Occurrence and removal of engineered nanoparticles in drinking water treatment and wastewater treatment processes: A review
Article information
Abstract
Engineered nanoparticles (ENPs) are widely used in various industrial products and consumer goods, resulting in their widespread existence, particularly in natural water systems and water and wastewater treatment plants. Their presence in surface water for human consumption may severely harm human health. Therefore, this review examines new findings and developments in the removal technology of ENPs in drinking water and wastewater treatment processes since the publication of the literature by Park et al. [1]. By evaluating recent articles, this review investigates the occurrence of ENPs, discusses the transport of nanoparticles (NPs) in various drinking water and wastewater treatment processes, and draws corresponding practical conclusions. Moreover, this review provides brief suggestions and predictions for the future development of NP removal technologies in water and wastewater treatment plants.
1. Introduction
Over the past decade, the increasing presence of engineered nanoparticles (ENPs) in the environment has gained global attention [2]. Most ENPs are widely used in cosmetics, coatings, pigments, personal care products, food packaging, and nanotechnology [3]. For example, silver (Ag) nanoparticles (NPs) have been studied for a long time and are extensively used owing to their antifungal, antiviral, and antimicrobial properties; moreover, they show applications in medicine because of their anti-inflammatory properties [4]. In natural water and wastewater, ENPs are a severe threat to human health and the survival of various organisms [1].
ENPs may bioaccumulate in important parts of the body, such as the brain, lungs, and/or liver, which can be harmful to health [5]. For example, Ag-NPs are likely to cause irreversible damage to the human body, such as changes in neurotransmitter levels and liver enzymes, and affect the human immune system [6]. TiO2-NPs are commonly used in various fields of industrial production owing to their unique properties. TiO2 also causes damage to human health; NPs with sizes of < 100 nm can interact with human cells, potentially causing the dysfunction of human cells and organs and even genetic damage [7]. To date, the long-term hazard of metal oxide ENPs to humans remains unknown. Moreover, the ingestion of large amounts of ENPs, particularly via natural water and wastewater, may cause dangerous health problems and considerable environmental damage [8]. The effects of ZnO-NPs on various aquatic plants and animals, including algae, daphnia, sea urchins, and some aquatic mammals, have been reported [9].
When textile fibers with NP components were washed, various NPs were readily discharged into water [10]. The amounts of Ag-NPs and TiO2-NPs in different commercial textiles were measured using mechanical methods [11]. Bratan et al. [12] reported the transport of NPs through the aquatic food chain and measured their concentration in different organisms. Additionally, a recent study showed that 8–20% of annual ZnO-NP production inevitably ends up in natural water bodies; the study also noted an increased risk of NPs to humans [13].
As the knowledge on NPs becomes clear, methods for treating NPs in water (e.g., coagulation/flocculation/sedimentation, membrane filtration, adsorption, and oxidation) have been actively explored [5, 14–16]. For example, Jiang et al. [15] found that the fluoroethylene fluoride microporous membrane is a new type of environmentally friendly industrial microporous membrane with a relatively low cost, exhibiting a ENP removal rate of up to 94.8%. This membrane was employed in the continuous treatment of water samples contaminated with Au-NPs, ZrO-NPs, Fe2O3-NPs, and NiO-NPs, showing high removal efficiency. Fluoroethylene fluoride is a high-potential adsorbent that considerably improves water treatment processes and contributes to water supply biosafety [15]. Flocculation is a conventional method for removing NPs. Oliveira et al. employed the flocculation–sedimentation method using Fe(OH)3, followed by a dissolved air flotation process after sedimentation [17]. Based on the size distribution and zeta potential analysis of the particles, NP flocculants showed unique microphotographic properties. This water treatment process eventually achieved an NP removal rate of > 95% with low turbidity (< 3 nephelometric turbidity units). At high NP concentrations, the removal rate of particles via sedimentation increased, which is sustainable in terms of its NP removal efficiency in water [17].
This review aims to examine new findings and developments in ENP removal technology since the publication of the literature by Park et al. [1]. This review is particularly important for further exploring the removal mechanisms and fate of different ENPs in various drinking water and wastewater treatment processes. The understanding of this new knowledge contributes to practical recommendations for fabricating optimal removal methods for different ENPs.
2. Environmental Occurrence of ENPs
NPs are currently present in various commercial commodities, and their tendency to release into the environment is gradually increasing [18]. In 2014, TiO2-NPs were the most produced and used NPs globally, with an annual production of 60,000–150,000 t, which is expected to increase at a compound annual growth rate of >20% from 2017 to 2022 [12]. The share of different NP materials in the global market in 2010 is listed: Ag-NPs (47%), carbon (16%), TiO2-NPs (11%), ZnO-NPs (10%), SiO2-NPs (9%), and Au-NPs (7%) [18]. Being the main point of entry, aqueous environments play a decisive role in NP dispersion [10]. In surface water, TiO2 exhibits the highest NP concentration of 2.2 μg/L, followed by Ag-NPs, with an NP concentration of 1.5 μg/L [19]. The benefits of ENPs are diverse, with the most common applications found in medicine, disease diagnosis, and environmental pollutant removal. Generally, Ag-NPs and carbon nanotubes (CNTs) are the most widely used ENPs owing to their antibacterial properties and applicability to medical devices [20]. In wastewater treatment processes, the use of NiO-NP–reinforced nanomolecular micromembranes has been recently reported for treating contaminants in water. NPs released by water treatment plants can be easily discharged into seawater and enter the ecosystem. A survey has shown that > 90% of NPs was used to enhance the physical strength of structures, such as rubber and concrete [21]. Fig. 1 depicts the possible fate of NPs in marine environments and organizations at risk of exposure [22].
The NP source can be divided into two main categories: naturally occurring NPs released into the environment and NPs attributed to intentional/unintentional human activities. For the second category, automobile exhaust is considered a significant source of NP pollution because burning fossil fuel, diesel, and gasoline engines release ENP pollutants with sizes of 20–130 nm [23]. The combustion process of cigarette burning releases approximately 100,000 ENPs with sizes of 10–700 nm [24]. Building demolition and the fabrication of home-furnishing supplies and building materials release considerable amounts of toxic NPs with sizes of <100 nm into the atmosphere [25]. Various commercial goods used in biopharmaceutical and medical applications, such as personal care products, mainly contain ZnO-NPs and TiO2-NPs that undergo variations in terms of the specific surface area and chemical changes in water, causing them to disperse in the aqueous environment, as reported in a study on nanomaterials in cosmetics [26].
