1. Introduction
During biological nitrogen removal from wastewater, ammonia nitrogen (NH4-N) is oxidized sequentially to nitrite nitrogen (NO2-N) by ammonia oxidizing bacteria (AOB) and then to nitrate nitrogen (NO3-N) by nitrite oxidizing bacteria (NOB) under aerobic conditions (nitrification), and following NO3-N is reduced to NO2-N, nitric oxide, nitrous oxide (N2O) and nitrogen gas (N2) by denitrifiers under anoxic conditions (denitrification). Particularly, characterized by alternative anoxic for denitrification and aerobic for nitrification, the intermittent aeration process has been widely applied for enhancing nitrogen removal from wastewater. Alternative anoxic and aerobic (AO) conditions can be realized timely within one sequencing batch reactor (SBR) as the intermittent aeration SBR (IASBR) or spatially in sequential reactors as the multiple anoxic and aerobic process [1]. For examples, total inorganic nitrogen (TIN) removal percentage in IASBR was 38.7% higher than in the AO SBR system [2]. In the intermittent aeration process, simultaneous nitrification and denitrification (SND) and denitrification with internal organic carbon could be enhanced [2]. In addition, AOB stimulation and NOB inhibition were observed under transition from anoxic to aerobic conditions, inducing partial nitrification [3]. However, N2O emission may be triggered with the short-cut nitrogen removal process because NO2-N is the precursor of N2O production and the inhibitor of nitrous oxide reductase (Nos) [4]. Therefore, N2O emission in the intermittent aeration process should be well evaluated.
N2O has a high greenhouse potential, with 300 times that of CO2. 0.03–18% of influent total nitrogen (TN) was emitted as N2O in intermittent aeration processes (lab-scale and full-scale), and the emission varied depending on the type of treatment processes, water quality, anoxic/aerobic period ratio and dissolved oxygen (DO) concentration [1]. Three pathways are responsible for N2O emission during wastewater treatment, including AOB denitrification, hydroxylamine oxidation and heterotrophic denitrification [5]. AOB denitrification was prominent under high NO2-N concentrations [6]. Especially, N2O emission was stimulated under low DO, high NO2-N concentration and with the heterotrophic activity in the intermittent aeration process [7, 8]. In the presence of heterotrophs, microbial competition among AOB, NOB and heterotrophs would be promoted, resulting in more N2O emission, which was highly dependent on the DO concentration [7]. Therefore, the selection of predominant microorganisms with the ability to reduce N2O was a key point to mitigate N2O emission. There was no consensus on what kind of nitrifiers in terms of r/K-strategist benefiting N2O mitigation. In a duckweed treatment ponds treating simulated stormwater, the growth of K-strategist (with a high substrate affinity and a low half saturation constant) may be responsible for low N2O emissions [9]. In contrast, N2O production was 25 times higher in completely stirred tank reactor (CSTR)-enriched biomass (K-strategist AOB) than in SBR-enriched biomass (r-strategist AOB) [10]. The mechanism behind this phenomenon needs to be further investigated.
SBR and CSTR have distinct regimes supplying wastewater for microorganisms and therefore may lead to diverse microbial community [10–12]. Due to the continuous organic supply pattern, system with the continuous feeding mode might benefit denitrifiers, especially for N2O-reducing related denitrifiers, resulting in low N2O emission [11]. In addition, SBR and CSTR regimes support different predominant AOB (K-strategist and r-strategist with different growth rates or substrate affinity constants) exhibiting distinct biokinetic properties [12]. SBR can select r-strategist AOB, while CSTR can select K-strategist AOB. Halophilic and halotolerant Nitrosomonas lineage were more highly enriched in SBR, whereas Nitrosospira spp. in CSTR [10]. As a result, the N2O emission characteristics may be entirely different. K-strategists of Nitrosomonas oligtropha and Nitrosospira spp. had a high potential in N2O production at lower NH4-N concentrations [13]. Additionally, SBR with high substrate gradients during the reaction cycle, could lead to more N2O emissions compared to CSTR [14]. N2O production during denitrification could be controlled by altering NO2-N feeding strategy. Compared to pulse feeding, N2O emission from the step-wise feeding and continuous feeding reactors were reduced by 3.1–4.2 and 8.2–11.7 folds, respectively [15]. Hence, continuous feeding might be efficient to minimize N2O emission. Taken together, a big controversy is that SBR regime may support fast-growing AOB and therefore favor the nitrogen removal but with high N2O emission potential. The discrepancy under these two regimes in terms of N2O emission and nitrogen removal needs to be in-depth studied.
This study aimed to investigate nitrogen removal and N2O emission induced by microbial community from two types of intermittent aeration processes operated under high and low DO conditions. Nitrification and denitrification activities were examined under different DO concentrations and electron acceptors, respectively. Furthermore, microbial community diversity and abundance and their correlation with N2O emission were analyzed.
