AbstractSulfate radical-based advanced oxidation processes have been applied in the remediation of polycyclic aromatic hydrocarbons (PAHs) contaminated soil, during which sulfate can be activated in different ways. In this work, effects of four different ways (FeSO4, NaOH, H2O2, and Heat) activated sodium persulfate (PS) on PAHs removal and soil physicochemical properties (pH, organic matter, functional groups, surface morphology, and partial heavy metal elements) were compared, and the influencing factors and mechanism of soil PAHs removal by thermally activated PS were also studied. The results showed that at the dosage of 3% of persulfate, the removal efficiency of PAHs followed the sequences of Heat (91.4%) > FeSO4 (86.6%) > H2O2 (86.2%) > NaOH (72.9%). However, thermal activation decreased the soil pH and organic matter content more significantly than other treatments. The reaction tended to reach equilibrium at 6 h when the dosage of persulfate was 3% and the activation temperature was 60 °C, and the reaction obeyed pseudo-first-order kinetics. Through quenching experiments, it was found that the free radicals playing a dominant role in the oxidation process were sulfate radicals. Compared with pH, liquid to soil (L/S ratio) and the temperature had more significant impacts on the degradation efficiency of PAHs.
Graphical Abstract
1. IntroductionPolycyclic aromatic hydrocarbons (PAHs) are among the most wide-spread organic contaminants distributed in soils around industrial sites [1, 2] such as petrochemical, coking, and steel. Because they have low volatility and are not easily soluble in water, they will be strongly absorbed by organic matter or minerals once they enter the soil [3]. Meanwhile, due to their characteristics of hardly degradable [4], highly carcinogenic, and highly mutagenic [5], they will eventually endanger human health without treatment properly. Therefore, the rehabilitation of PAHs contaminated soil is an urgent necessity.
Chemical oxidation has been widely used for remediating PAHs contaminated soil due to its advantages, such as short remediation cycle, high pollutant removal efficiency and low cost [6]. Meanwhile, chemical oxidants such as Fenton reagent, permanganate, and PS have been proved to be able to degrade PAHs in soil [7–10]. Among them, PS is widely employed because of its lower cost, milder reaction, higher selectivity, longer environmental retention time, and wider pH range [11].
PS is relatively stable and reacts slowly at ambient temperature. When only PS is implemented, the treatment of high pollutant concentration soil may not reach the remediation target because of the limited amount of oxidant added [12], and the soil even requires secondary oxidation, which greatly increases the cost [13]. PS can be activated to produce sulfate radicals, which in turn contribute to the degradation of target pollutants [14]. Commonly used activation reagents or methods include ferrous iron, alkaline, H2O2 and heat. Some previous literature has investigated the effect of activated PS on PAHs removal. It was reported that Fe2+-activated persulfate oxidation can reduce soil pollution and avoid the excessive soil acidification in in-situ remediation [4]. In addition, thanks to the strong tolerance of the Fe2+-activated persulfate system, the reduction of pyrene degradation caused by the background electrolytes (Cl−, HCO3 − and humic acid) in soil was not remarkable. Ranc et al. found that persulfate effectiveness was largely due to its thermal activation [15]. The thermal activation of persulfate (65 °C for 6 h) led to the degradation of more than 90% of both PAHs after 7 days of treatment [16]. Johansson et al. [17] reported that the main disadvantage of thermally activated PS oxidation was the formation of sulfates and sulfuric acid, resulting in a sharp drop of pH. PS may also affect soil organic matter, functional groups, structure, and heavy metal elements. However, no study has compared the effects of the above four commonly used activated PS on soil physicochemical properties so far. There were a few in-depth studies on thermally activated PS to degrade organic pollutants. Chen et al. [18] studied the effects of various operating parameters on the degradation of PNP by thermally activated persulfate, and explored the reaction pathway mechanism. Peng et al. investigated various factors (initial persulfate or BDE209 content, temperature, pH and inorganic ion) for thermally activated persulfate oxidation of BDE209 [19]. Liu et al. evaluated the effect of soil composition on available oxidant demand and CVOC removal rate [20]. It is also necessary to analyze the key factors and the mechanism involved in the degradation of PAHs by thermally activated PS.
In this work, Phenanthrene and Benzo [a] pyrene with high detection rate and great harm in PAHs contaminated sites were selected as target pollutants. This study aimed to: (1) compare the effects of different activation methods on PAHs removal and soil physicochemical properties; (2) analyze reactive radicals and experimental kinetics to provide mechanistic insights; (3) investigate the influence of key factors on the degradation of PAHs by thermally activated PS to find better working parameters.
2. Materials and Methods2.1. Chemicals and Original SoilDichloromethane and n-hexane were of HPLC grade, Phenanthrene (PHE, 97%) and Benzo [a] pyrene (BaP, 96%) were obtained from Maclean’s Reagent Co., Ltd., Shanghai, China. Sodium persulfate (Na2S2O8, PS, 99%,) and other chemical reagents were analytically pure, water used in the experiment was ultra-pure water. The original (non-PAHs-polluted) soil was taken from a park in Nanjing, China. The soil was naturally air-dried indoors, after removing stones, plants and other impurities, it was ground through a 60-mesh sieve and then stored in a brown bottle at a low temperature in a dark place for further use. Physicochemical characteristics of the soil were measured and are listed in Table S1.
