1. Introduction
Rhodamine B is a representative of xanthenes cationic dyes and is known for its stability [1]. It is harmful if swallowed and capable to cause irritation to the skin, eyes and respiratory tract. Photocatalytic oxidation is highly efficient in generating · OH for the degradation of the pollutants [2].
Several techniques have been developed to treat RB. Ozone-ultraviolet (O3/UV) photocatalytic oxidation is highly efficient in generating · OH for the degradation of pollutants [2]. The choice of photocatalysis is essential in RB removal, and TiO2 is a good candidate. TiO2-photocatalyzed reactions are non-selective oxidation processes in a level of microgram per liter, and the use of TiO2 is effective for inactive substrates [3]. Moreover, the presence of solid particles in TiO2 can provide additional nuclei for bubble formation, thereby enhancing the degradation efficiency of pollutants [4].
Compared with its powder form, TiO2 nanotubes have a larger Brunauer-Emmett-Teller (BET) surface area (SBET), which is advantageous in one-electron oxidation during photocatalytic reactions [5, 6]. However, the relatively large bandgap of TiO2 (3.2 eV) limits the efficiency of photocatalytic reactions because of the high recombination rate of photogenerated electrons and holes [7]. To solve this problem, metal ions were doped into the TiO2 nanotubes to narrow the bandgap of TiO2, as well as reduce the recombination rate of electrons and holes because of the generation of crystal defect caused by ion-doping [7–9]. In addition, the doped ions can act as shallow trapping sites for electrons and holes, and then reacted with H2O2, OH- and O2 to generate · OH and O2- · radicals.
In this study, the effect of metal ion-doping on the photocatalytic activity of TiO2 nanotubes for RB removal via photocatalytic oxidation was investigated. Metal ions, including Ag+, Al3+, Mn2+, Ni2+ and Zn2+, were investigated, and Zn2+ was observed to be the optimum type. Characteristic analyses of the TiO2 nanotubes were conducted to determine the factors that affect the photocatalytic activities of the catalysts.
2. Materials and Methods
2.1. Materials
P-25 TiO2 was provided by the Degussa Corporation (Richfield Park, USA). The powder has an SBET of 50 m2/g, an average crystallite size of 21 nm, and a crystal structure of 80% anatase and 20% rutile. RB (A.R.) was purchased from Beijing Chemical Reagent Company (China). Microporous membrane (0.45 μm) (MPM) was purchased from Agela Technologies (China).
A photocatalytic reactor (60 mm diameter and 750 mm length), which was equipped with a 37 W 254 nm UV lamp (15 mm Dia. And 793 mm Lth.) purchased from Haili Lighting Equipment Company (China), was used in this experiment. O2, which was generated by an oxygenator (Yaao, China), was used to produce O3 with the help of an ozone generator (Tonglin, China). O3 was guided into the photocatalytic reactor through the bottom port, and the samples were acquired from the sampling port in the middle of the reactor.
2.2. Preparation of TiO2 Nanotubes
One gram of P-25 TiO2 was added into 16 mL of a 10 mol/L NaOH aqueous solution in a Teflon vessel. The mixture was then stirred for 2 h at room temperature. The mixture was then heated at 110°C for 24 h in an oven, and cooled to room temperature in air. The as-prepared precipitate (Na2Ti3O7) was washed with distilled water to achieve a pH of 7 and then subsequently dispersed in a 0.1 mol/L HCl aqueous solution. After ultrasonication for 30 min, the precipitate was continuously washed to achieve a pH of 7 and then dried at 60°C. The nanotubes were prepared by calcining the precipitate at 400–600°C for 2 h.