2. Materials and Methods
2.1. SBR and CMR Systems
Two 27-L continuous-flow multiple AO reactors were operated under high DO (CMR-H) and low DO conditions (CMR-L). The system consisted of seven sequential reaction tanks, including anaerobic (3 L), aerobic I (3 L), anoxic I (3 L), aerobic II (3 L), anoxic II (3 L), aerobic III (3 L) and settling tank (7 L). The anoxic tanks were mixed by mechanical stirrers. The sludge recirculation ratio and the internal recycling ratio were both 100%. The DO concentration in the aerobic III tank was controlled at 1 mg/L in CMR-L and 2 mg/L in CMR-H.
Two 8-L SBRs were operated also under high DO (SBR-H) and low DO conditions (SBR-L). Both SBRs included four cycles per day and each cycle consisted of a 120-min anaerobic phase, a 120-min intermittent aeration phase with aerobic I (30 min), anoxic I (30 min), aerobic II (30 min) and anoxic II (30 min), a 60-min aerobic phase (aerobic III) and a 60-min settling phase. During aerobic phases, DO concentrations was controlled at 1 mg/L in SBR-L and 2 mg/L in SBR-H. In each cycle, 4 L of treated supernatant was discharged and 4 L of synthetic waste-water was pumped into the reactor by peristaltic pumps.
All reactors were inoculated with activated sludge taken from a domestic wastewater treatment plant in Shenzhen, China. Sludge retention time and hydraulic retention time were 15 d and 12 h, respectively. All reactors were operated under room temperature (24 ± 0.5°C).
The synthetic wastewater consisted of 510 mg/L sodium acetate (NaAc), 153 mg/L NH4Cl, 14 mg/L CaCl2·2H2O, 90 mg/L MgSO4·7H2O, 46 mg/L Na2HPO4, 10 mg/L yeast extract, 100 mg/L NaHCO3 and 0.4 mL/L trace elements. The composition of trace elements was according to Smolders et al. [16]. The synthetic wastewater contained around 400 mg/L of chemical oxygen demand (COD) and 40 mg/L of NH4-N.
2.2. Nitrifying Batch Experiments
Nitrification batch experiments were conducted to examine nitrifying activities and N2O emission characteristics. The examined conditions included effects of DO concentrations (2.0 and 1.0 mg/L) and the presence of heterotrophic activity.
800-mL activated sludge was taken from the aerobic III tank/phase in CMR or SBR systems, respectively. After settlement, supernatant was discarded and the activated sludge was resuspended with 800 mL synthetic wastewater without the addition of NaAc and NH4-N. During each batch experiment, 800 mL of mixed liquor was divided into two parts and filled into batch reactors. The set DO concentrations in batch reactors were controlled by adjusting aeration rates of air and N2. For examining the effect of DO on the ammonia oxidation activity, 30 mg/L of NH4-N was dosed. For examining the effect of DO on nitrite oxidation activity, 20 mg/L of NO2-N was initially dosed. For examining the effect of DO with the presence of heterotrophic activity on ammonia oxidation activity, 30 mg/L of NH4-N and 500 mg/L of NaAc were applied. For examining the effect of DO with the presence of heterotrophic activity on nitrite oxidation activity, 20 mg/L of NO2-N and 500 mg/L of NaAc were applied.
Each experiment was carried out in duplicate. During all batch experiments, gas and liquid samples were taken at intervals of 10 min to analyze concentrations of N2O in the gas phase and NH4-N, NO2-N and NO3-N in the liquid phase. Suspended solids (SS) and volatile suspended solids (VSS) were measured before the beginning of each batch experiment. Liquid N2O, DO concentrations and pH were measured on-line.
2.3. Denitrifying Batch Experiments
Denitrifying batch experiments were conducted to examine the activity of denitrifiers and N2O emission under conditions with electron acceptors of NO2-N and NO3-N and electron donors of NaAc and polyhydroxyalkanoates (PHAs), respectively.
For denitrification with PHAs, 800-mL activated sludge was taken from the anaerobic tank. Then, it was divided into two parts and filled into two batch reactors. After stripping with N2, batch experiments were started after the addition of 20 mg/L of NO2-N or 30 mg/L of NO3-N, respectively, to investigate the effect of electron acceptor on the denitrifying activity and N2O emission.
For denitrification with NaAc, 800-mL activated sludge was taken from the aerobic III tank/phase in CMR and SBR systems. After settlement, supernatant was discarded and the activated sludge was resuspended with 800 mL synthetic wastewater without the addition of NaAc. 800 mL of mixed liquor was divided into two parts and filled into two batch reactors. After stripping with N2, batch experiments were started with the addition of 20 mg/L of NO2-N and 500 mg/L of NaAc, or 30 mg/L of NO3-N and 500 mg/L of NaAc, respectively, to investigate the effect of electron acceptor on the denitrifying activity and N2O emission.