2.2. Preparation of Contaminated SoilAn appropriate amount of dichloromethane solution with phenanthrene and benzo [a] pyrene dissolved was added to every 200 g of the original soil, and the mixture was stirred with a glass rod to promote homogeneous distribution of PAHs. Then placed the spiked soil in a fume hood to eliminate the solvents. After the solvent volatilized, the soil was sealed with the sealing film and aged for 2 weeks. The concentrations of PHE and BaP in the contaminated soil were 162.63 mg kg−1 and 149.18 mg kg−1, respectively, which were measured by GC-MS. Unless otherwise specified, soil in the following text generally refers to the contaminated soil.
2.3. Experimental SetupIn the degradation experiments, in order to evaluate the influence of PS oxidation process on PAHs removal and soil properties, a total of 17 treatments were set up. The dosage of PS was set as 1%, 3%, 6% and 9% of the mass fraction of the soil. The activation methods of PS were set as citric acid chelated iron activation (FeSO4), alkaline activation (NaOH), hydrogen peroxide activation (H2O2) and thermal activation (Heat) (Table 1). The PS oxidation experiments were carried out in some 40 ml brown sample bottles, 10 g of contaminated soil and a certain amount of ultrapure water were added to each brown sample bottle, and then oxidants and activators were added. The ratio of liquid to soil was set at 1:1 (g: mL) unless stated otherwise, and only 10 mL ultrapure water was added to the control group (CK). The sample bottles were sealed and shaken to ensure uniform reaction conditions and then placed in a water bath oscillator of darkness and were shaken at 180 rpm (the temperature of thermal activation treatments was 40~90°C, and the rest treatments was 25°C). After 72 h, the sample bottles were taken out and the reactions were terminated by ice bath for 2 hours. In quenching studies to identify free radicals, the radical scavengers, tert-butyl alcohol (TBA) and ethanol (EtOH) were applied to determine predominant free radicals in the thermal activation system responsible for the degradation of PAHs. The ratios of scavengers to PS were set as 30:1 (mol: mol). In the pH experiment, 25 mL of ultrapure water was first added to the soil sample, and then the required pH was adjusted with 10% NaOH or HCl solution prior to adding the oxidant. In the reaction kinetics experiment, the reaction was quenched by adding sodium thiosulfate solution at predefined time period. All experiments were performed in triplicate. All the soil samples after the reactions were freeze-dried and stored at 4°C for testing. The freeze-dried soils were characterized and their physicochemical properties (pH, organic matter and heavy metal elements) were determined one week later.
2.4. Analytical Procedures of PAHsFreeze-dried sub-samples (2 g) were collected, weighed, and mixed with dichloromethane (10 mL). The mixture was conducted by ultrasound assisted extraction for 15 min and then centrifuged at 3800rpm for 10 minutes. The supernatant was purified by filtration membrane and transferred to the test tube. The purified extract was concentrated to nearly 0.5 mL using a nitrogen blowing instrument, and internal standard was added and the volume diluted to 1 mL with n-hexane. The concentrations of PAHs in soil samples were analysed by gas chromatography (GC) (Agilent 8890) with a mass spectrometer (MS) (Agilent 5977A) with HP-5MS column (30 m × 0.32 mm × 0.25 μm). The injection volume was 1 μL, and the sample was injected in split mode with a split ratio of 10:1. The setting of column temperature program was as follows: started at 80°C for 2 min, and increased to 180°C at 20 °C min−1, then incessantly increased to 290°C at 10 °C min−1 and hold for 5 min at 290°C.
2.5. Physicochemical Properties and CharacterizationsPhysicochemical properties of soil samples, such as pH, organic matter content and total heavy metal content, were determined by technical provisions for soil sample analysis and testing methods. Heavy metal speciation was identified by BCR continuous extraction method [21], including acid soluble/exchangeable, reducible and oxidizable and residual state. The extractants of the first three forms of heavy metal are 0.11 mol L−1 acetic acid solution, 0.1 mol L−1 hydroxylamine hydrochloride solution (pH 2.0) and 1.0 mol L−1 of ammonium acetate solution (pH 2.0). A laser particle analyser (Malvin, Mastersizer 2000, UK) was used to determine the distribution of soil particle size distribution; the surface morphology of soil was performed by field emission scanning electron microscopy (SEM, Sigma 300, Zeiss, Germany); the groups in the soil and the functional group structures of the characteristic components in PAHs were analyzed by Fourier transform infrared spectroscopy (FTIR, Nicolet 6700, Thermo Fisher, USA).