The ion-doped TiO2 nanotubes were prepared based on the aforementioned process except for adding AgNO3 (0.0213 g), AlC13 (0.0167 g), Mn(NO3)2 · 4H2O (0.0313 g), NiC12 (0.0162 g) or ZnCl2 (0.0170 g) into the 10 mol/L NaOH solution. The amounts of doping ions which were added in the solution were 1.0% (atom percentage). According to the atlases of energy distribution, the doping concentrations of Ag+, Al3+, Mn2+, Ni2+ and Zn2+ ions were 0.251%, 0.618%, 0.498%, 0.459% and 0.372%, respectively. A 0.1 mol/L HNO3 solution was used instead of an HCl solution for the preparation of Ag+-doped TiO2.
2.3. Characterization of TiO2 Nanotubes
X-ray diffraction (XRD) patterns were collected using a Rigaku Dmax-RB diffractometer (Tokyo, Japan). The surface elemental composition of TiO2 was recorded via X-ray photoelectron spectroscopy (XPS, AXIS ULTRADLD, Kratos Analytical, UK). Transmission electron microscopy (TEM) images were obtained using a HITACHI HT-7700 electron microscope (Tokyo, Japan). SBET was measured using the Quadrasorb SI-MP apparatus (Quantachrome Instrument, USA). Diffuse reflectance spectroscopy (DRS) analyses were performed using a HITACHI U-3010 UV-vis scanning spectrophotometer (Tokyo, Japan).
2.4. Photocatalytic Activity Tests
Aqueous slurries were prepared by adding 0.15 g of TiO2-based photocatalyst to 1500 mL of 20 mg/L RB aqueous solution at pH 7. The aqueous slurries were stirred and bubbled with O3 (1 L/min, 10%) for 30 min and then irradiated with a 254 nm UV lamp. Every 10 min, a 20 mL sample was acquired and mixed with 0.5 mL of 0.5 mol/L Na2S2O3 solution to stop the reaction. The sample was filtered through an MPM to separate the catalyst. The concentration of RB was determined using a UV-visible spectrophotometer (DR5000, Hach).
3. Results and Discussion
3.1. Characterization of Photocatalysts
3.1.1. Morphology of the catalysts
Transmission electron microscopy was performed to observe the morphology of the catalyst. The morphologies of Zn2+-doped TiO2 nanotubes calcined at 550°C are shown in Fig. 1. The TiO2 nanotube was successfully synthesized. Other ion-doped nanotubes shared a similar morphology (figure not shown). The TiO2 nanotubes had an average diameter of approximately 10 nm with a hollow and open-ended structure, and the tube wall thickness was approximately 1 nm.
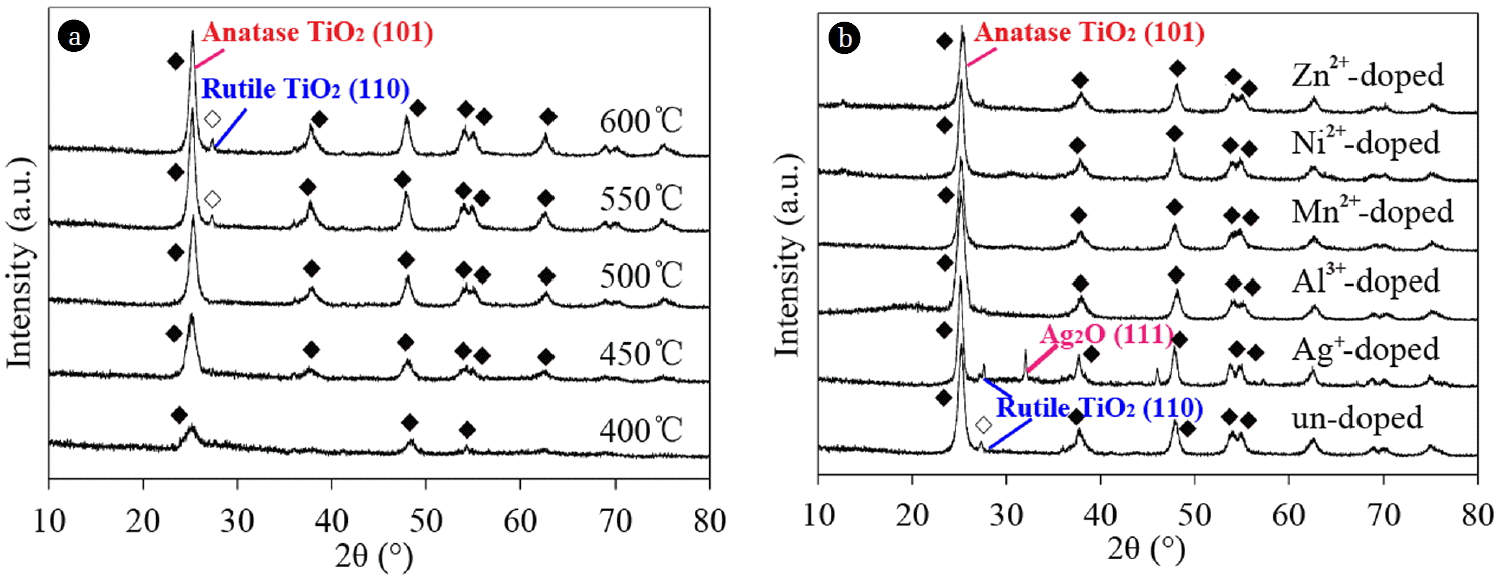
3.1.2. XRD analysis