Each experiment was carried out in duplicate. During all batch experiments, gas and liquid samples were taken at intervals of 10 min to analyze concentrations of N2O in the gas phase and NO2-N and NO3-N in the liquid phase. SS and VSS were measured before the beginning of the batch experiment. Liquid N2O, DO concentrations and pH were measured on-line.
2.4. Analytical Methods
NH4-N, NO3-N, NO2-N, SS and VSS were measured according to standard methods [17]. DO and pH were measured by a portable DO meter (Flexi, HACH, USA) and a pH meter (pH 30d, HACH, USA), respectively. N2O was tested by a gas chromatograph (GC-9720, Fuli, China) with an electron capture detector and a HP-PLOT/Q column (J&W GC Columns, Agilent Technologies, USA) according to Wang et al. [7]. The N2O emission factor was calculated according to Kimochi et al. [18]. The efficiency of SND was calculated based on Wang et al. [7].
DNA was extracted from activated sludge using a Fast DNA Spin Kit (Laboratories, Inc., Carlsbad, USA) according to the manufacturer’s instruction. The DNA sample was amplified by polymerase chain reaction in the V4 region of the target fragment 16S rRNA gene, and analyzed by high-throughput sequencing using the Illumina Miseq platform. The primers were 515F and 806R [19]. After screening, species classification of the representative spliced sequences for each OTU was performed using the Qiime software. Results were classified to the phylum, class, order, family and genus levels. The taxonomies for 16S rRNA microbial communities had been deposited in the NCBI BioProject database with the accession ID of PRJNA342302.
3. Results and Discussion
3.1. Performance of SBR and CMR Reactors
Table 1 shows the performance of SBR and CMR systems under steady state. Under steady state, the SS/VSS in SBR-L, SBR-H, CMR-L and CMR-H were 3.50/2.81, 3.88/3.12, 3.34/2.68 and 3.44/2.75 g/L. Four systems achieved desirable removal efficiencies for NH4-N (99.3–99.7%). CMR-L achieved the best effluent quality for TIN, with the NO3-N concentration of 3.55 mg/L and NH4-N of 0.28 mg/L, and the corresponding removal efficiencies of TIN and NH4-N were 91.5% and 99.3%, respectively. In contrast, the highest effluent NO3-N concentration of 10.46 mg/L was obtained in SBR-H, with the TIN removal efficiency of 75.2%. TIN removal efficiencies under low DO conditions were obviously higher than under high DO conditions, which might be due to high SND efficiency under low DO conditions [20]. In addition, TIN removal efficiencies under CMR conditions were higher than that under SBR conditions. Hence, CMR systems with a low DO condition benefited nitrogen removal. Chen et al. [21] also obtained that enhanced nitrogen removal was achieved in a continuous-flow intermittent aeration process with low DO (0.3–1.0 mg/L), with the TN removal efficiency up to 94.9%. While in a continuous plug-flow step feed process with the intermittent aeration (DO = 1.5–2.0 mg/L), TN removal efficiency of 86% was achieved [3].
3.2. Nitrifying Activity and N2O Emission
Nitrifying activity was tested under different DO concentrations and in the presence of organic carbon, with results shown in Table 2.
With increasing DO concentrations from 1.0 mg/L to 2.0 mg/L, activities of AOB increased, while that of NOB was less affected. Correspondingly, the ratio between nitrite oxidizing rate (rNOB) and ammonium oxidizing rate (rAOB) decreased from 1.06–1.37 to 0.74–1.08. Importantly, in the presence of organic carbon, NOB activity was significantly suppressed, independent of DO concentrations. The rNOB/rAOB ratio markedly reduced from 1.06–1.37 to 0.47–1.21 under DO of 1 mg/L and from 0.74–1.08 to 0.52–0.98 under DO of 2 mg/L. The balance between AOB and NOB could be destroyed in the presence of organic carbon, which might result in NO2-N accumulation in the intermittent aeration process [2]. With organics, competition for oxygen between heterotrophs and nitrifiers would be promoted, with the suppressed nitrifying activity [22]. Furthermore, the suppression of NOB in SBR systems was more remarkable (with relatively low rNOB/rAOB ratio) in comparison with CMR systems, especially in the presence of organics. The result was consistent with Wang et al. [2], who demonstrated that the rNOB/rAOB ratio was 0.32 in the multiple AO SBR process, which was lower than 0.98 of the AO SBR process [2]. In the presence of organics, AOB activity was inhibited in sequencing batch biofilm reactors, where the rNOB/rAOB ratio was 0.21–1.08 without and 0.25–1.25 with the addition of organics [23].