3. Results and Discussion3.1. Effect of PS Activated in Different Ways on PAHs DegradationThe removal rate of PAHs under the four treatments (FeSO4, NaOH, H2O2, and Heat) all increase noticeably when the dosage of oxidant enhanced from 1% to 3% (Fig. 1a), similar to the results of Gou et al. [11]. With further increase in the amount of oxidant, the removal rate of PAHs did not increase significantly, possibly attributed to SO4•−, which played a vital role in the removal of PAHs in the PS oxidation process, reacted with excess PS [22] (Eq. (1)). In the FeSO4 activation treatment groups, even when the amount of PS was increased from 3% to 6%, the degradation efficiency of PAHs decreased slightly, which may be ascribed to that the excess Fe2+ consumed SO4 •− [14] (Eq. (2)). At the dosage of 3%, the overall removal efficiency of PHE and BaP by the four treatments was as follows: thermal activation (91.4%)>citric acid chelated iron activation (86.6%)>hydrogen peroxide activation (86.2%)>alkali activation (72.9%). Thermal activated PS had the best degradation effect on PAHs in this paper. The overall degradation efficiency was the highest when 9% PS was applied, reaching 98.8%, the degradation efficiency of PHE was 97.5%, and BAP can be almost completely removed (Figs. 1a, 1b and 1c). Through the detection of reactive radicals, Zhao et al. [23] found that the high redox potential of thermally activated PS and the efficient removal of PAHs were due to the formation of hydroxyl radicals in the activation process.
The degradation of BaP in the soil treated with different activation methods was significantly better than that of PHE (Figs. 1b and 1c), indicating that within the condition of limited oxidant, SO4 •− tends to react preferentially with high-ring PAHs. Brown et al. [24] reported that the reactivity of PAHs is strongly correlated with their electronic structure (Clar structure). At the same time, high-ring BaP is likely to take longer to reach and combine with the soil organic matter, so its availability for oxidants will be higher than PHE [25]. In addition, BaP may also generate PHE during its degradation process when the PS dosage is limited.
3.2. Variations in Soil Properties3.2.1. Changes in soil pH and organic matterIt can be noted from Fig. 2a that the soil pH decreased after oxidation, which was because S2O8 2− would leave some SO4 2− and H+ in the soil during the process of degrading organic pollutants [26], as shown in Eqs. (3), and (4). Except for NaOH can alleviate the acidification issue, soil pH declined significantly with the other three activation methods and continued to decline with the increase of oxidant addition. In NaOH activation treatment, when the PS amount was over 3% the pH value increased. This may be because at alkaline pH (i.e., pH > 7), the reaction shown in Eq. (5) [27] occured rather than hydrolysis of persulfate anions (see Eqs. (3), and (4)). When the PS dosage exceeded 3%, SO4 • − reacted with excess PS (Eq. (1)), the generation of H+ was relatively reduced, and the presence of a high initial concentration of OH− (NaOH>0.9%) prevented the decrease of pH. With the addition of 3% PS, thermal activation treatment reduced pH from 7.86 to 3.01, a decrease of 61.70%, the decreasing degree of soil pH was as follows: thermal activation>citric acid chelateded iron activation>hydrogen peroxide activation>alkali activation. However, the pilot scale experiment of Wu et al. [28] was not like the small-scale experiment in which a small amount of soil was fully mixed. They observed that the buffering capacity of the soil resulted in less pH change; thus, the soil acidification phenomenon that occurred in the laboratory may not appear in the field projects.
As depicted in Fig. 2b, organic matter of the soil treated with different activation methods all decreased when PS dosage increased. When the dosage of PS was more than 1%, the decrease of organic matter slowed down. The possible reason was that the remaining organic matter was not easily degraded by PS and belonged to refractory organic matter. The soil organic matter reduced most obviously after thermal activation, when 3% PS was applied, it declined from 13.27 to 7.17 g kg−1, decreased by 45.97%. While the other three activation methods had less damage to the soil organic matter, which may be due to the strongest oxidation ability of thermal activated PS in this study. Liu et al. [20] also found that the reaction between soil organic matter and PS was speeded up at a higher temperature (50°C), thereby augmenting the relative contribution of soil organic matter to PS consumption.
Among the four activation methods, NaOH activated PS had the least effect on soil pH, and H2O2 activated PS had the least damage to organic matter. However, the soil pH and organic matter reduced most obviously after thermal activation.
3.2.2. Soil functional groups analysisNo significant variations were found in the functional groups of the soil treated by different activation methods at the dose of 3% PS (Fig. 3). The peak at 1650cm−1 is attributed to the C=C stretching vibration in the aromatic rings which are conjugated with another C=C bond, an aromatic nucleus, or a C=O bond [29]. This peak was weakened after oxidation, suggesting that activated PS might have the potential to remove aromatics from soil. After oxidation, the band at around 1320 cm−1 was enhanced, and this band is attributed to C-H bending vibration or C-O stretching vibration [30], which indicates that other hydrocarbons or oxygenated compounds may be produced in the soil. GC-MS scanning mode was used to identify PAHs products, and it was found that there were indeed hydrocarbons and oxygenated compounds generated. The total ion chromatograms of the two PAHs and their degradation products are detailed in Fig. S1. The recommended structures of the two PAHs degradation products based on total ion chromatogram and mass spectrometry are shown in Table S2. The peak intensity of the compound with retention time of 7.449 min was the highest, and the possibility of direct degradation of PHE and BaP into chain compounds with high carbon number was small. Therefore, the product should be mainly 3,4-dimethyl benzaldehyde (C9H10O), and its toxicity is much lower than that of PHE and BaP. In addition, thermally activated PS can significantly reduce the concentrations of PHE and BaP in soil. Therefore, this method can reduce the environmental risk of soil.