Most TiO2-NPs and Ag-NPs released as byproducts of the aforementioned applications are treated in wastewater treatment plants, which are one of the essential ways of diverting ENPs from the environment [27]. Keller et al. [28] used a mass flow model to derive the influent and effluent concentrations of TiO2-NPs and Ag-NPs in wastewater treatment plants, obtaining influent and effluent concentrations of 100–200 and 1–20 μg/L for TiO2 and 2–18 and 0.003–0.26 μg/L for Ag, respectively. Previously, all elemental titanium concentration measurements in wastewater treatment plants were based on the fact that titanium only originated from ENPs and researchers never considered natural factors [29]. However, a recent study has shown that titanium concentrations in wastewater treatment plants are often higher than expected because titanium present in rocks and minerals is transported into groundwater and surface water via openings in sewer system and soil seepage because of weathering and corrosion [30]. Furthermore, in nature, TiO2 is mixed with trace amounts of other elements; therefore, NP contamination caused by natural factors must be considered [31].
Owing to weathering, ENPs (e.g., TiO2-NPs, Ag-NPs, and ZnO-NPs) may fall off the facade of buildings and transport to nearby lakes or rivers, which are downstream water sources, via rainwater [32]. Generally, wastewater treatment plants can remove > 90% of ENPs in water sources using physical, biological, or chemical approaches; however, untreated ENPs are still discharged into lakes or rivers [28]. Approximately 30% of ENPs with sizes of < 100 nm, such as various cell fragments, fibers, and inactivated viruses, is discharged into water bodies [33]. In the US, drinking water treatment plants are usually located downstream from sewage treatment plants. These findings suggest that wastewater treatment plants are generally one of the major sources through which ENPs enter the environment [34].
Recent data based on several studies have suggested that in our current society, ENPs are widely used everywhere in urban environments; hence, urban runoff has become an important source of ENP emission [23]. In urban environments, TiO2-NPs are added to most paints because TiO2 can be used as a photocatalyst and exhibit a unique self-cleaning surface function. Such paints are excessively used in the US, including white paint reinforced with TiO2 for painting road signs [35]. The use of such paint in the US is estimated to be as high as 450,000 t in 2025, which will lead to the discharge of large amounts of TiO2 into the environment through urban runoff. Parker et al. found that the concentration of TiO2 in urban runoff increases considerably after a rainstorm [36]. Another study reported a high concentration of TiO2 in roadside sludge and soil after rainwash [37]. Urban runoff must cause considerably lesser ENP pollution than sewage treatment plants, although the quantitative data of related research are difficult to obtain. Therefore, further research can assess the long-term effects of urban runoff.
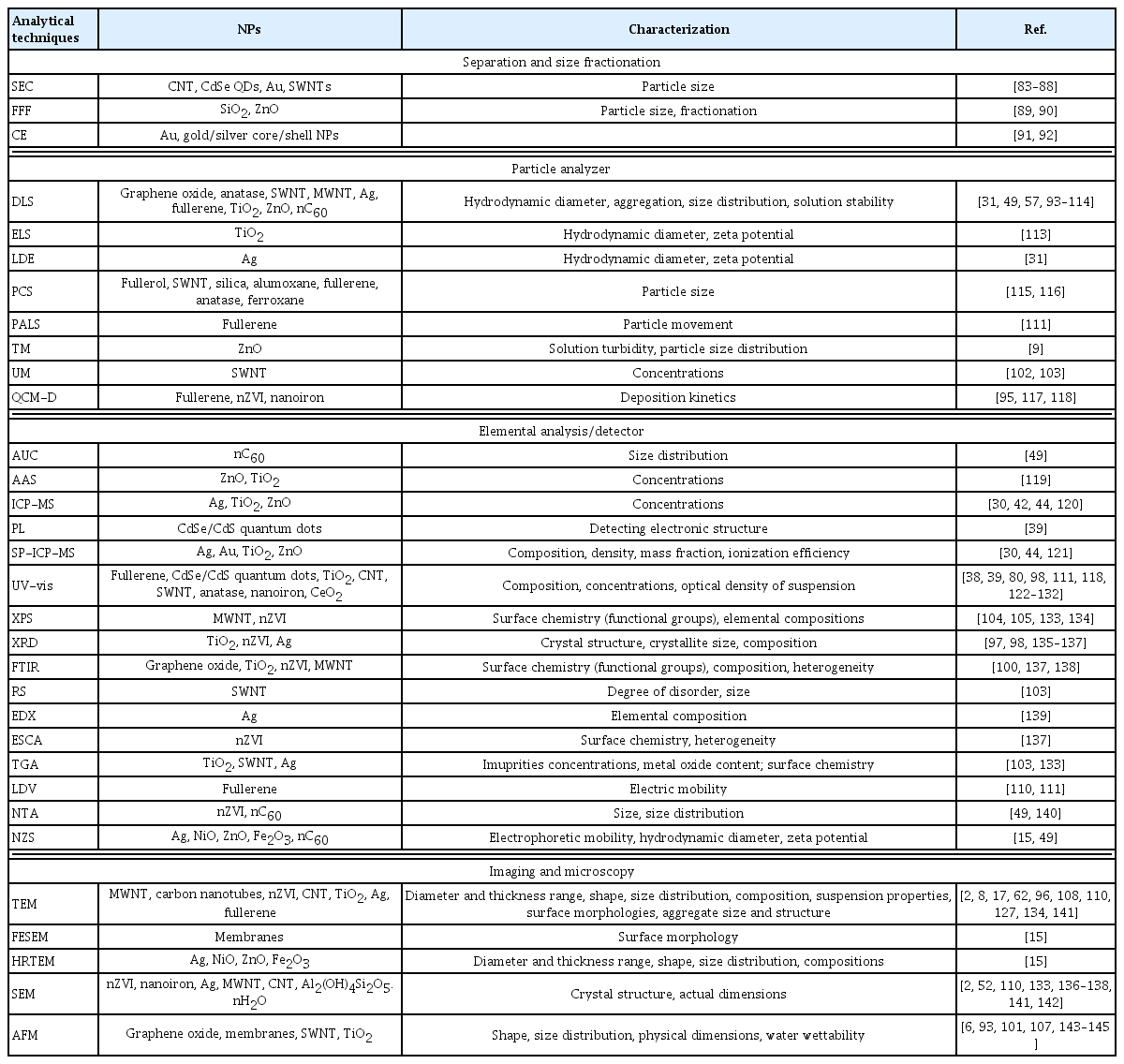
Aiming at determining the content of various ENPs in sewage, Keller et al. [25] reported that in a drinking water treatment plant, nanomaterials, such as TiO2, ZnO, SiO2, and Ag2O, have been commonly found in the decreasing order of their concentrations [28]. Additionally, sewage water contains both nonadhesive and adhesive NPs. However, nonadhesive NPs, adhesive NPs, and ENPs show similar structures, shapes, and elemental composition, making the measurement and distinction of different NPs difficult in water. Various analytical techniques have been used to characterize different ENPs in the environment (Table 1).
