AbstractIn this study, a series of acid-base bi-functional catalysts were prepared by mixing different amounts of basic amino acids (AAs) and phosphotungstic acid (PTA), which exhibited the excellent performance in catalyzing conversion of oleic acid (OA) for the biodiesel production. The physicochemical properties of the catalysts were characterized and analyzed using modern testing techniques and characterization methods such as XRD, FT-IR, XPS, SEM, TEM, and Hammett titration. The various influence parameters were optimized using the central composite design based the response surface methodology, where the maximum biodiesel yield of 97.0% was achieved at the MeOH/OA molar ratio of 5.9, the catalyst loading of 8%, reaction time of 6 h, and reaction temperature of 65°C. Furthermore, the stability and reusability of the prepared catalyst were also demonstrated. At last, the possible catalytic mechanism of the prepared catalyst was comprehensively described.
1. IntroductionThe increasing fossil fuel combustion, stimulating people to find a green and efficient alternative fuels [1]. Biodiesel is environment-friendly, biodegradable, non-toxic, similar to petro-diesel [2, 3], which is a preferred renewable energy resource. Selecting appropriate catalyst is especially important for the efficient biodiesel production. Homogeneous acids such as sulfuric acid and hydrochloric acid are the most widely used catalysts in acid reduction treatment. Among the homogeneous acids, phosphotungstic acid (PTA) is not only an environmentally friendly catalyst but also has strong Brønsted solid acidity and excellent redox properties [4]. However, this is limited because they are soluble in polar solvents and are challenging to recover [5]. Also, their specific surface area is small, which is not rendering better mass transport and diffusion in reactions [6]. Besides, the active sites are prone to be leached into the liquid reactants. Cardoso [7] used pure PTA to catalyze the fatty acid esters from triethylene glycol and methacrylic acid. Although the product purity was higher, the recycling process was cumbersome and the recycling performance was poor. Shanmugam [8] compared PTA, sulfuric acid, and trifluoroborate ether as catalysts to synthesize methacrylates. PTA had higher catalytic activity and did not require radical inhibitors such as toluene. However, it could only be recycled 3 times, and the conversion yield was greatly reduced. One way to improve the actively and stability of the catalyst is to introduce base groups into the PTA based catalyst. Amino acids (AAs) as the most plentiful natural source are considered suitable functional groups due to their various side chains and physiological importance [9]. Basic amino acids (arginine, lysine, and histidine) have stronger basicity, which could react with PTA molecules to form composite catalysts with two active centers of acid and base [10, 11]. However, there are few reports on the combination of basic amino acids and phosphotungstic acid to prepare catalysts for biodiesel production. The optimization of reaction conditions for the preparation of biodiesel is a complicated process involving a large number of experimental studies. Response surface methodology (RSM) overcomes the shortcomings of traditional single factor experiments such as large number of experiments, long experiment period and inaccurate results. It is suitable for this study of many reaction conditions in this study and determines the optimal reaction conditions.
In this study, we selected phosphotungstic acid (H3PW12O40) and basic amino acid to self-assemble series of acid-base bifunctional composite catalysts (AA/PTA). The strength of acids and bases could be adjusted by changing the concentrations and types of amino acids and then used the prepared catalyst for producing biodiesel from oleic acid (OA). The effects of process parameters such as reaction time, reaction temperature, catalyst loading, and methanol/oleic acid (MeOH/OA) molar ratio on the catalytic reaction were investigated through a series of experiments and model design experiments.
2. Experimental Section2.1. Preparation of AA/PTA CatalystsThe reagents used in this experiment were all analytical or chemically pure. A series of different acid-base bi-functional catalysts were prepared according to the ref. [12]. AA powder (1, 2, 3 mmol) and PTA (1 mmol) were separately added slowly into a 50 mL of 1 M hydrochloric acid solution at room temperature. The suspension was then stirred separately for 30 min. The prepared PTA solution was introduced into the AA solution. The mixture was then stirred at 30°C for 12 h. After that, the white precipitate was appeared immediately. The product was collected by centrifugation and washed with distilled water for 3 times. The amount of amino acids changed sequentially, and the resulting catalysts were expressed as ArgxPTA (x = 1 – 3), LysxPTA (x = 1 – 3), and HisxPTA (x = 1 – 3).