The XRD patterns of the un-doped and ion-doped TiO2 nanotubes are shown in Figs. 3(a) and 3(b), respectively. The weight fractions of anatase in the anatase-rutile mixtures in the samples were calculated using Eq. (1) [10].
where f is the weight fractions of anatase in the mixtures, and IR and IA are the maximum X-ray diffraction peak intensities of rutile phase (110) and anatase phase (101), respectively. The weight fractions of anatase in various samples are listed in Table 1.
Fig. 2 and Table 1 show that both anatase and rutile phases might exist in the TiO2 nanotubes. When the calcination temperature was increased, the intensity of the anatase phase decreased, whereas that of the rutile phase increased. The dopant ions could substitute Ti4+ in the TiO2 nanotubes lattice, which resulted in the deformation of the crystal lattice. As a result, broadening diffraction peak and decreasing peak intensity were observed.
Among the dopant ions, only Ag+ could be observed in the XRD patterns in the form of Ag2O based on the characteristic peak of the (111) crystal plane of Ag2O at 32.8° and the XPS spectra of Ag 3 d [11]. Ag+ (ionic radius of 115 pm) could be observed because it had difficulty entering the TiO2 lattice [12]. Ions were partially dispersed in the bulk of TiO2 nanotubes [9]. The metallic oxides from other doped ions might also be formed on the surface of TiO2. However, these oxides were not observed in the XRD patterns because their metal sites (0.5 wt.% – 2.0 wt.%) were expected to be below the visibility limit of X-ray analysis [13].
3.1.3. XPS analysis
The XPS analysis was performed to determine the surface composition and the chemical state of the elements in the ion-doped TiO2 nanotubes calcined at 550°C (Fig. 3). Fig. 3(a) shows the XPS survey scan of the un-doped TiO2, and Figs. 3(b) – 3(k) show the XPS spectra of Ag 3d, Al 2p, Cu 2p, Fe 2p, Mn 2p, Ni 2p, V 2p, Zn 2p, Ti 2p, and O 1 s, respectively. The sharp XPS peaks for Ti, O, doped ions, as well as C and Na, are shown in Fig. 3(a). The presence of C was mainly attributed to carbon contamination when the samples were transferred to the XPS device [14]. The presence of Na 1s could be explained by the incomplete exchange of protons with Na+ during acid washing [9]. The results also indicate that a part of the catalyst still remained at the Na2Ti3O7 nanotubes where a lot of Na existed [15].