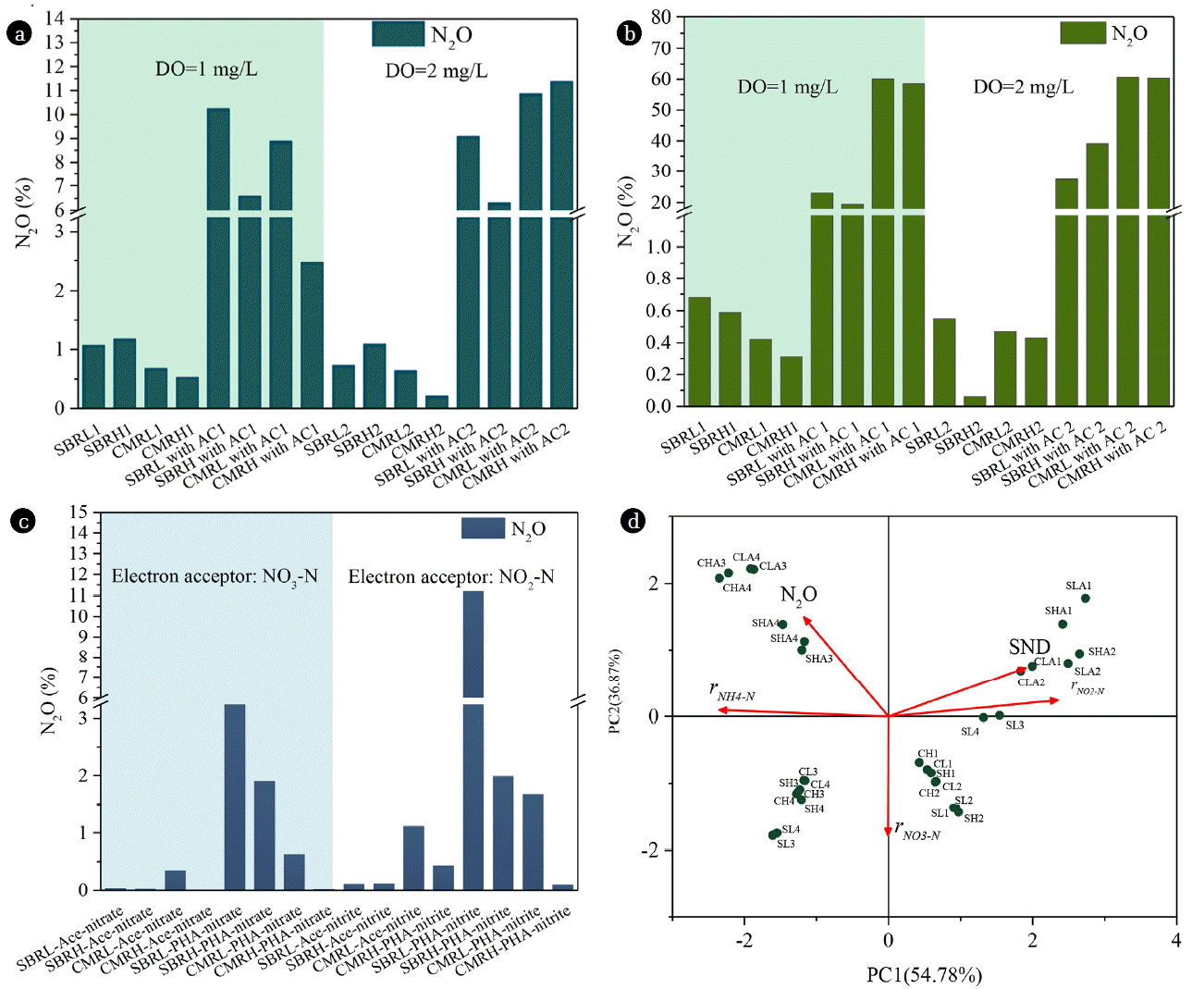
As shown in Fig. 1(a), N2O emission was triggered during ammonia oxidation in the presence of organic carbon. Herein, N2O emission factors ranged from 0.21–1.18% in the absence of organic carbon, which were significantly lower than those with the presence of organic carbon (2.47–11.39%). Particularly, in the absence of organic carbon, SBR systems possessed higher N2O emission potential (with N2O emission factors of 0.73–1.18%) compared to CMR systems (with N2O emission factors of 0.21–0.68%). In the presence of organic carbon, however, more N2O emission occurred in CMR systems compared to SBR systems under the DO of 2 mg/L. SBR-L and CMR-L had higher N2O emission potential (with 10.24% and 8.89% of N2O emission factor) comparing to SBR-H and CMR-H (with 6.60% and 2.47% of N2O emission factor) under DO of 1 mg/L. From Fig. 1(d), high SND efficiency under low DO conditions might be the reason responsible for this phenomenon. N2O emission was positively correlated with rNH4-N. Besides, low DO concentrations (less than 0.2 mg/L) facilitated N2O generation from AOB denitrification [24]. N2O from AOB denitrification pathway decreased as DO concentration increased from 0.35 to 3.5 mg/L [6]. Interestingly, SBRL and CMRL possessed the higher N2O emission factors comparing to SBRH and CMRH when the oxygen concentration was limited (1 mg/L), suggesting that oxygen was the determining factor affecting N2O emission. However, CMR systems exhibited high N2O emission when oxygen concentration was not limited (2 mg/L). The supply regime might be the decisive factor under this condition. Besides, the N2O emission factors observed under this condition were higher than that under DO of 1 mg/L, which further demonstrated that CMR affected the N2O emission intrinsically rather than DO. This was consisted with the result that N2O emission during nitrification correlated with rNH4-N (Fig. 1(d)) and the lower rNH4-N in CMR. The microorganisms’ selection by CMR system might be the underlying reason for N2O emission, which was in line with Terada et al. [10] that CSTR predominantly enriched slow-growing (K-strategist) AOB, contributing to more N2O production.