3.2.3. SEM analysis of soil morphologyIn order to identify the difference in surface morphology and micro-structure between the original soil samples and the soil samples treated with 3% PS addition, SEM characterization was performed. Since the changes under the four treatments were similar (Fig. S2), the thermally activated PS is taken as an example in this article. As depicted in Figure 4, the lamellar structure of the soil was clear before and after oxidation, but after oxidation the soil particles showed obvious agglomeration. Although the soil particle size changed, there was no obvious difference in the surface structure. Therefore, the possibility of plugging aquifer pores and reducing permeability coefficient after activated PS treatments is slight [31].
3.2.4. Analysis of heavy metals in soilSoil pH, organic matter, structure, morphology and other properties are affected by chemical oxidation process, these factors may further have influence on the occurrence form of heavy metal elements in the soil [32]. In this manuscript, the total amount of the six common heavy metal elements (Fe, Mn, Cu, Ni, Pb and Zn) in soil and the four forms of them before and after oxidation (3% sodium PS added amount) were measured, as shown in Table S1 and Fig. 5.
It is noteworthy that the chemical oxidation really had a certain impact on the heavy metals in these soil samples. The total amount of Fe and reducible Fe in the soil active with citric acid chelateded iron increased, which was mainly due to the introduction of exogenous Fe. And the increase of reducible Fe indicates that Fe oxide was formed after oxidation treatment, the other three treatments had less effect on the partitioning of Fe. However, due to the high background value of Fe in the soil, the impact of increasing Fe content in the FeSO4 activation system was almost negligible. Moreover, Fe in the residual fraction accounted for more than 96% in all treatments, this fraction stably exists in crystal lattices such as quartz and clay minerals, and has little effect on the migration and bioavailability of heavy metal. A decrease in pH may enhance the competition between H+ and dissolved metals for negatively charged surfaces (such as clay minerals), thereby reducing the adsorption of heavy metals in the soil and improving its mobility [33]. The reducible Mn decreased in different degrees after oxidation treatment (thermal activation declined the most), meanwhile, the increase of acid soluble Mn after thermal activation possibly be ascribed to the decline of pH leading to metal dissolution. Due to the high affinity of Cu for organic matter, and PS oxidizes soil organics [31], the organic bound (oxidizable) form of Cu declined and transformed into the acid-soluble form and reducible form. It can be noted that Ni in the organic bound fraction decreased after oxidation, and the acid soluble Ni increased significantly in the soil treated by thermal activation. There was no obvious change in the occurrence form of Pb in the soil before and after treatments. However, it seems that the chemical oxidation treatments could transform the Zn in the soil to a more stable residual fraction, further research may be needed to explain this phenomenon.
3.3. Degradation Behavior of Thermally Activated PS3.3.1. Radical quenching studiesIn order to identify the dominant free radicals (HO• or SO4 • −) in the thermal activation system, quenching experiments were carried out with free radical scavengers (EtOH and TBA) [34, 35]. The reaction rate of tert-butyl alcohol (TBA) with HO• is fast (reaction rate constant k= (3.8–7.6) × 108 M−1 s−1), while the reaction rate with SO4 •− is slow (k= (4–9.1) × 105 M−1 s−1). In contrast, EtOH reacts quickly with both HO• (k = (1.2–2.8) × 109 M−1 s−1) and SO4 •− (k= (1.6–7.7) × 107 M−1 s−1) [36]. Therefore, TBA is utilized to scavenge HO•, MeOH is used as a quencher for both HO• and SO4 •−. It can be seen from Fig. 6 that the PAHs removal after addition of 3.78 M TBA showed a tiny (2%) difference compared to the treatment with PS only within 48 h. When 3.78 M EtOH was added, the removal rate of PAHs declined from 85.6% to 52.3% at 48h. The quenching effects from TBA and EtOH indicate that the contribution of SO4 •− was more dominant than HO• in the thermal activation system. The result is different from that obtained by Zhao et al. [23] through ESR technique, they found the HO• was the predominant radical produced in the thermal activation of PS. It may be because they conducted ESR test in a liquid persulfate system, while in soil system SO4 •− is more selective than HO• and is less easily removed by some species (such as HCO3 −) as well as natural organic matter [37, 38].