Fig. 3(a) shows that the peak intensities of Ag2O were much higher than those of other metallic oxides. Moreover, the Ag2O peak could be observed in the XRD patterns of Ag+-doped TiO2. Both observation results indicate that Ag2O existed on the surface of the samples. The binding energies of Ag 3d, Al 2p, Mn 2p, Ni 2p and Zn 2p indicate that the doping ions existed as Ag+, Al3+, Mn2+, Ni2+ and Zn2+ in the TiO2 crystal lattice (Figs. 3(a) – 3(e)).
Fig. 3(f) shows that binding energies of 458.15 eV to 459.00 eV for TiO2 nanotubes correspond to the peaks of Ti 2p3/2, whereas binding energies of 464.05 eV to 464.90 eV correspond to Ti 2p1/2. The binding energy of Ti 2p indicates that Ti was incorporated into the lattice of TiO2 as Ti4+. The Ti 2p binding energy of the ion-doped TiO2 nanotubes increased compared with that of pure TiO2 because the electrons in the conduction band of TiO2 may be transferred to the doped metal ions on the surface of TiO2, which resulted in a decrease in the outer electron cloud density of Ti ions [8].
3.1.4. BET surface area analysis
The catalytic efficiencies of the TiO2 nanotubes are related to the SBET of the catalyst[16, 17]. As summarized in Table 1, the SBET of TiO2 nanotubes decreased with increasing calcination temperature. The decrease in SBET could be attributed to the aggregation of nanotubes, which resulted in a closely coagulated structure[17].
Table 1 indicates that the SBET of ion-doped TiO2 nanotubes was more or less smaller than that of un-doped TiO2 nanotubes, particularly for Ag+-, Mn2+-, and Ni2+-doped TiO2 nanotubes. The SBET decreased when metal ions were doped because of partial pore blockages and framework defects [9].
The SBET of TiO2 nanotubes was significantly larger than that of P-25 TiO2. This finding is attributed to the inner and outer surfaces of the layered-tubular structure, which is one of the special qualities of this catalyst [6].
3.1.5. UV-vis DRS analysis
The UV-vis DRS analysis was performed on the catalysis to obtain the energy band gaps (Eg) (Fig. 4). The Eg of the catalysts are calculated [18] and shown in Table 1.
Fig. 4(a) shows that Eg decreased with increasing calcination temperature. This trend occurred because the Eg of rutial TiO2 (3.0 eV) was narrower than that of anatase TiO2 (3.2 eV), and the rutile phase of TiO2 began to form when the calcination temperature was increased. The Eg of un-doped TiO2 nanotubes was between 3.30 and 3.22 eV, which was a little higher than that of anatase TiO2 in powder form. This blue shift was due to the quantum size effect on different morphologies of the catalysts [19]. The reflectance spectra of ion-doped TiO2 slightly shifted toward a longer wavelength (red shift) compared with those of un-doped TiO2, and the Eg of ion-doped TiO2 was visibly narrower. The results show that the doped elements involved were indeed integrated into the lattice of the TiO2 nanotubes, thereby altering the crystal and electronic structures of the TiO2 nanotubes [20]. The reduction in Eg of the TiO2 nanotubes allows the excitation of the catalyst at a lower irradiation power. Thus, the photocatalytic activity of the catalyst is enhanced [19].