In the presence of organics, heterotrophic activities affected N2O emission during nitrification significantly. The N2O emission obtained from this study (2.47% – 11.39%) was within the 0.70% – 14.50% from Shen et al. [25] and obviously lower than 15% (the ratio between the N2O production rate and the ammonia oxidation rate) from Wunderlin et al. [5]. Presumably, heterotrophic activities might decrease the DO availability for AOB. Therefore, AOB denitrification pathway would be enhanced.
For N2O emission during NO2-N oxidation (Fig. 1(b)), 0.06% – 0.68% of NO2-N was emitted as N2O in the absence of organic carbon independent of DO concentrations, whereas N2O emission was stimulated in the presence of organic carbon, with N2O emission factors of 23.2% – 60.7%. Besides, N2O emission under DO = 2.0 mg/L conditions in all four systems was higher than that under DO = 1.0 mg/L, which was likely a result of activity inhibition of Nos by the high DO concentrations [15]. CMR systems possessed more N2O emission capacities (with 58.54% – 60.70% of N2O emission factors) independent of organic carbon compared to SBR systems (with 19.45% – 39.08% of N2O emission factor). It’s worth mentioning that higher N2O emission under DO = 2.0 mg/L might be triggered by air stripping. Under anoxic or low DO conditions, N2O produced could be reduced in addition to emitted. However, under stripping conditions, N2O emission was triggered significantly and N2O emission amount was increased by 6-fold compared to that without stripping [26].
Compared with ammonia oxidation, N2O emission from NO2-N oxidation might be from heterotrophic denitrification as no NO3-N production occurred. Under the 1.1 mg O2/L condition, heterotrophic denitrification could be responsible for 40% of the N2O production based on Tallec et al. [27]. NO2-N heterotrophic denitrification was responsible for 60% of N2O production during partial nitrification [28]. The present study demonstrated that heterotrophic denitrification dominated the N2O emission during NO2-N oxidation because the addition of organics. Based on Wang et al. [29], the organic utilization efficiency of heterotrophic denitrifiers might be a regulator for N2O emission. Thus, the high N2O emission capacity in CMR systems was attributed to less abundant heterotrophs reducing N2O to N2.
3.3. Denitrifying Activity and N2O Emission
Denitrifying activities were examined with NO2-N or NO3-N as the electron acceptor and acetate or PHA as the carbon source, with results shown in Table 3. SBRs had a higher denitrifying rate when NO3-N was used as the electron acceptor, independent of carbon sources. In contrast, a higher denitrifying rate was observed in SBR systems with NO2-N and acetate as the electron acceptor and donor, respectively. Overall, SBRs had a higher denitrifying rate regardless of carbon types and electron acceptors. The possible reason could be due to that SBR enriched more abundant denitrifiers. Specially, higher denitrifying rates with NO2-N were observed in CMR-L with acetate as the organic carbon, meaning that denitrifiers enriched in CMR, especially under low DO conditions, were liable to utilize NO2-N as the electron acceptor. SBR (especially SBR-H) facilitated denitrification with PHAs as electron donor due to the higher storage capacity of PHAs under pulse feeding conditions compared to continuous feeding conditions [30].
N2O emission was stimulated in SBRs with PHAs as the electron donor. As shown in Fig. 1(c), SBR-L emitted more N2O when using PHAs, especially with NO2-N as the electron acceptor. In addition, SBR with low DO concentrations were prone to emit more N2O, which was likely a result of less abundant of N2O reduction related denitrifier [11]. SBR owned less abundant N2O-reducing denitrifiers compared to continuous feeding systems. The PHAs-type denitrification (with NO2-N) caused more N2O emission, likely due to the slow degradation rate of PHAs inducing the competition for electrons between denitrifying enzymes [23, 31]. Denitrifiers of IASBR preferred to utilize PHAs for denitrification [2]. As a result, more N2O emission was observed for denitrifiers in SBR.