3. Fate of ENPs in Drinking Water Treatment
3.1. Coagulation/Flocculation/Sedimentation
A new coagulant, namely, SiO2–tetraethylenepentamine was synthesized to remove Ag-NPs [38]. During coagulation, Ag-NPs interacted with this coagulant in many ways, the most common way being via electrical neutralization, to produce metal ligands. Further, hydrogen bonds and an oversea bridging function were observed. The length of the nitrogen chain of SiO2–tetraethylenepentamine was a key factor affecting the coagulation process. Another significant factor was the nitrogen content. Under optimized conditions, the maximum Ag-NP removal rate of 98.3% was obtained in 35 min using the SiO2–tetraethylenepentamine coagulant [38]. The corresponding study provides a novel idea for developing other new coagulants. In another study, the latest characterization method, namely, AF4 inductively coupled plasma mass spectrometry (ICP-MS), was employed to measure Ag-NPs in a solution and analyze their size distribution and shape structure [31]. AF4 ICP-MS can detect some Ag-NPs with sizes of < 20 nm; however, single-particle ICP-MS cannot detect these NPs. In the experiment, Ag-NPs were extracted from primary particles and their aggregates and the Ag concentration in particles with sizes of 100 and 450 nm remained similar.
Chen et al. [39] evaluated the removal effects of Fe3+ and Al3+ coagulants on the aggregation activities of 3-mercaptopropionic acid–capped CdSe/CdS core–shell quantum dots. The stability of polyadenylic acid quantum dots was affected by the type of cation rather than the concentration. This finding indicates that the complexation and depletion of carboxyl groups on the surface by Ca2+ considerably affected the compression of the electric double layer than those of carboxyl groups on the surface by a high-concentration K+. The presence of humic acid increased the stability of polymethylmethacrylate quantum dots, which may have increased the negative surface charge by coating or bonding to the polymethylmethacrylate quantum dot surface. The kinetic aggregation models of NPs were similar in tap water and seawater but different in lake water owing to the total calcium and magnesium concentration of 2.3 mM. Al3+ and Fe3+ can combine with the carboxyl groups of surface-terminated ligands, neutralize negative charges on the surface, reduce electrostatic repulsion, and induce the aggregation of surface-terminated ligands. Moreover, polymethylmethacrylate quantum dots can be combined and wrapped with hydrolysate floc coagulants [39].
The TiO2-NP removal efficiency was investigated in surface water using conventional techniques, such as coagulation, flocculation, and precipitation, during drinking water treatment to explore the relationship between the treatment process and natural organic matter in water [40]. Results showed that the TiO2-NP removal rate in both hydrophobic and hydrophilic aqueous solutions exceeded 90%. Furthermore, the initial TiO2 concentration was 0.2–10 mg L−1 and the coagulation, flocculation, and precipitation processes at this concentration had no obvious influence on the removal rate of natural organic matter. Therefore, it is scientific and reasonable to remove TiO2-NPs and biological nitrogen and organic oxygen compounds using polyaluminum chloride and conventional coagulation/flocculation/precipitation treatments. The characteristics of the water substantially affect the NP removal efficiency because hydrophobic water requires additional coagulant concentrations than hydrophilic water to ensure that the TiO2 removal efficiencies for these two types of solution are roughly equal; thus, water with a high UV254 value can be obtained [41]. The core of removing TiO2-NPs lies in neutralizing the positive and negative charges [14]. In another study, different ionic species and strengths were examined to remove TiO2-NPs from water via coagulation [42]. Appropriate contents of coagulants were used based on the Derjaguin–Landau–Verwey–Overbeek theory, as well as 0.1–0.2 mM of polyaluminum chloride combined with FeCl3, increasing the TiO2-NP removal rate to > 90%. The mean particle size of the suspended TiO2-NPs decreased from 145 to 43 nm after coagulation and sedimentation owing to the successful floc formation using 0.2 mM FeCl3 as a coagulation agent.
The large-scale production and widespread use of ZnO-NPs has induced the risk of ZnO-NPs entering aqueous environments [9]. The relationship between organic ligands and the adsorption and precipitation treatments of ZnO-NPs under different water characteristics was explored. Moreover, the coagulation effects of ZnO-NPs, Zn ions, dissolved organic carbon, and UV254 in synthetic water under different FeCl3 concentrations were discussed. Results showed that the concentrations of humic acid, salicylic acid, and cysteine were negatively correlated with the zeta potential and hydrodynamic diameter of the particles. Furthermore, a higher concentration signified increased stability of the nanoions. The adsorption of organic ligands on ZnO-NPs followed the Langmuir model, and the adsorption peaks of hydroxyapatite, salicylic acid, and l-cysteine were observed at 143, 40.5, and 66.1 mg/g, respectively [9]. In the effective coagulation zone, the removal efficiency of ZnO-NPs and Zn2+ exceeded 95%; the effective coagulation zone, the excessive charge induced by the coagulant stabilized the suspended NPs again. Additionally, the removal efficiency of dissolved organic carbon and UV254 was related to water chemistry conditions [43]. The removal efficiency of hydrophobic water was higher than that of hydrophilic water, and the width of the effective coagulation zone was directly related to the water characteristics. Water with hydrophobic natural organic matter and higher UV254 value require additional coagulant concentrations than hydrophilic water to achieve a similar removal efficiency of ZnO-NPs and Zn2+. The Fourier transform infrared (FTIR) spectroscopy analysis of ZnO-NP composite pollutant flocs showed that the interaction of meshing and charge neutralization can be a possible removal mechanism.
The fate of TiO2-NPs in a simulated drinking water treatment was characterized using both powder and suspension mode TiO2 [44]. TiO2-NP solutions with concentrations of 0.1, 1, and 10 mg/L were prepared to investigate the impact on the TiO2-NP activity using deionized water and raw water at two water treatment plants in Taiwan. In these both plants, the residual Ti content in separate treatment units was 2.7–47.4% for the laboratory-simulated water treatments of prechlorination, coagulation, sedimentation, filtration, and postchlorination, with the total removal rate of 52.6–97.3%, except for the 0.1-mg/L TiO2-NP solution. The overall removal rate of was 9.3–53.5% for the 10-mg/L TiO2-NP solution. The main processes for TiO2-NP removal, partly attributed to particle agglomeration, sedimentation (after coagulation), and filtration, have been verified using particle size distribution and zeta potential measurements [45]. During sedimentation, the size of TiO2-NPs increased from 21–36 to 4,490 nm in the supernatant; after filtration, all agglomerates with sizes of 1 μm were further extracted.
3.2. Membrane Filtration
Different ionic species and strengths were evaluated to remove TiO2-NPs from water via microfiltration (MF) [42]. An uncoagulated TiO2-NP suspension (nominal pore sizes: 0.45 and 0.10 μm) was subjected to MF, resulting in lower NP concentrations of the filtrate than the NP concentration of the precoagulated water filtrate [46]. This is because high concentration NPs in feed water reduced particles smaller than the pores by blocking or covering the membrane pores via pure coagulation and settlement. Results indicate that the elimination of NPs via coagulation/sedimentation accompanied by membrane MF may be less effective than direct membrane filtration. The addition of suspended TiO2-NPs until coagulation (0.3–15 mM NaCl or 0.1–5 mM CaCl2) considerably decreased the TiO2 concentrations (0.28–0.47 mg/L) in the supernatant after sedimentation, leading to increased NP aggregation because electrostatic repulsion between negatively charged particles was mitigated [47]. The addition of phosphates (0.01, 0.1, and 1.0 mM) increased the TiO2-NP concentration during coagulation and settlement using FeCl3 or polyaluminum chloride. However, the removal rate of TiO2-NPs via MF increased owing to phosphate addition, except for the 1.0 mM phosphate suspension filtered using a 0.45-μm membrane.
