). In a previous study, Chang et al. [27] found that the combined effect of BIOS and BIO degraded 77% of petroleum-hydrocarbons after 35 d of treatment. In another research a similar higher degradation rate of 81% was found by Ruberto et al. [30] when inorganic N and P were amended in the petroleum hydrocarbon contaminated soil. The amendment with nutrients stimulates the metabolic activity of bacteria assisting the removal of hydrocarbons from the soil [27, 29, 31]. This indicates that the combined action of nutrient addition and bacterial treatment can degrade substantial amount of diesel oil in soil microcosms.
3.3.2. Identification of bio-recalcitrant aliphatic fraction
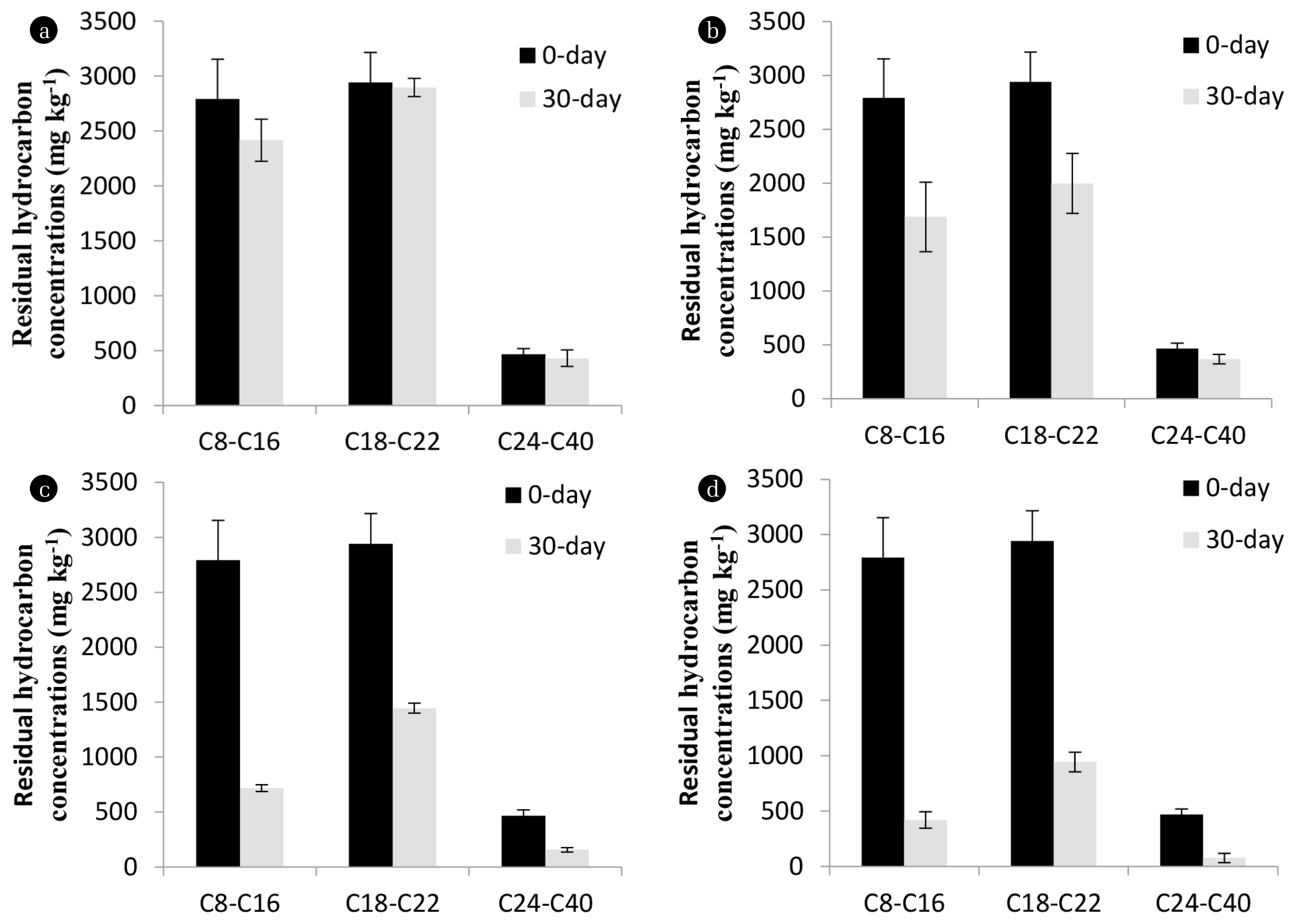
Diesel oil is composed of a large proportion of aliphatic hydrocarbons which are prevalent in soils as a consequence of spills [32]. The aliphatic hydrocarbons are usually degradable compared to the aromatic hydrocarbons [33]. However, different fractions of aliphatic hydrocarbons vary in their degradability pattern [20, 34]. To study the biodegradability pattern of the saturated aliphatic hydrocarbons, fractional analysis of the residual diesel oil after 30 d treatment in different soil microcosms was performed. For this, all the remaining hydrocarbons ranging from C8-C40 were quantified and grouped into three fractions: low molecular weight semivolatile, C8-C16; medium-length nonvolatile, C18-C22, and high molecular weight nonvolatile, C24-C40. Among the three fractions, a higher residual concentration of the medium-length nonvolatile C18-C22 fraction was detected compared to the low molecular weight semivolatile and high molecular weight nonvolatile fractions, in all the treatment methods (see Fig. 3). BIO with other bacterial strains in this study also found higher concentration of C18-C22 compared to the C8-C16 and C24-C40 fractions (see Fig. S3).
Fig. 3 shows that the concentration of C18-C22 hydrocarbons was higher in the BIO treatment (1,500 mg kg−1) compared to the BIOS + BIO (940 mg kg−1) experiment. The results imply that the C18-C22 hydrocarbons may be identified as the bio-recalcitrant aliphatic fraction of diesel oil. The non-volatile, inherent hydrophobicity, poor leaching ability, and limited bioavailability properties of the mid-length saturated alkanes restrict their microbial degradation [10, 11, 35].