NO2-N triggered the N2O emission especially for SBR-L and CMR-L. Lemaire et al. [32] also confirmed that the addition of NO2-N led to five times higher net N2O production rate compared to the addition of NO3-N. Besides, the difference in the rate of NO3-N and NO2-N reduction could also lead to the accumulation of N2O. Free nitrous acid (FNA) might be the true inhibitor because FNA could bind to the active sites of copper-contained N2O reductase, resulting in the inhibition to the N2O reduction. Fifty percent inhibition concentration of FNA was 0.0007–0.001 mg HNO2-N/L, equivalent to approximately 3–4 mg NO2-N/L at pH 7 [33]. For denitrifying polyphosphate accumulating organisms (DNPAOs), denitrifying NO2-N to N2O was enhanced as soon as NO2-N increased to above 1 mg/L [34]. Batch experiments showed that four systems enriched DNPAOs because NO2-N or NO3-N and PO4-P could be reduced simultaneously (data not shown). Presumably, DNPAOs might be better enriched under low DO conditions (SBR-L and CMR-L), leading to more N2O emission.
3.4. Microbial Community Diversity and Abundance in SBR and CMR Reactors
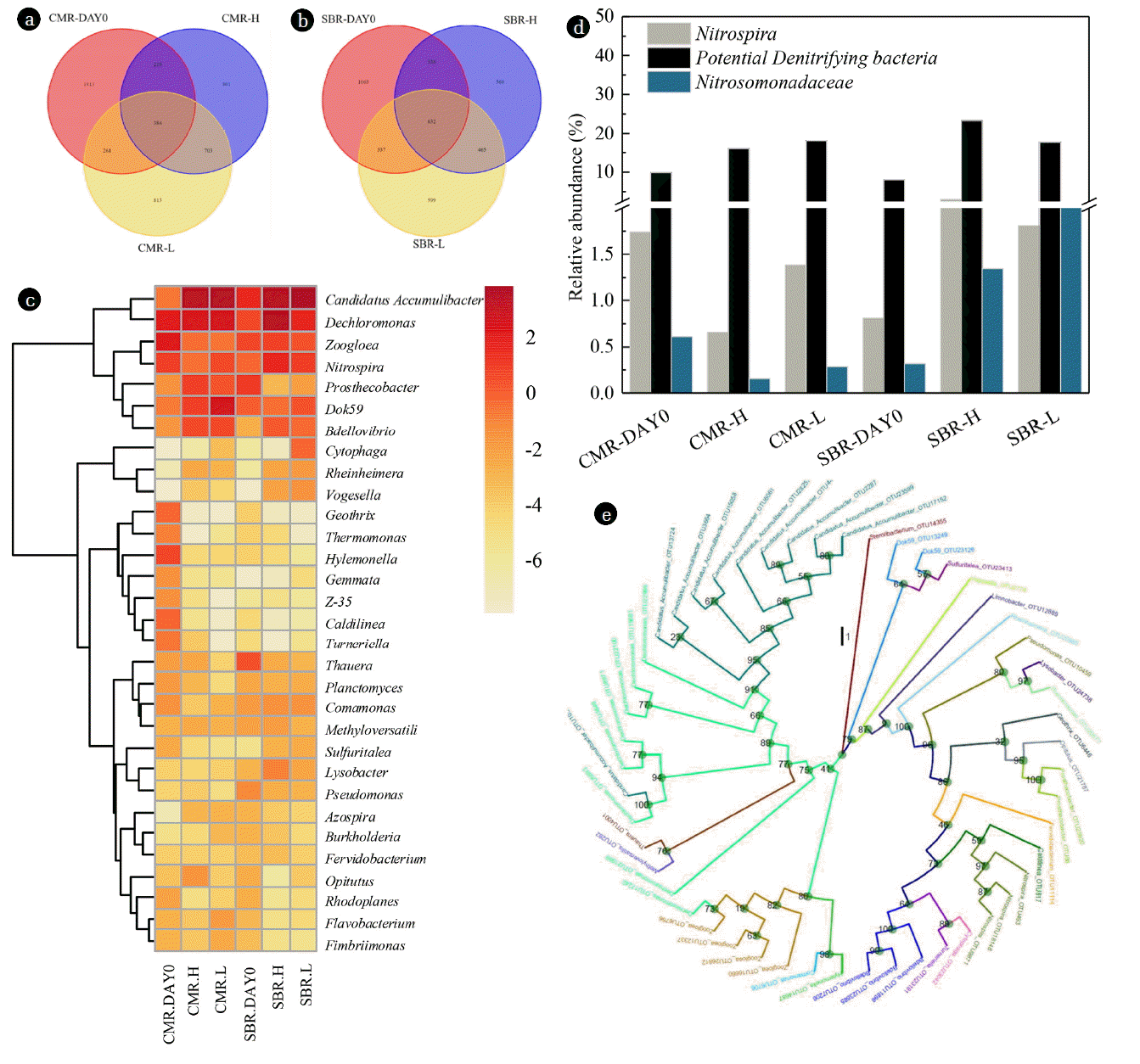
Table 4 depicts the microbial community abundance (Top 30) and diversity in four reactors. CMR-L had the highest microbial diversity, whereas SBR-H had the lowest diversity. In addition, from Figs. 2(a) and 2(b), 7.7% and 15.8% of species were the same among CMR and SBR conditions, respectively, indicating that SBR could maintain more species from the seed. 34.1% and 42.3% of species were common between CMR-H and CMR-L and between SBR-H and SBR-L, respectively. Noted that 813 and 801 unique species were enriched in CMR-L and CMR-H, while 599 and 560 species were enriched in SBR-L and SBR-H, suggesting that low DO and CMR conditions offered more favorable environment for microbial diversity. CMR systems could supply adequate nutrient supporting more organisms’ growth, resulting in diverse microbial community [11].