For the first time, Jiang et al. [15] proved that the polyvinylidene fluoride microporous membrane could be used as a low-cost and environmentally stable adsorbent to capture NPs from water. Using Au-NPs and Ag-NPs as models, they studied four industrial microporous membranes, namely, polyvinylidene fluoride, nylon, mixed cellulose ester, and polyethersulfone, using intermittent adsorption experiments. The polyvinylidene fluoride microporous membrane showed the best removal efficiency [15]. Moreover, this membrane can be used as a filter for the continuous treatment of water samples contaminated with Au-NPs, Ag-NPs, ZrO2-NPs, ZnO-NPs, Fe2O3-NPs, and NiO-NPs [48]. Furthermore, the viability of this approach was confirmed by comparing its performance with that of an NP-contaminated water treatment using a commercial substance. The polyvinylidene fluoride microporous membrane is a possible adsorbate for capturing NPs from water. This useful application can boost water management processes and increase the biosafety of water sources.
Because ENPs, such as buckminsterfullerene nC60, in water pose possible environmental and health hazards, they must be removed during drinking water treatment processes [49]. The corresponding study focused on the removal mechanism and the function of the membrane pore size during the removal of nC60 fullerene NP suspensions from dead-end MF and ultrafiltration systems, mimicking the separation process in industrial water treatment plants. The membrane content and aperture ratio were the key factors affecting the affinity between nC60 and the membrane [50]. Membranes with pore sizes of 18–500 nm were selected. Experiments were conducted on a dead-end bench-scale device using a hydraulic backwash cleaning process under continuous flux conditions. The obtained removal rate was ~20% higher than those reported in the literature with similar membrane pore sizes [51]. Although the nC60 filtration behavior appeared to be distinct, the low-pressure membrane technology with pores that are smaller and, surprisingly, primarily identical or larger than the particle size effectively extracted the nC60 NPs. The ratio of cake-layer deposition resistance to the clean membrane resistance and the ratio of nC60 size to the membrane pore size affected the nC60 filtration activity and backwash process efficiency in restoring the original membrane filtration conditions. These findings are significant for drinking water treatment processes, as they provide useful information for determining the best membrane processes and operating criteria for removing nC60 from drinking water.
3.3. Adsorption
Halloysite has been effectively introduced to remove Ag-NPs in water dispersed with iron oxide NPs [52]. The efficiency of halloysite to adsorb natural and magnetic materials was high; however, the overall adsorption potential of magnetically sensitive halloysite (67.9 mg/g) was higher than that of the natural halloysite (40.9 mg/g). This low-cost, advanced composite material that can be conveniently isolated from water using an external magnetic field is a promising adsorbent for treating Ag-NP–contaminated water, particularly in potential environmental technology. In a separate study, both natural and magnetically sensitive halloysite materials were studied as potential Ag-NP adsorbents [53]. Ag-NPs were prepared using disaccharide maltose by reducing diamino Ag complex cations, which exhibit an average diameter of 26–31 nm. In the preliminary experiment, under standard laboratory conditions, the Ag-NP adsorption reached equilibrium within 30 min. The higher magnetically enhanced adsorption of Ag-NPs by halloysite can be attributed to the broad surface region of the substance, although other reasons were most likely responsible [53]. This substance is interesting for future environmental technology applications owing to its high magnetic adsorption capability and easy isolation from treated solutions and suspensions. The findings of other studies have shown that halloysite, particularly magnetic halloysite, isolates Ag-NPs from water systems. The advantage of magnetically adjusted halloysite is that it can be easily removed from large quantities of solutions or suspensions using a magnetic separator.
Suhartono proposed the use of activated carbon extracted from natural sources to remove Ag-NPs from water [54]. Activated carbon was extracted from cocoon shells using a chemical method. Several kinetic adsorption models were used to describe the kinetic process, such as pseudo-first, pseudo-second-order, pseudo-first-order fractal, and pseudo-second-order fractal kinetic models. This study showed that the proposed activated carbon could remove Ag-NPs from water systems. The mixed first-order and second-order adsorption behaviors were well defined compared with the other models. Taylor et al. removed TiO2-NPs from water by first covering them with polydopamine and then encapsulating with liposomes before being adsorbed on poly-L-lysine glass surfaces covered with lecithin [55]. Atomic force microscopy with a thickness of 30 nm verified the poly-L-lysine coating. The average removal rate of 58% was obtained with a regular variation of 18% for concentrations of 5–125 mg/L. This approach offers a promising solution for mitigating the health hazard of TiO2-NPs. Water quality factors, such as alkalinity, ionic strength, and humic acid, had negligible effects on the TiO2-NPs removal efficiency. In this case, coagulation, flocculation, and sedimentation were not needed. TiO2-NPs were first covered with polydopamine and then encapsulated with lecithin liposomes before being adsorbed on glass surfaces covered by poly-L-lysine [56]. The transmission electron microscopy images showed that the poly-L-lysine concentration, with a smooth coating thickness of 30 nm on the glass surfaces, was a key factor owing to solid electrostatic interactions between the positively and negatively loaded poly-L-lysine glass surfaces. Additionally, the removal rate of NPs confirms our theory that lecithin liposome–encapsulated polydopamine–TiO2-NPs can electrostatically bind to poly-L-lysine–coated glass surfaces.
Piplai et al. [57] investigated the impact of pH values of 5–9 and activated carbon concentrations of 0.01, 0.1, 1, 10, and 100 g/L on the removal efficiency of colloidal NPs (ZnO and CuO) from their solutions in batch kinetic experiments. Results indicated that at pH values of 8 (ZnO) and 6 (CuO), the adsorption potentials of activated carbon for ZnO-NPs and CuO-NPs (removal efficiency of NPs using activated carbon) were 0.9 (91.3%) and 0.95 (95.6%) mg/g in deionized water, respectively, and 0.6 (78%) and 0.75 (83%) mg/g in wastewater, respectively. The adsorption potentials of Zn in the colloidal solution using activated carbon were 0.71 (74.7%) and 0.58 (69%) mg/g in deionized water and wastewater, respectively, whereas for CuO, they were 0.82 (81.2%) and 0.6 (71.5%) 0.6 mg/g in wastewater and deionized water, respectively. Generally, the corresponding research presented a thorough overview of the removal capacities of activated carbon and suggested that activated carbon could be used as an effective adsorbent to remove ENPs, such as ZnO-NPs and CuO-NPs (single and mixed solutions), from water [58]. As removing NPs from wastewater is essential, an activated carbon–based adsorptive filter is a promising technique.