3.3.2. Reaction kinetics of PHE and BaPIn the experimental study of reaction kinetics, three soils with different initial pollutant concentrations were prepared, of which the total concentrations of PAHs were 176.5 mg kg−1 (PHE: 82.1 mg kg−1, BaP: 94.4 mg kg−1), 321.7 mg kg−1 (PHE: 159.0 mg kg−1, BaP: 162.8 mg kg−1), and 600.5 (PHE: 293.6 mg kg−1, BaP: 306.9 mg kg−1), respectively. The degradation effects of PAHs were investigated at 60°C and fixed PS dosage (3%) for 0, 0.5, 1, 3, 6, 12, and 24 h. As can be seen from Fig. 7a, the degradation rate did not change significantly after the reaction lasted for 6 h. According to Chen et al. [36], the degradation curves of PAHs at different pollutant concentrations were fitted with the pseudo-first-order kinetic model (Fig. 7b):
Pseudo first order reaction kinetic equation:
where C0 and Ct are PHE and BaP concentrations at time zero and t, respectively; k, the reaction rate constant (h−1). Due to the limitation of oxidant addition, the overall removal efficiency of PAHs decreased with the increase of pollutant concentration. The reaction rate of PAHs decreased may be because the demand for reactive free radicals increased at a higher initial concentration of PAHs. Higher concentrations of PAHs also result in the accumulation of intermediates, thus reduced the ratio of free radicals ‘to PAHs molecules [39]. Furthermore, thermally activated PS at 60°C even cannot make the contaminated soil with PHE and BaP concentrations of 82.1 mg kg−1 and 94.4 mg kg−1 reach our remediation target (PHE<5 mg/kg and BaP<0.55 mg/kg), suggesting that this method under this working condition can only be used to remediate soils with lower pollutant concentrations.
3.3.3. Impacts of key factors on thermal activationIn this study, thermal activation has the highest removal efficiency on PAHs, thus the effects of the three key factors of liquid to soil (L/S) ratio, pH and temperature on it was further investigated. The vital influence of the L/S ratio on the degradation of PAHs in soil during chemical oxidation was shown in Fig. S3a. This part of the research was carried out at different L/S ratios (1:2, 1:1, 2:1, 3:1) with a fixed dose of oxidizer (3%). When the L/S ratio rose from 1:2 to 1:1, the PAHs removal rate increased, but the removal rate of 2:1 and 3:1 was lower than 1:1. The concentration of oxidant was affected by the L/S ratio, which in turn had an impact on the oxidizer consumed by the oxidizable substances in the soil [31]. At the L/S ratio of 1:2, the lowest degradation efficiency of PAHs was observed. This is caused by the incomplete mixing of liquid and soil, resulting in the ineffective contact between a part of the oxidant and pollutants. Appropriate increase of L/S ratio can make the pollutants in the soil more easily desorption, while too large L/S ratio dilutes the oxidant concentration, reduces the collision between free radicals and pollutants. In the research system of this paper, 1:1 was the most suitable L/S ratio.
In order to explore the ability of thermally activated PS to degrade PAHs in soil with different properties, the pH conditions (3, 5, 7, 7.86, 9, 11) of the soil were changed (Fig. S3b). It seems that thermally activated PS has the best overall degradation efficiency of PHE and BaP under the condition of close to neutral. However, the degradation efficiency of PHE and BAP was not significantly different under these pH conditions, which was not as significant as the L/S ratio. Although degradation efficiency of organic compounds varies at different pH values, advanced oxidation based on PS is still suitable for degradation of organic compounds with a wide pH range [38].
As depicted in Fig. S3c, the enhancing effect of low temperature( 40°C) on PAHs degradation was not obvious, with the rising of thermal activation temperature, the removal rate of PHE and BAP increased as well. When the temperature reached 90°C, the overall degradation efficiency reached the highest 99.1%. Due to high temperature (>50°C) energy input, PS O-O bond was broken and produce sulfate radicals [14]. Generally, the higher the temperature, the higher the generation rate of sulfate free radical SO4 •−. What’s more, the amount of oxidant used can be greatly reduced by properly increasing the reaction temperature [40]. Yang et al. [41] studied the degradation of Acid Orange 7 (AO7) by thermally activated PS at 25°C, 50°C, 60°C, 70°C and 80°C, and found that the decolorization rate of AO7 was close to 0% at 25°C within 180 min. With the increase of temperature, the degradation rate of AO7 by PS was higher, at 80°C, AO7 was almost 99% decolorized within 40 min. Meanwhile, at higher temperature, the pollutants in soil may also volatilize and desorb, which also enhances the pollutant removal. However, it is important to highlight those excessive temperatures cause free radicals to generate at too fast a rate, which favors radical-radical reactions over radical-organic reactions, and may lower degradation efficiency [42]. The results showed that compared with pH, L/S ratio and temperature had more significant impacts on the degradation efficiency of PAHs.
4. ConclusionsThere is an optimal amount of oxidant, further increasing the dosage of oxidant had no obvious effect on the degradation efficiency but increased the remediation cost and caused more serious damage to soil organic matter. The concentration of PHE and BaP in the soil could not be reduced to below the remediation target even in the heat activation treatment with 3% PS. Therefore, for sites contaminated with high concentrations of PAHs similar to this study, perhaps the thermal activation temperature can be further increased, or the heating method can be changed to achieve the remediation goal. Compared with the heat conduction heating method and the steam enhanced extraction method, the resistance heating technology has the advantages of relatively uniform heating, high utilization of heat energy, less damage to the stratum and soil, and relatively cheap price. The combination of the two technologies deserves further study. The chemical oxidation treatments had a certain impact on the physical and chemical properties of the soil. After the thermal activation treatment, the degree of pH reduction and the degree of destruction of organic matter are both the largest, which may be a disadvantage caused by its strong oxidation capacity. The effect of activated PS treatments on the heavy metal elements in the soil was generally mild, while the effect of thermal activation on Mn, Cu and Ni was slightly greater, making them transform into forms that are easier to migrate. This should be considered during the remediation process of soil contaminated by organic compounds and heavy metals.