Detailed microbial community abundance was investigated at the genus level (Fig. 2(c)). There were significant distinctions between the seed and the acclimated sludge in CMR reactors, however, only slight shifts in the distribution of genus was observed between the seed and the SBR sludge. In CMRs, the major genera in the seed were Dechloromonas (3.99%), Zoogloea (4.43%), Nitrospira (1.74%), Caldilinea (0.82%), Geothrix (0.81%), Dok59 (0.55%), Candidatus Accumulibacter (0.58%) and Turneriella (0.61%). Other minor genera included Prosthecobacter (0.32%), Bdellovibrio (0.29%), Thermomonas (0.49%), Z-35 (0.34%) and Gemmata (0.33%). The dominating genera in CMR-H and CMR-L were Candidatus Accumulibacter (9.76% and 11.26%), Dechloromonas (5.03% and 5.61%) and Dok59 (1.73% and 5.76%), followed by Prosthecobacter (1.69% and 1.14%), Bdellovibrio (1.32% and 1.43%), Zoogloea (0.72% and 0.63%) and Nitrospira (0.66% and 1.36%). As for microorganisms in SBRs, Candidatus Accumulibacter (2.93%), Dechloromonas (1.41%), Zoogloea (1.47%), Prosthecobacter (1.92) and Thauera (1.23%) were the major genera in the seed. Whereas Candidatus Accumulibacter (10.50% and 12.74%), Dechloromonas (10.29% and 3.10%), Dok59 (0.70% and 1.17%), Zoogloea (1.66% and 1.18%), Nitrospira (2.99% and 1.81%) and Bdellovibrio (1.03% and 0.75%) dominated in SBR-H and SBR-L. Candidatus Accumulibacter and Dechloromonas were the most abundant genera in all four reactors, which were polyphosphate accumulating organisms [35] or DNPAOs [36]. Dechloromonas survived in SBR-H, which was contrary to Liang et al. [11] that Dechloromonas adapted well to the continuous feeding system, which could be due to the enhancement of denitrifiers with PHAs in SBRs [2]. As a bacterial predator, Bdellovibrio could significantly shift the species composition in wastewater treatment systems, and might disturb the operational performance [37]. Prosthecobacter, Zoogloea, Thauera and Dechloromonas genera were commonly genera distributed in wastewater treatments plants [38, 39]. Prosthecobacter was thriving in CMR, which belongs to oligotrophic heterotrophs enriched in low-nutrient environments [40].