The removal of ZnO-NPs and CuO-NPs from a colloidal solution via sedimentation/settlement was also investigated (removal rate of < 5% for both NPs) [57]. In both single and blended solutions, the overall adsorption potential of ZnO-NPs and CuO-NPs using activated carbon was examined. The equilibrium times required for removing ZnO-NPs and CuO-NPs from deionized water were found to be 480 and 390 min, respectively. Based on the spectrum shifting of depleted activated carbon, FTIR analysis showed that functional classes participated in the removal of ZnO-NPs and CuO-NPs. Both NPs were also removed via sedimentation. As observed from FTIR analysis, electrostatic attraction and surface complexation removed some NPs, while some NPs may have entered the pores, as depicted from Brunauer–Emmett–Teller (BET) analysis [59]. Only characterization analysis was used to determine the removal process. No quantitative approach was employed to test the concentration of NPs accumulating on the activated carbon. Thus, activated carbon should be used to quantify the concentration of NPs in water.
Iron oxide magnetic particles were synthesized and used to remove Ag-NPs via a simple solvothermal reaction [2]. The prepared iron oxide magnetic particles showed strong adsorption potential for Ag-NPs in an aqueous medium. Kinetic experiments have shown that the adsorption of Ag-NPs is a pseudo-second-order process [60]. The Langmuir isotherm model was studied, followed by the test results for adsorbing Ag-NPs, with the maximal adsorption ability of 19.9–62.8 mg/gat obtained at a pH of 6.2 and 298 K. The measured sorption free energy was 4.09–5.17 kJ/mol, indicating physisorption, which can be primarily attributed to electrostatic interactions. After four adsorption–desorption cycles, the iron oxide magnetic particle adsorbents retained substantial removal efficiencies, indicating that the formed iron oxide magnetic particles were reusable [61]. Further, strong removal efficiencies (63.3–99.9%) and recoveries (67.1%–99.9%) were obtained for the actual Ag-NPs spiked up to a level of 10 μg/L, indicating that aged iron oxide magnetic particles can be used as a low-cost, effective adsorbent to remove and recover Ag-NPs from natural water.
3.4. Other Technologies
Zhang et al. [62] presented a basic method for removing CNTs from industrial and laboratory wastewater using sodium hypochlorite. CNTs were completely degraded into carbon oxides or carbonate ions when aqueous dispersions were directly treated with a sodium hypochlorite solution. The findings demonstrated that two popular commercially available CNTs (single-walled and multiwalled CNTs) exhibit similar properties. To study the structural modifications of CNTs, a CNT dispersion solution (100 mg/L) was ultrafiltered using a 10-kDa molecular weight cutoff membrane and rinsed with water before and after NaClO treatment for 1, 3, and 24 h. Transmission electron microscopy and Raman spectroscopy were used to characterize the collected CNTs. Additionally, the study discovered that graphene oxide could be an intermediate product of single-walled CNT degradation, implying that CNT degradation may require an unzipping mechanism compared with CNT oxidation using a strong acid treatment [63]. However, the reaction rates using sodium hypochlorite were considerably higher than those using a strong acid; moreover, no obvious nanoribbon structures were discovered. Additionally, because the components of raw wastewater are more complicated than those CNT dispersions used in the analysis, it was suggested that the method’s feasibility must be confirmed using raw or simulated wastewater [62].
In summary, their findings show that CNTs can be completely isolated from aqueous solutions, such as wastewater, using a simple chlorination treatment [62]. In the presence of sodium chloride, the degradation rate of CNTs varied based on their type; in other words, single-walled CNTs deteriorated more rapidly than double-walled CNTs, while multiwalled CNTs degraded more steadily than both single-walled and double-walled CNTs. Unlike the degradation of CNTs, the energy-dispersive X-ray spectroscopy spectra and elemental mappings of C, Si, O, Na, and Cl revealed no hydrocarbon component. However, some issues, such as the detailed mechanism of CNT degradation using NaClO, relationship between the degradation rate and the type of CNT, and effect of impurities and other substrates in practical wastewater on CNT degradation, remain unresolved [64]. The results presented in that study demonstrate a novel route for developing a suitable method for removing CNTs.
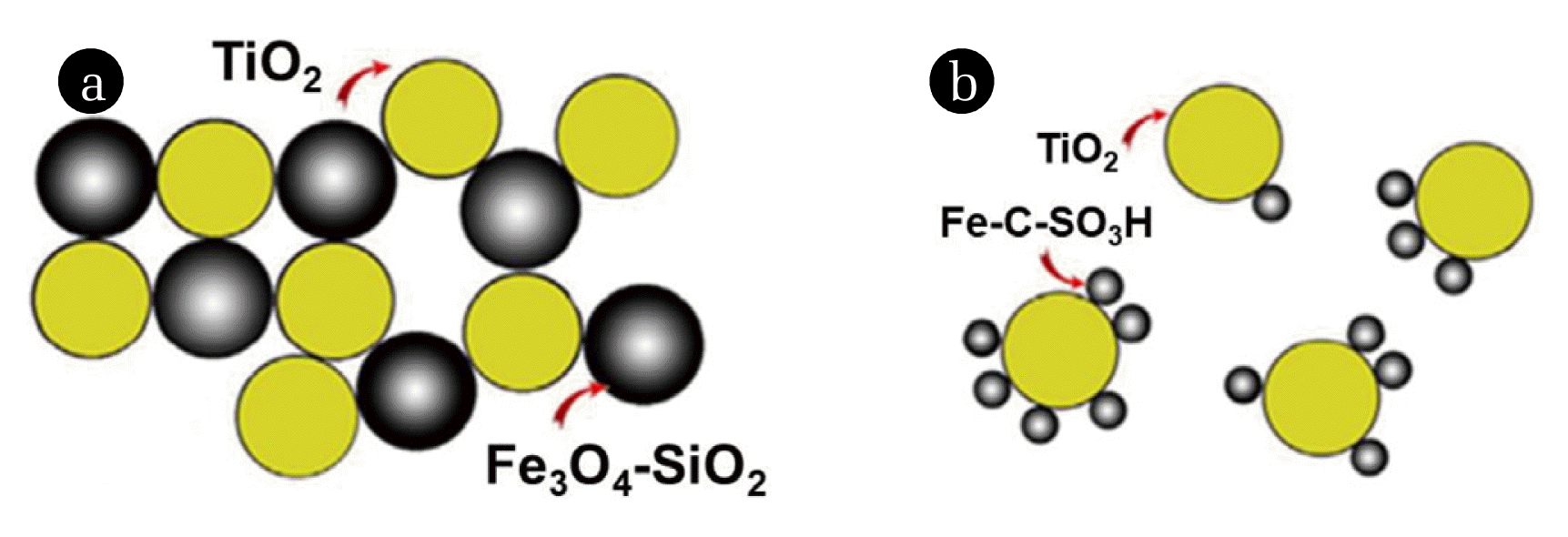
Another study investigated a novel method for removing TiO2-NPs from water [8]. Composite materials, such as Fe3O4–SiO2 and Fe–C–SO3H-NPs, with magnetic cores (5 nm) were engineered for the magnetic sedimentation of nonmagnetic TiO2-NPs in water. The sedimentation dynamics of the heterogeneous water suspensions of (TiO2)x(Fe3O4–SiO2)x(x = 0.15–0.55) and (TiO2)1x(Fe–CSO3H)x(x = 0.01–0.17) were investigated in a gradient magnetic field (B = 0.3 T, dB/dz = 0.16 T/m) for pH values of 3–6. After magnetic sedimentation, the residual concentrations of Fe3O4–SiO2, Fe–C–SO3H, and TiO2-NPs in water were determined using nuclear magnetic resonance spectroscopy relaxometry and ultraviolet spectrophotometry [65]. The hydrodynamic sizes of the heteroaggregates (dh) depended on the pH and were proportional to the mass content of the magnetic variable x. The dh limit of 5 m and the quickest sedimentation were achieved for the mixed (TiO2)1x(Fe3O4–SiO2)x water suspension when x = 0.35–0.5 at pH = 3 and 4 and x = 0.2 at pH = 5. Under this regime, the maximum removal rate of TiO2 particles of 99.7% from water was observed in 30 min. The highest heteroaggregates and quickest sedimentation were obtained for the mixed (TiO2)1x(Fe–CSO3H)x water suspension when x = 0.03–0.05 at pH = 3 and 4 and x = 0.03 at pH = 5. However, when using Fe–C–SO3H-NPs as a flocculant, the minimum residual concentration of TiO2-NPs in water increased by two orders of magnitude [66]. These findings were discussed in the context of a particular distribution of Fe-based NPs formed via heteroaggregation with TiO2-NPs. Fig. 2 demonstrates the heteroaggregation in the mixed suspensions.