3.4. Biodegradation of Bio-recalcitrant Aliphatic Fraction C18, C20, and C22
3.4.1. Biodegradation of C18, C20, and C22 hydrocarbons
The above mentioned data (Fig. 3 and Fig. S3) clearly indicated that the mid-length, non-volatile aliphatic hydrocarbons C18-C22 in diesel oil are bio-recalcitrant. Based on this fact, three hydrocarbons namely octadecane (C18), icosane (C20), and docosane (C22) were selected to investigate in detail the degradability efficiencies using different treatment approaches. Fig. 2(b) and Table 3 present the degradation efficiency of C18-C22 hydrocarbons using four different remediation treatments (NA, Ctrl, BIO, and BIOS + BIO).
After 30 d incubation, the NA experiment showed removal effi ciency of 6.8% only (5,017 to 4,848 mg kg−1) as shown in Table 3. NA appears ineffective in removal of the bio-recalcitrant fraction C18-C22, containing the non-volatile hydrocarbons with indigenous bacteria (102 CFU g−1). Soil washing by spraying sterile water without disturbing the soil in the Ctrl experiment showed that 11.2% of the hydrocarbons were removed from the soil (Fig. 2(b)). Wu et al. [28] have shown a similar pattern of removal rate (16%) of TPH when sterile water was added to soil. Water tends to flush out hydrocarbons and other contaminants from soil [36]. In the BIO microcosms, there was no marked difference in degradation until 15 d of incubation (Table 3). A notable increase in degradation rate was observed after 15 d, and resulted in 56.4% of C18-C22 hydrocarbons being removed by the end of the experiment, which The initial concentration of hydrocarbons was 5,202 mg kg−1. Values represent average of triplicate with ± standard error (n = 3). was significantly higher removal compared to the NA and Ctrl treatments (p < 0.05). This indicates that prolonged incubation of Acinetobacter sp. strain K-6 plays a key role in removing the most prominent residual hydrocarbons C18-C22 of diesel oil from the soil. BIO is a promising technique which, when applied in the early phases of bioremediation process, promotes the remediation of contaminants from soil.