3.2. Photocatalytic Activity of the Catalysts
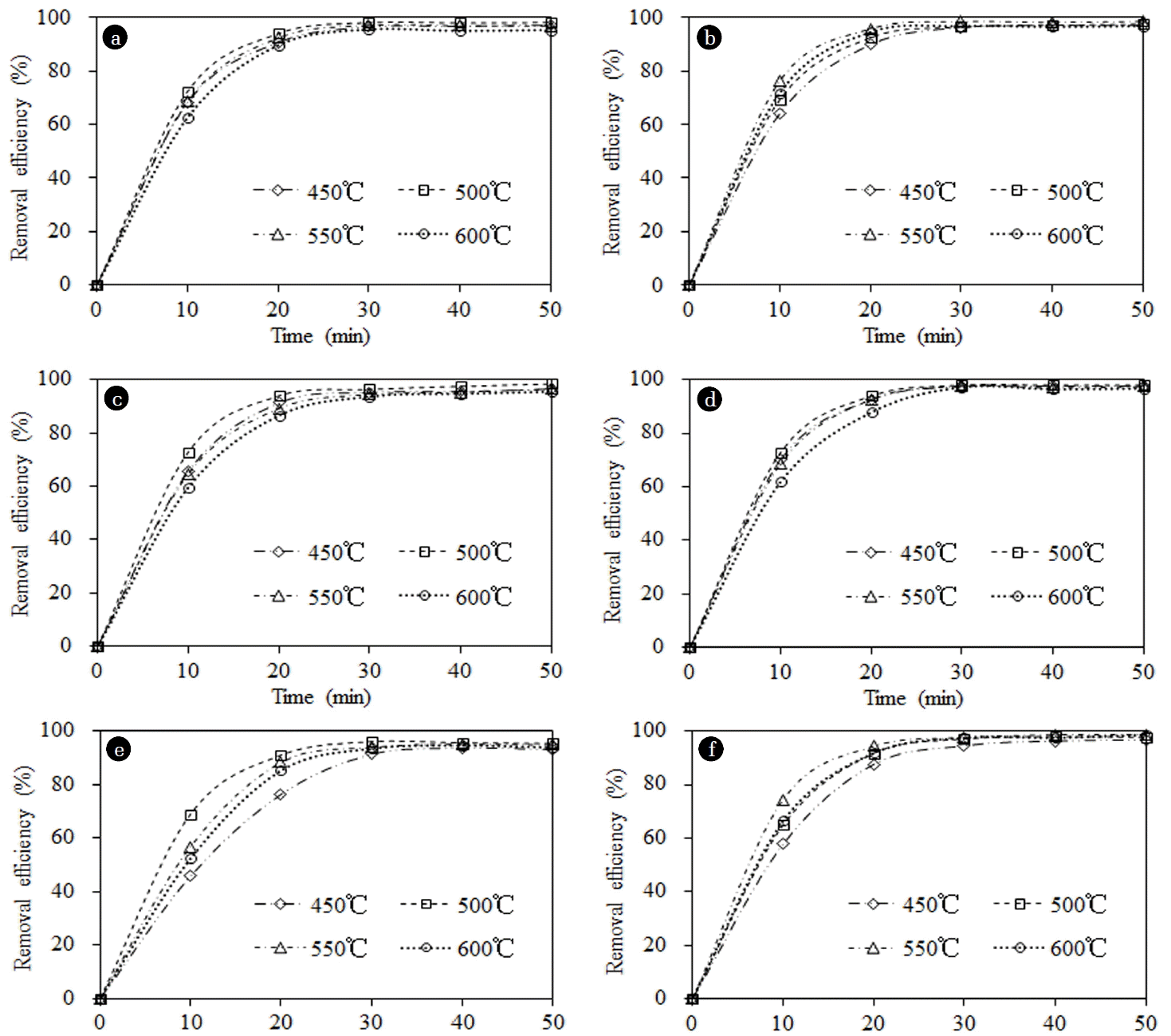
As shown in Fig. 5(a), the highest catalytic activity for the un-doped TiO2 nanotubes was observed at a calcination temperature of 500°C, with an RB removal efficiency of 98.1%. The SBET, crystalline phases, crystallinity, and Eg of TiO2 nanotubes were considered as crucial factors that affect the photocatalytic activity of the catalyst. When the calcination temperature was increased, the SBET decreased (see Table 1). The SBET could affect the photocatalytic activities of the catalyst because a larger SBET indicates that more pollutants are adsorbed onto the surface of the catalyst, and more areas become available for electron-hole pair separation. The anatase phase exhibited a higher photocatalytic activity compared with other phases. Although the SBET of TiO2 nanotubes calcined at 450°C were larger, the crystalline phases were predominantly amorphous, which resulted in low photocatalytic activity, and broadened Eg of TiO2. For TiO2 nanotubes calcined at 550°C, and 600°C, although the main crystalline phases were anatase, the amount of rutile phase increased, and the SBET decreased; thus, the overall efficiencies were low.