Nitrosomonadaceae family and Nitrospira genus were responsible for NH4-N oxidation and NO2-N oxidation, respectively. The abundances of Nitrosomonadaceae family and Nitrospira genus in SBRs (2.99% in SBR-H and 1.81% in SBR-L) were obviously higher than in CMRs (0.66% in CMR-H and 1.38% in CMR-L) (Fig. 2(d)). Compared to the seed (CMR-DAY0 and SBR-DAY0), different shifts were observed for Nitrosomonadaceae family and Nitrospira genus in SBRs and CMRs. These two nitrifiers could be well maintained in SBRs rather than in CMRs. This observation was consistent with Liang et al. [11] that abundant AOB (Nitrosomonadales) and NOB (Nitrospira) were confirmed in batch feeding systems in comparison with continuous feeding systems. Nitrospira, possessing a low maximum specific growth rate, was a K-strategist that could be well-adapted to low substrate concentrations [41]. Park and Noguera [42] suggested that Nitrospira community had a dynamic change under different DO conditions. Kim and Kim [12] concluded that Nitrospira genus was preferring to live in continuous reactors comparing to SBRs. Therefore, the different composition of these nitrifying genus (probably r-strategist in SBRs and K-strategist in CMRs) enriched in different feeding patterns and DO concentrations might result in different nitrifying activity and N2O emission characteristics. Nitrosomonas europaea within the Nitrosomonadales family, harbored nir (encoding nitrite reductase) and nor (encoding nitric reductase) genes while lack of nosZ (encoding nitrous oxide reductase) [43], was a source of N2O production. Some species within Nitrospira genus including Candidatus Nitrospira inopinata, Candidatus Nitrospira nitrosa and Candidatus Nitrospira nitrificans are known as Comammox that oxidize NH4-N to NO3-N. However, data about the above species was absent. Hence, further researches are needed to evaluate their contributions on nitrogen conversion. In addition, based on the 16S rRNA result, ammonia oxidizing archaea of Candidatus Nitrososphaera within Crenarchaeota pylum were detected in all four reactors, with abundance of 0% – 0.016%. Their contributions in nitrogen need to be further evaluated.
Diverse denitrifiers including Dechloromonas, Zoogloea, Thauera, Comamonas, Methyloversatilis, Flavobacterium and Pseudomonas were obtained (Fig. 2(e)). Total abundance of these potential denitrifiers was 9.85%, 16.05%, 18.09%, 8.03%, 23.34% and 17.74% in seed sludge of CMR (CMR-DAY0), CMR-H, CMR-L, seed sludge of SBR (SBR-DAY0), SBR-H and SBR-L (Fig. 2(d)). Relative high abundances were observed in SBR-H and CMR-L. This observation could be an explanation about high denitrifying activity in SBR-H and CMR-L. Denitrifiers enriched in CMR might prefer to utilize NO2-N as the electron acceptor, whereas prefer to NO3-N in SBRs, indicating diverse microbial denitrifiers enriched. Particularly, Methyloversatilis spp. was reported as incomplete denitrifiers (reducing nitrate or nitrite to nitrogen oxide intermediates instead of N2), and Pseudomonas, Thauera and Burkholderia were incomplete nitrite reducers (capable of reducing nitrite to N2O intermediates harbor cd1-type nitrite reductase) [44, 45]. Dechloromonas, Comamonas and Flavobacterium were complete denitrifiers [46–48]. Dechloromonas carried both nirS (encoding nitrite reductase) and nosZ genes [46]. Flavobacterium enriched in biofiltration systems was nosZ-containing bacterial genera [48]. Some members of Zoogloea could utilize glucose and sodium acetates to remove nitrate [49]. Strand et al. [50] reported that one Zoogloea genus could reduce NO3-N at the DO concentration up to 8 mg/L. Thauera was dominant in IASBR treating digested piggery wastewater [51]. Although Thauera was found to be the nosZ owner in an intermittent aeration process [8], some members of Thauera was found to be a partial denitrifier that produces N2O as the end-product [43].
Candidatus Accumulibacter, which was reported as DNPAOs, proliferated especially in CMR-L (11.26%) and SBR-L (12.74%). These bacteria might be partly responsible for N2O emission during denitrification. This might explain why N2O emission was stimulated during NO2-N denitrification. For N2O emission related denitrifying genera, Methyloversatilis, Thauera, Burkholderia and Pseudomonas, accounted for 0.44% in CMR-H, 0.32% in CMR-L, 0.61% in SBR-H and 0.41% in SBR-L. In contrast, Dechloromonas, Comamonas and Flavobacterium, which might be N2O reducers, accounted for 5.16% in CMR-H, 5.99% in CMR-L, 10.64% in SBR-H and 3.45% in SBR-L. The ratios between N2O producers and reducers were 0.09, 0.05, 0.06 and 0.12 in CMR-H, CMR-L, SBR-H and SBR-L, respectively. The lowest abundance of N2O-reducing microorganisms and the highest producer and reducers ratio of SBR-L could be a response for high N2O emission during denitrification. The low abundances of N2O reducers and the high microbial diversity in CMR systems might be the reason for high N2O emission during nitrification in the presence of organics.
4. Conclusions
The present study attempted to explore similarities and differences between SBR and CMR systems from nitrogen removal, N2O emission and microbial diversity. CMR systems had obviously higher nitrogen removal capacity over SBR systems. SBR systems enriched nitrifiers and denitrifiers with high specific activities. More N2O emission was observed for CMR during nitrification, whereas for SBR during denitrification. More microbial diversity was observed in CMR systems. The dominant DNPAOs and low abundances of N2O reducers might be responsible for N2O emission during nitrification and denitrification.