Furthermore, studies have revealed that the rate of hydrocarbon degradation in such soils can be improved by a combination of BIO and BIOS [37, 38]. This study applied a combined treatment strategy by supplementing bacteria with nitrogen and phosphorus. The BIOS + BIO treatment lowered the C18-C22 hydrocarbons levels from 5,202 mg kg−1 to 2,999 mg kg−1 in 15 d as shown in Table 3. At the end of experiment, BIOS + BIO technique showed 73.7% degradation of hydrocarbons (Table 3, Fig. 2(b)). The degradation rate in the BIOS + BIO treatment was significantly higher than in the BIO alone treatment (p < 0.05). The amount of nitrogen and phosphorous is a rate-limiting factor that determines the survival of inoculated bacteria in petroleum oil-contaminated soils [39].
3.4.2. Biodegradability pattern of individual C18, C20 and C22 hydrocarbons
Fractional analysis of the individual hydrocarbons showed that a large amount of all the three hydrocarbons (C18, C20 and C22) were present in the NA and Ctrl experiments (Fig. 4(a) and 4(b)). In the BIO experiment, the final concentrations of C18 and C20 were below 500 mg kg−1 whereas the final concentration of C22 hydrocarbon was below 1,000 mg kg−1 (Fig. 4(c)). In the BIOS + BIO experiment, the final concentration of C18 could not be determined as it was below the detection limit of GC and the final concentrations of C20 and C22 were below 150 and 600 mg kg−1, respectively (Fig. 4(d)). Overall, the fractional analysis found that C22 was hardly degraded compared to the C18 and C20 hydrocarbons. These hydrocarbons are non-volatile hydrophobic compounds and cannot be removed through the natural physical processes. The mid-length aliphatic hydrocarbons such as C18, C20, and C22 possess low bioavailability which makes difficult for microbial degradation [10, 11]. Therefore, the BIOS + BIO treatment method may be a better remediation trial option for soil containing high bio-recalcitrant fraction contaminants. Substantial amounts of bio-recalcitrant hydrocarbons may be degraded through the combined effect of BIO with Acinetobacter sp. strain K-6 and BIOS with nutrients.
3.5. Bacterial Count and Its Viability in Different Soil Microcosms
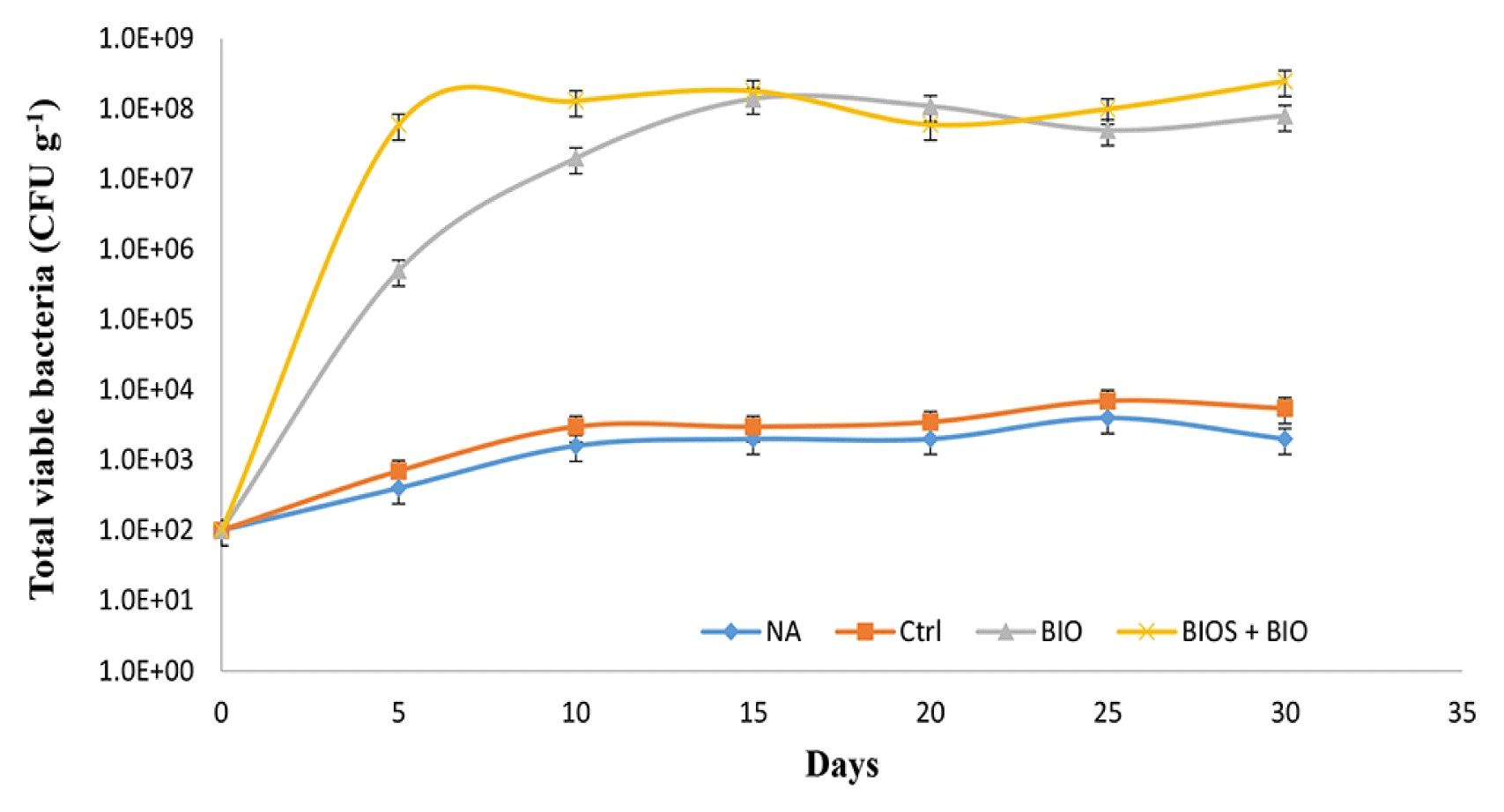