Photocatalytic activity was also affected by ionic radius, valence state, and configuration. The photocatalytic activities of the catalysts on RB removal increased when Ag+, Al3+ and Zn2+ were doped into the TiO2 nanotubes, whereas such activities decreased as a result of Mn2+ or Ni2+ doping. In the presence of Zn2+-doped TiO2 nanotubes calcined at 550°C, the removal efficiency of RB within 50 min was 98.7%. The catalytic activity was affected by the following factors. First, the existing impurity band can reduce the recombination of photoinduced electron-hole, which can transfer electrons more efficiently to the oxygen adsorbed on the surface of TiO2 nanotubes [21]. Second, the weight fractions of the anatase phases of the catalyst were larger than those of un-doped TiO2 nanotubes, the Eg of the nanotubes were narrower, and the SBET of the catalyst were smaller (see Table 1). Third, the ionic radii of the doped ions affected the photocatalytic activities.
For Ag+, the ionic radius was significantly larger than that of Ti4+ (60.5 pm). Ag+ was observed on the surface of TiO2. The electrons continuously transferred from TiO2 to Ag2O, which allowed the pollutant with negative charges to be easily adsorbed by the TiO2 crystals. The weight fraction of the anatase phase of the catalyst was larger than that of the un-doped TiO2 nanotubes, and the Eg of the catalyst was narrower. Therefore, the catalytic activity of Ag+-doped TiO2 increased.
The photocatalytic activity increased when Al3+ was doped into the catalyst. The ionic radii of Al3+ (53.5 pm) was smaller than that of Ti4+; thus, the ions were easily substituted in the lattice. Two doped ions replaced two Ti4+ ions, and one O2- hole appeared. The shape of the crystal changed, which facilitated the generation of electron-hole pairs. The weight fraction of the anatase phase of the catalyst increased after ion-doping, and the Eg was narrower. Therefore, the photocatalytic activities of Al3+-doped TiO2 nanotubes increased.
When Mn2+ and Ni2+ ions were added, the photocatalytic activity of the catalyst decreased. Mn2+ and Ni2+ ions, whose ionic radii were 67 and 69 pm, respectively, had a slight difficulty in entering the lattice, therefore, MnO and NiO were generated on TiO2 nanutubes. The formation of MnO or NiO could hinder the separation of electrons and holes. In addition, the SBET of Mn2+-and Ni2+-doped TiO2 were extremely small. Therefore, the photocatalytic activities of the Mn2+-and Ni2+-doped catalysts were limited.
The ionic radii of Zn2+, which was 74 pm, was slightly larger than that of Ti4+. ZnO might be formed on the surface of the TiO2. The combination of TiO2 with the doped metallic oxide results in the generation of Ti3+. The electrons were transferred to Ti3+ and then to the oxygen, which could reduce the probability for recombination between electrons and holes. In addition, the photogenerated holes left in the valence band would have more opportunity to participate in the oxidizing reactions because photogenerated electrons can be effectively scavenged by Zn2+ ions [22]. Therefore, the removal efficiencies increased.
4. Conclusions
Ion-doped TiO2 nanotubes were successfully synthesized for RB removal. The ions were effectively incorporated into the TiO2 lattice, except for Ag+. When the ions were doped and the calcination temperature was increased, the SBET of TiO2 decreased, and the Eg of ion-doped TiO2 became visibly narrower. The photocatalytic activities of catalysts on RB removal could be increased when Ag+, Al3+ and Zn2+ were doped into the TiO2 nanotubes, and the Zn2+-doped TiO2 nanotubes had the highest catalytic activity because of the effects of the weight fractions of the anatase phase, SBET, Eg, and the doped ions. When Mn2+ and Ni2+ were used as dopants, the photocatalytic activity of the catalyst decreased.