The removal of TiO2-NPs (mean volumetric diameter = 450 nm) via flocculation accompanied by settlement or dissolved air flotation was investigated in bench-scale studies (4 bar saturation pressure and 30% recycling ratio) [17]. After heterocoagulation, iron hydroxide (30–40-mg/L Fe3+) and gelatinized corn starch (10–20 mg/L) were formed as flocculants at a pH value of 7. The particle size and zeta potential distributions, time removal efficiencies, and floc microphotographs were studied. The removal mechanisms involved the precipitation of ferric hydroxide, heterocoagulation the NPs, and flocculation of the filled carrier with gelatinized starch. Optimum findings reported of the TiO2-NP removal rates of 95–100% by settlement or flotation after 5 min. Clear treated water at a concentration of < 1 mg/L and a low turbidity (< 3 nephelometric turbidity units) and TiO2-NPs were collected. Table 2 summarizes the removal rates of selected ENPs in drinking water treatment processes.
4. Fate and Transport of ENPs in Wastewater Treatment
The development of nanomaterials using different NPs has led to the release of NPs in wastewater tanks and wastewater sludge. Studying the fate and transport of NPs is essential for environmental impact assessment because NP biosolids are present in wastewater treatment facilities. Multiple techniques are involved in the removal of CuO-NPs, ZnO-NPs, TiO2-NPs, and CeO2-NPs from wastewater [67]. The wastewater treatment plant could remove up to 6% of CeO2, while up to 95% of the CuO-NPs are removed via the water treatment facility.
In contrast to biosorption, which is a common technique for copper removal, the predominant mechanisms for removing Cu-NPs are agglomeration and settlement [68]. Cu-NPs are also completely removed from wastewater, with only a small amount of waste returned to the water supply. Owing to the large content of ZnO in wastewater, a considerable amount of ZnO-NPs is transported to the sludge. Most NPs aggregate at the wastewater bottom and then attach to the biomass. Most of the Ag-NPs can remain in sulfate and inorganic wastewater treatment plants and can be extracted from the plant surfaces [69]. Moreover, in effluents and sludge, most polysaccharide NPs are present in the form of Ag2S. Ag-NPs are transported to sewers with a relatively minor impact on the biofilm transport capacity. Additionally, owing to their electrocatalytic activity, Ag-NPs can be efficiently removed from wastewater treatment systems. In wastewater treatment plants, titanium concentrations in raw sewage vary from 181 to 1,233 mg/L. Wastewater treatment plants can remove > 96% of influent TiO2-NPs, while the effluent removal rate is found to be < 25 mg/L in all facilities [70]. Several studies have indicated that on average, > 90% of NPs derived from sewage are removed; however, the removal efficiency of SiO2-NPs is considerably low.
SiO2-NPs in wastewater are unstable during removal via sedimentation and show considerably slow settlement during primary treatments [67]. The corresponding study showed that 96% of SiO2-NPs in wastewater was removed in 6 days. However, the concentration of SiO2-NPs in the effluent gradually increased and NP levels in the wastewater reached 65% of the input after 30 days of feeding. The biosorption of SiO2-NPs directly resulted from low dissolution levels and limited biosorption propensity in wastewater. Environmental factors can significantly affect the migration of NPs. Attrition, erosion, aggregation, and agglomeration were the four main factors that can affect the particle size of metal NPs in a suspension [67]. Note that salinity influenced activated sludge and its microorganisms. A better understanding of these techniques will improve the bioavailability, uptake, and toxicity of NPs [68]. Various properties of exposure media, such as concentration and pH, may influence the rate of metal NP dissolution and aggregation in, for example, natural organic matter. The release of Zn2+ from ZnO-NPs under acidic conditions was considerably significant; however, a large amount of Zn2+ was bound to suspended or dispersed particles in the filtrate when the pH exceeded 10. The dissolution or ionization of Cu nanoplates was observed in wastewater treatment systems [69].
Ag-NPs can be converted to Ag+ in wastewater, which is more harmful to the microbial species in activated sludge and the nitrification phase than Ag-NPs. However, dissolved organic matter will considerably stabilize Ag-NPs [71]. Sulfuration may cause Ag-NPs and ZnO-NPs to aggregate. In aqueous conditions, ZnO-NPs are mildly soluble and can dissolve to form Zn2+. At a ZnO-NP concentration of > 40 mg/L, Shewanella oneidensis MR-1 and Escherichia coli are insensitive to the released Zn2+ [72]. Overall, the data show that biological wastewater treatment plants utilizing activated sludge will effectively remove most forms of NPs from wastewaters. NPs, such as Ag-NPs, ZnO-NPs, and TiO2-NPs, can be effectively extracted because of their interactions with biomass [3]. Under such circumstances, Ag-NPs, Cu-NPs, and ZnO-NPs will partly turn into Ag+, Cu2+, and Zn2+, respectively. The involvement of both extracellular polymeric substances and ionic compounds will efficiently improve NP removal [38]. The volatility of NPs influenced by the sewage ionic strength and NP entrapment by extracellular polymeric substances are essential in NP removal using activated sludge. This is a synopsis of the fate and transformation of NPs during urban wastewater treatments [73]. In a separate study, samples with a high organic load were collected from different locations of two typical municipal sewage treatment plants to study the characteristics and distribution of ENPs in the entire system [74]. By designing and refining the pretreatment procedures, ENPs, coarse particles, and ionic substances were separated from wastewater and sludge particles. By measuring and analyzing the concentration and mass flow rate of selected NPs at different time points, the whereabouts and distribution of ENPs in sewage treatment plants can be known [11].