AcknowledgmentsThis research was supported by National Natural Science Foundation of China (No. 42077126).
NotesAuthor Contributions Y.H. (Postgraduate), X.Y. (Ph.D. student), L.Z. (Postgraduate) and B.L. (Postgraduate) completed the experiments; Y.H. (Postgraduate), X.Y. (Ph.D. student) and Y.Z. (Professor) wrote the manuscript; K.S. (Professor) and W.J. (Professor) provided technical guidance and experimental funding. References1. Kuppusamy S, Thavamani P, Venkateswarlu K, Lee YB, Naidu R, Megharaj M. Remediation approaches for polycyclic aromatic hydrocarbons (PAHs) contaminated soils: Technological constraints, emerging trends and future directions. Chemosphere. 2017;168:944–968. doi:10.1016/j.chemosphere.2016.10.115
2. Usman M, Hanna K, Haderlein S. Fenton oxidation to remediate PAHs in contaminated soils: A critical review of major limitations and counter-strategies. Sci. Total. Environ. 2016;569:179–190. doi:10.1016/j.scitotenv.2016.06.135
3. Zhang P, Chen Y. Polycyclic aromatic hydrocarbons contamination in surface soil of China: A review. Sci. Total. Environ. 2017;605:1011–1020. doi:10.1016/j.scitotenv.2017.06.247
4. Guo J, Gao Q, Yang S, et al. Degradation of pyrene in contaminated water and soil by Fe2+-activated persulfate oxidation: Performance, kinetics, and background electrolytes (Cl−, HCO3 − and humic acid) effects. Process. Saf. Environ. 2021;146:686–693. doi:10.1016/j.psep.2020.12.003
5. Li GL, Lang YH, Gao MS, Yang W, Peng P, Wang XM. Carcinogenic and mutagenic potencies for different PAHs sources in coastal sediments of Shandong Peninsula. Mar. Pollut. Bull. 2014;84(1–2)418–423. doi:10.1016/j.marpolbul.2014.04.039
6. Li Y, Liao X, Huling SG, et al. The combined effects of surfactant solubilization and chemical oxidation on the removal of polycyclic aromatic hydrocarbon from soil. Sci. Total. Environ. 2019;647:1106–1112. doi:10.1016/j.scitotenv.2018.07.420
7. Boulange M, Lorgeoux C, Biache C, Saada A, Faure P. Fenton-like and potassium permanganate oxidations of PAH-contaminated soils: Impact of oxidant doses on PAH and polar PAC (polycyclic aromatic compound) behavior. Chemosphere. 2019;224:437–444. doi:10.1016/j.chemosphere.2019.02.108
8. Pardo F, Peluffo M, Santos A, Romero A. Optimization of the application of the Fenton chemistry for the remediation of a contaminated soil with polycyclic aromatic hydrocarbons. J. Chem. Technol. Biot. 2016;91:1763–1772. doi:10.1002/jctb.4767
9. Sun Y, Niu WK, Hu XJ, et al. Oxidative degradation of polycyclic aromatic hydrocarbons in contaminated industrial soil using chlorine dioxide. Chem. Eng. J. 2020;394:124857–124869. doi:10.1016/j.cej.2020.124857
10. Tian H, Chen C, Zhu T, Zhu B, Sun Y. Characterization and degradation mechanism of bimetallic iron-based/AC activated persulfate for PAHs-contaminated soil remediation. Chemosphere. 2021;267:128875–128909. doi:10.1016/j.chemosphere.2020.128875
11. Gou YL, Zhao QY, Yang SC, et al. Removal of polycyclic aromatic hydrocarbons (PAHs) and the response of indigenous bacteria in highly contaminated aged soil after persulfate oxidation. Ecotox. Environ. Safe. 2020;190:110092–110100. doi:10.1016/j.ecoenv.2019.110092
12. Usman M, Faure P, Ruby C, Hanna K. Application of magnetite-activated persulfate oxidation for the degradation of PAHs in contaminated soils. Chemosphere. 2012;87:234–240. doi:10.1016/j.chemosphere.2012.01.001