Fig. 5 shows the total viable bacterial enumeration monitored over a period of 30 d. The number of the viable bacteria revealed a low number (102–103 CFU g−1) in the NA and Ctrl experiments, while the number increased (107–108 CFU g−1) in the BIO and BIO + BIOS experiments. Some studies of BIOS and BIO reported that there was a decrease in the microbial population at the final stages of treatment [28, 29]. But, in this study the periodic supplement of bacteria and nutrients helped to maintain the bacterial numbers and enhanced their remediation activities in the hydrocarbon-contaminated soil until the end of the treatments period (Fig. 5).
4. Conclusions
The present study reported the isolation, identification, and characterization of the petroleum-hydrocarbon-degrading strain Acinetobacter sp. K-6. This strain can utilise a variety of carbon sources for their optimal growth and can be considered as competitive bioresource for remediation of petroleum hydrocarbons from soils. The biodegradability pattern and fractional analysis identified C18, C20, and C22 hydrocarbons as the most bio-recalcitrant residual fraction in diesel oil. This is the first study that reports the use of Acinetobacter sp. strain K-6 for the selective remediation of octadecane, icosane, and docosane. The evaluation of biodegradability patterns of C18, C20, and C22 hydrocarbons only using several remediation approaches revealed that BIO alone cannot lower the amount of mid-length, non-volatile, and saturated aliphatic hydrocarbons to a substantial level.
Furthermore, nutrients amendment and inoculation with bacteria boost the remediation efficiency to achieve the bioremediation goal. The combinatorial approach of BIOS and BIO improved the degradation rate and removed adequate amounts of mid-length, non-volatile, and saturated aliphatic hydrocarbons. The findings of this study may play a valuable role in developing efficacious bioremediation strategies for the removal of large quantities of bio-recalcitrant aliphatic hydrocarbons deposited in soil over a long period of time. Further investigations on the degradation pathways of C18, C20, and C22 hydrocarbons by Acinetobacter sp. K-6 are essential to provide deeper insights into the bio-recalcitrant properties of these hydrocarbons.