A study outlined the qualitative and quantitative findings of ENPs in wastewater treatment systems, focusing on pretreatment schemes and chemical analysis techniques [75]. The primary pretreatment techniques are acid digestion and particle separation. Using pretreatment techniques, the identification and quantification of ENPs in wastewater samples with high organic materials, soluble metal ions, and coarse nonNPs become challenging [76]. The most popular pretreatment methods for detecting metal oxide NPs are hot-plate and microwave acid digestions. To digest water/wastewater samples containing TiO2, nitric and sulfuric acids are often used [77].
Choi et al. [75] summarized a review of the current literature on related topics, revealing that overall, existing methods focused mostly on single ENPs and were mainly applicable to samples with low organic loadings or under well-controlled laboratory conditions in wastewater treatment systems. In another study, Khosravi et al. [32] used ammonium persulfate and nitric acid in the melting process of titanium; however, for the dissolution of Ag-NPs, a pure nitric acid solution or aqua regia was used in the hot-plate and microwave acid digestion processes. The weak oxidation ability of acid digestion enables the treatment of wastewater samples with a high organic load and NP concentration. Owing to acid digestion, NPs in samples can be quantified using different analytical tools. However, if ENPs are not separated from soluble metals, the risk of overestimating the ENP concentration exits. When filtration is performed after acid digestion and before ICP analysis, it still cannot prevent the overestimation of NP concentration owing to the influence of coarse particles. Direct particle extraction is widely used as a popular pretreatment process. For example, cloud point extraction and ion exchange resins have shown promising results in separating Ag-NPs from wastewater. However, the organic load in the analyzed wastewater samples was unexpectedly low [78]. The results of extracting Ag-NPs from samples with a large organic load (such as wastewater sludge) were negligible. It is relatively easy to extract CNTs or fullerenes from water because they are nonmetals. However, if the organic load in a sample is large, it becomes challenging to remove carbon-based NPs. With or without an additional pretreatment, ENPs have been separated from wastewater via membrane filtration and centrifugation. Although this method is useful for extracting small scales, at high organic load concentrations, organic materials will adhere to NPs. Thus, this method cannot effectively separate the total mass of ENPs in samples. The most commonly used methods for quantitatively measuring the metalloid forms of NPs are ICP-MS, ICP emission spectrometry, and graphite furnace atomic absorption spectrometry [75]. In a study, these three elemental detection methods showed high recognition rates under various particle concentrations [79].
The aforementioned SiO2–tetraethylenepentamine process has also been used in sewage treatments [67]. For the same contact period, the removal performance of Ag-NPs steadily improved as the SiO2–tetraethylenepentamine concentration increased. When the SiO2–tetraethylenepentamine concentration was increased, the functional groups interacting with Ag-NPs and the contact region increased, improving the Ag-NP removal performance. Silver chloride NPs were almost completely removed when the SiO2–tetraethylenepentamine concentration was increased to 66.7 mg/L (for 134.8-mg/L silver chloride NPs), achieving the removal efficiency of 98.3%. In other words, to fully dissolve Ag-NPs (134.8 mg/L) in 35 min, at least 66.7-mg/L SiO2–tetraethylenepentamine should be present [17]. In any case, the concentration of the coagulant should be determined based on the initial concentration of Ag-NPs and the optimal removal period. Low concentrations and long removal times necessitate the use of a less concentration of the coagulant. Under the same experimental conditions, such as pH, the presence of sodium chloride, stirring, and the initial concentration of Ag-NPs, only 4.5-mg/L SiO2–tetraethylenepentamine was needed and Ag-NPs could be fully extracted within 24 h. Clearly, compared with other coagulants, SiO2–tetraethylenepentamine showed a considerably higher removal potential for activated sludge particles.
The concentration of Ag-NPs seemed to exceed their original quantity, which can be attributed to the Ag-NP fusion caused by SiO2–tetraethylenepentamine [30]. Coagulation mechanisms are often dynamic, and there is rarely a simple factor [79]. Several factors contributed to the removal of Ag-NPs during the SiO2–tetraethylenepentamine phase. Ag-NPs and SiO2–tetraethylenepentamine charge the neutralization in tetraethylenepentamine owing to an interaction between Ag and the metal ligand of the N atom. The special force between N and Ag atoms is well understood [39]. SiO2–tetraethylenepentamine strongly prefers to react with Ag-NPs regardless of the pH. The silanol group and Ag-NP stabilizer form a hydrogen bond. Although some silanol groups react with 3-chloropropyl trimethoxy silane, other silanol groups can form hydrogen bonds with the carboxylic acid groups of sodium citrate, serving as an Ag-NP stabilizer. Because of the aforementioned mechanisms, large amounts of Ag-NPs can simultaneously interact with SiO2–tetraethylenepentamine. Because of its solid state and broad scale, SiO2–tetraethylenepentamine can act as a “bridge” and facilitate the rapid solidification of Ag-NPs. All the above processes aid the adsorption of Ag-NPs using SiO2–tetraethylenepentamine [30].
Polyaluminium ferric chloride (PAFC) combines the advantages of aluminum and iron salt coagulants. As a high-molecular-weight organic flocculant, cationic polyacrylamide (CPAM) has a good removal effect on flocculated negatively charged colloids [80]. The flocculation efficiencies of PAFC and PAFC/CPAM for three initial concentrations (0.2, 2, and 30 mg/L) of TiO2-NPs in pure water and kaolin suspension systems (kaolin concentration: 30-mg/L) were investigated under different coagulant concentrations, pH values, precipitation times, and hydraulic conditions. Results showed that the TiO2-NP removal rate in the kaolin suspension system was slightly higher than that in the pure water system and the TiO2-NP removal rate obtained using PAFC/CPAM was considerably higher than that obtained using only PAFC [80]. The removal efficiency of TiO2-NPs increased as the initial concentration of TiO2-NPs increased. Under the optimal conditions, the removal rates obtained using PAFC/CPAM for 30-mg/L TiO2-NPs in pure water and kaolin suspension systems were 97.98 and 98.62%, respectively. Studies on the fractal dimension of flocs showed that the flocs produced using PAFC/CPAM exhibited a larger fractal dimension than those produced using only PAFC [81]. The corresponding study demonstrated that water turbidity improved the removal efficiency of TiO2-NPs and enhanced coagulation can efficiently remove TiO2-NPs from water.