13. Gou Y, Yang S, Cheng Y, et al. Enhanced anoxic biodegradation of polycyclic aromatic hydrocarbons (PAHs) in aged soil pre-treated by hydrogen peroxide. Chem. Eng. J. 2019;356:524–533. doi:10.1016/j.cej.2018.09.059
14. Zhou Z, Liu X, Sun K, et al. Persulfate-based advanced oxidation processes (AOPs) for organic-contaminated soil remediation: A review. Chem. Eng. J. 2019;372:836–851. doi:10.1016/j.cej.2019.04.213
15. Ranc B, Faure P, Croze V, Lorgeoux C, Simonnot MO. Comparison of the effectiveness of soil heating prior or during in situ chemical oxidation (ISCO) of aged PAH-contaminated soils. Environ. Sci. Pollut. R. 2017;24:11265–11278. doi:10.1007/s11356-017-8731-0
16. Peluffo M, Rosso JA, Morelli IS, Mora VC. Strategies for oxidation of PAHs in aged contaminated soil by batch reactors. Ecotox. Environ. Safe. 2018;151:76–82. doi:10.1016/j.ecoenv.2017.12.067
17. Johansson C, Bataillard P, Biache C, et al. Ferrate(VI) oxidation of polycyclic aromatic compounds (PAHs and polar PACs) on DNAPL-spiked sand: degradation efficiency and oxygenated by-product formation compared to conventional oxidants. Environ. Sci. Pollut. R. 2020;27:704–716. doi:10.1007/s11356-019-06841-0
18. Chen X, Murugananthan M, Zhang Y. Degradation of p-Nitrophenol by thermally activated persulfate in soil system. Chem. Eng. J. 2016;283:1357–1365. doi:https://sci-hub.et-fine.com/10.1016/j.cej.2015.08.107
19. Peng HJ, Zhang W, Xu LY, Fu RB, Lin KF. Oxidation and mechanism of decabromodiphenyl ether (BDE209) by thermally activated persulfate (TAP) in a soil system. Chem. Eng. J. 2016;306:226–232. doi:https://sci-hub.et-fine.com/10.1016/j.cej.2016.07.066
20. Liu J, Liu Z, Zhang F, Su X, Lyu C. Thermally activated persulfate oxidation of NAPL chlorinated organic compounds: effect of soil composition on oxidant demand in different soil-persulfate systems. Water. Sci. Technol. 2017;75:1794–1803. doi:10.2166/wst.2017.052
21. Davidson CM, Duncan AL, Littlejohn D, Ure AM, Garden LM. A critical evaluation of the three-stage BCR sequential extraction procedure to assess the potential mobility and toxicity of heavy metals in industrially-contaminated land. Anal. Chim. Acta. 1998;363:45–55. doi:10.1016/s0003-2670(98)00057-9
22. Yu XY, Bao ZC, Barker JR. Free Radical Reactions Involving Cl•, and Cl2 −•, and SO4 −• in the 248 nm Photolysis of Aqueous Solutions Containing S2O8 2− and Cl−
. J. Phys. Chem. A. 2004;108:295–308. doi:10.1002/chin.200414021
23. Zhao D, Liao XY, Yan XL, Huling SG, Chai TY, Tao H. Effect and mechanism of persulfate activated by different methods for PAHs removal in soil. J. Hazard. Mater. 2013;254:228–235. doi:10.1016/j.jhazmat.2013.03.056
24. Brown GS, Barton LL, Thomson BM. Permanganate oxidation of sorbed polycyclic aromatic hydrocarbons. Waste. Manage. 2003;23:737–740. doi:10.1016/s0956-053x(02)00119-8
25. Peluffo M, Pardo F, Santos A, Romero A. Use of different kinds of persulfate activation with iron for the remediation of a PAH-contaminated soil. Sci. Total. Environ. 2016;563:649–656. doi:10.1016/j.scitotenv.2015.09.034
26. Liang C, Lee IL, Hsu IY, Liang CP, Lin YL. Persulfate oxidation of trichloroethylene with and without iron activation in porous media. Chemosphere. 2008;70:426–435. doi:10.1016/j.chemosphere.2007.06.077
27. Marchesi M, Thomson NR, Aravena R, Sra KS, Otero N, Soler A. Carbon isotope fractionation of 1,1,1-trichloroethane during base-catalyzed persulfate treatment. J. Hazard. Mater. 2013;260:61–66. doi:10.1016/j.jhazmat.2013.05.011