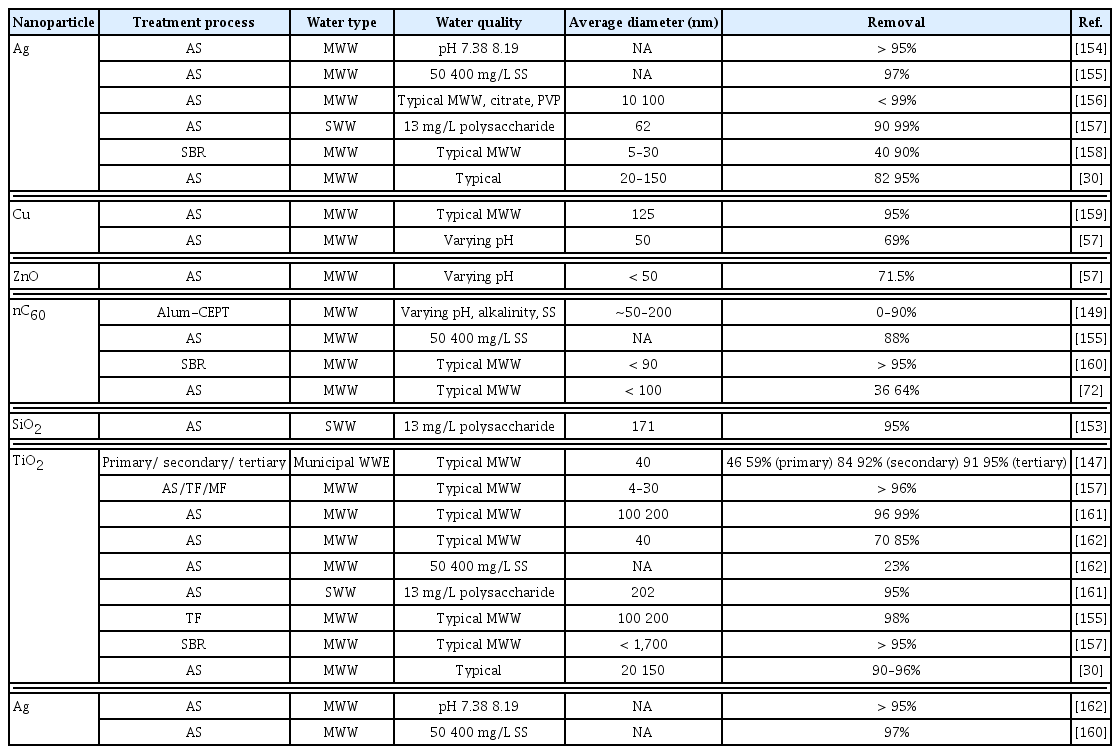
The optimal coagulant concentration was obtained by removing three mass concentrations of TiO2-NPs [80]. The optimal concentrations of PAFC and CPAM were 60 and 3 mg/L, respectively. The turbidity and TiO2-NP removal rates using PAFC and PA FC/CPAM increased as the coagulant concentration increased. When the concentration exceeded the optimal critical point, the removal effect of PAFC considerably decreased, whereas that of PAFC/CPAM remained relatively stable. Generally, the removal effect of TiO2-NPs at a high initial mass concentration exceeded that of TiO2-NPs at a low initial mass concentration [82]. Under the same flocculation conditions, the addition of kaolin improved the removal rate of the two NP materials. When the only TiO2-NP system was used, with initial mass concentrations of 0.2, 2, and 30 mg/L, the maximum removal rates of TiO2-NPs using PAFC were 65.9, 90.7, and 93.0%, respectively. The removal efficiency of TiO2-NPs improved in the presence of kaolin. After adding kaolin to the TiO2 solution at an initial concentration of 30 mg/L, the maximum removal rate of TiO2-NPs using PAFC increased from 93.0 to 97.8%. When only the TiO2-NP system was used, at an initial TiO2-NP concentration of 30 mg/L, the TiO2-NP removal rates using PAFC and with and without CPAM were 93.0 and 97.9%, respectively. The turbidity removal rate was consistent with the TiO2-NP removal rate. When the optimal critical point was exceeded, the removal rate of turbidity and TiO2-NPs decreased as the flocculant concentration increased. Table 3 summarizes the removal efficiency of selected ENPs in wastewater treatment processes.
5. Conclusion and Areas of Future Study
In the natural environment, ENPs are highly dynamic. It is necessary to consider the fate of each ENP component and understand the global fate of nanomaterials. Most ENPs penetrate the soil and flow into water treatment plants via groundwater, whereas some ENPs enter the biological cycle. With continuous developments in the nanotechnology industry, the environmental burden has increased considerably. In this review, both conventional and advanced methods developed in the last five years (2016–2021) for removing NPs from drinking water and wastewater are described in detail. Findings have shown that the removal of NPs varies significantly depending on both conventional and advanced treatment processes in both water and wastewater treatment plants. During coagulation and flocculation the characteristics of the water substantially affect the NP removal efficiency; for example water containing hydrophobic natural organic matter requires additional coagulant concentrations than water containing hydrophilic natural organic matter. Development of new adsorbents could greatly enhance the safe use of ENPs when in contact with water treatment materials. Furthermore, the removal mechanisms of various composite filtration membrane materials are discussed, and relevant opinions are suggested to considerably improve the reuse rate of these adsorbents and membranes and increase the removal rate of various NPs in waters with different qualities using low-pressure membranes.
Although the risks of using nanomaterials are well known, several uncertainties persist. Because of environmental exposure of nanomaterials, there are potential risks to the environment and human health that cannot be ignored. Therefore, it is necessary to further integrate technological innovations and risk assessments to ensure full access to technological benefits, minimize possible negative impacts, and ensure the long-term sustainability of the nanotechnology industry. Based on existing research, further studies must focus on expanding the applicability to different pollutants, improve recovery and enhance the reusability of NPs, and increase the efficiency of various methods for treating contaminated water. In the future, research attention should be directed toward investigating methods for removing mixed NPs in water. Employing these methods in water treatment apparatus requires designing an optimal removal system, with the possibility of subsequent scaling to larger volumes of treated water, which is already in progress. Additionally, to address pollution because of new ENPs, comprehensive experiments should be conducted to determine the most suitable removal method and minimize potential harm to the environment and human health. Extensive research activities on removing ENPs in the water environment and strict laws formulated by government agencies can have a far-reaching impact on identifying and preventing ENP contamination.
Acknowledgments
This study was supported by the University of South Carolina ASPIRE program. This work was also supported by the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (No. 2021R1A2C2003734).
Notes
Author Contributions
C.Y. (M.S. student) performed the literature review and wrote the paper, edited the manuscript. C.P. (Associate Professor) and Y.Y. (Professor) conceived and designed the study, reviewed and edited the manuscript. S.K. (post-doc) reviewed and edited the manuscript.
M.J. (faculty) reviewed and edited the manuscript. All authors read and approved the manuscript.