28. Wu H, Sun L, Wang H, Wang X. In situ sodium persulfate/calcium peroxide oxidation in remediation of TPH-contaminated soil in 3D-sand box. Environ. Technol. 2018;39:91–101. doi:10.1080/09593330.2017.1296029
29. Yuan X, Choi SW, Jang E, Lee KB. Chemically activated microporous carbons derived from petroleum coke: Performance evaluation for CF4 adsorption. Chem. Eng. J. 2018;336:297–305. doi:10.1016/j.cej.2017.11.168
30. Guo Y, Rockstraw DA. Physical and chemical properties of carbons synthesized from xylan, cellulose, and Kraft lignin by H3PO4 activation. Carbon. 2006;44:1464–1475. doi:10.1016/j.carbon.2005.12.002
31. Liang C, Chien YC, Lin YL. Impacts of ISCO Persulfate, Peroxide and Permanganate Oxidants on Soils: Soil Oxidant Demand and Soil Properties. Soil. Sediment. Contam. 2012;21:701–719. doi:10.1080/15320383.2012.691129
32. Huang F, Wei X, Zhu T, Luo Z, Cao J. Insights into Distribution of Soil Available Heavy Metals in Karst Area and Its Influencing Factors in Guilin, Southwest China. Forests. 2021;12:609–620. doi:10.3390/f12050609
33. Sungur A, Soylak M, Ozcan H. Investigation of heavy metal mobility and availability by the BCR sequential extraction procedure: relationship between soil properties and heavy metals availability. Chem. Spec. Bioavailab. 2014;26:219–230. doi:10.3184/095422914x14147781158674
34. Zhang W, Su Y, Zhang X, Yang Y, Guo X. Facile synthesis of porous NiCo2O4 nanoflakes as magnetic recoverable catalysts towards the efficient degradation of RhB. Rsc. Adv. 2016;6:64626–64633. doi:10.1039/c6ra12706a
35. Zhu J, Zhu Z, Zhang H, Lu H, Qiu Y. Efficient degradation of organic pollutants by peroxymonosulfate activated with MgCuFe-layered double hydroxide. Rsc. Adv. 2019;9:2284–2291. doi:10.1039/c8ra09841g
36. Chen L, Hu X, Yang Y, et al. Degradation of atrazine and structurally related s-triazine herbicides in soils by ferrous-activated persulfate: Kinetics, mechanisms and soil-types effects. Chem. Eng. J. 2018;351:523–531. doi:10.1016/j.cej.2018.06.045
37. Ji Y, Dong C, Kong D, Lu J. New insights into atrazine degradation by cobalt catalyzed peroxymonosulfate oxidation: Kinetics, reaction products and transformation mechanisms. J. Hazard. Mater. 2015;285:491–500. doi:10.1016/j.jhazmat.2014.12.026
38. Ji Y, Dong C, Kong D, Lu J, Zhou Q. Heat-activated persulfate oxidation of atrazine: Implications for remediation of ground-water contaminated by herbicides. Chem. Eng. J. 2015;263:45–54. doi:10.1016/j.cej.2014.10.097
39. Zou X, Li X, Chen C, et al. Degradation performance of carbamazepine by ferrous-activated sodium hypochlorite: Mechanism and impacts on the soil system. Chem Eng J. 2020;389–399. doi:10.1016/j.cej.2019.123451
40. Peng LB, Deng DY, Guan MY, Fang XT, Zhu QY. Remediation HCHs POPs-contaminated soil by activated persulfate technologies: Feasibility, impact of activation methods and mechanistic implications. Sep. Purif. Technol. 2015;150:215–222. doi:10.1016/j.seppur.2015.07.002
Fig. 1(a) Removal effect of PAHs in the soil after different oxidation treatments (dosages of PS: 0, 1%, 3%, 6%, and 9%; activation methods: FeSO4, NaOH, H2O2, and Heat). Residual concentration of (b) PHE and (c) BaP after different oxidation treatments. 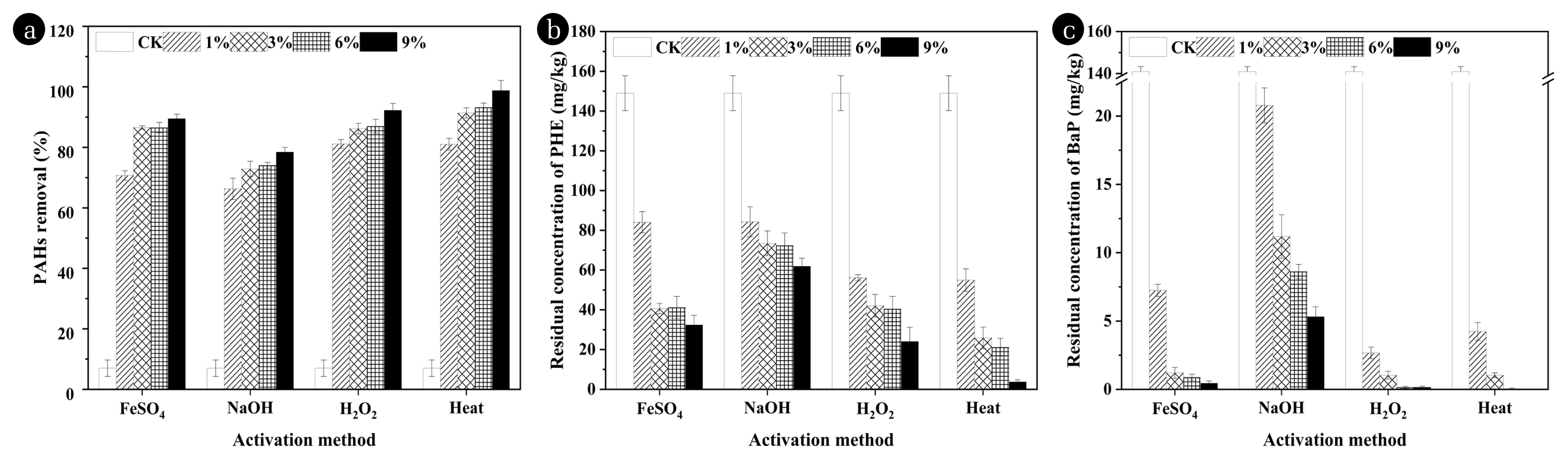
|
|
|||||||||||||||||||||||||||||||||||