2.2. Catalyst CharacterizationThe morphology and structure of the prepared catalyst were investigated using SEM and TEM, respectively. The crystalline phase of the prepared catalyst was analyzed using X-ray diffraction (XRD). The elemental composition and chemical state of the material surface are detected by X-ray photoelectron spectrometer (XPS) and Fourier transform infrared spectroscopy (FTIR), respectively. The acid-base strength of the catalyst was measured by Hammett acid/base strength (H0), and the acid-base content was represented by acid-base titration.
2.3. Catalyst activity testA certain amount of OA, methanol, and catalyst was added into a 250 mL flask. This mixture was stirred at a specific temperature for a certain time. After completion, the reaction mixture was immediately cooled to room temperature, the excess methanol was distilled off under reduced pressure, and the catalyst was recovered by centrifugation. The reaction parameters i.e. the catalyst loading, MeOH/OA molar ratio, reaction time, and reaction temperature were regulated within a certain range to optimize the conversion yield of OA. Finally, the conversion yield of biodiesel was calculated based on formula:
Where AVBIO and AVOA are the acid value of OA and product, respectively.
3. Results and Discussion3.1. Catalyst ScreenThe catalytic performance of various catalysts in the catalytic process were investigated (Fig. 1). Remarkably, there was a significant difference for different types of catalysts. The conversion yield was 89.54% for pure H3PW12O40 catalyst. The maximum conversion yield (94.92%) was attained by employing Arg2PTA catalyst. The conversion performance of Lys2PTA (90.94%) was slightly lower than that of Arg2PTA. Meanwhile, the lowest conversion yield was using His1PTA (80.81%) as assisted medium in this study. There are four possible reasons that caused the difference in conversion yield: (1) Amino acid is a kind of basic material. The numbers of active sites increases with increasing amino acid concentrations, which significantly increasing the conversion yield. (2) The oleic acid containing high acid value was used as lipid feedstock in this study. The conversion process is more based on the esterification reaction. The pure phosphotungstic acid is a super strong Brønsted acid, which is more active in the esterification reaction. Compared to the content of the pure phosphotungstic acid, the content of phosphotungstic acid in the composite catalyst would be decreased based on our experiment steps when the molar ratio of AA/PTA was 1:1. The acid strength of the composite catalyst (1:1) was lower than that of the pure phosphotungstic acid from the Table 1. Whilst the base strength of the composite catalyst was also low. Hence, the pure phosphotungstic acid has a higher catalytic efficiency than the composite catalyst (1:1). (3) The acid and base strength of the composite catalyst would be significantly improved when the molar ratio of AA/PTA was 2:1. The synergistic effect between the AA and PTA was enhanced, resulting in greater conversion yield. Additionally, arginine with amino group and guanidine group exhibits more activity than other amino acids in esterification reactions. (4) Saponification was easy to occur due to the higher base strength of the composite catalyst for the OA with high acid value when the molar ratio of AA/PTA was 3:1, which was detrimental to the conversion of biodiesel.
3.2. Characterization3.2.1. SEM and TEMThe SEM was used to analyze the microscopic morphology of Arg2PTA (Fig. 2(a), (b)). Both pure phosphotungstic acid and Arg2PTA had a smooth surface and a flaky structure. However, the size after the introduction of arginine becomes less uniform, which may be due to the electrostatic effect between arginine and PTA.
In order to further determine the morphological characteristics of the catalyst, TEM was used for Arg2PTA (Fig. S1). It could be seen from the figure that the particle diameter of Arg2PTA is about 1.5 μm, and the result was consistent with SEM.
3.2.2. FT-IRThe FT-IR spectrums of the pure arginine, PTA, and Arg2PTA catalysts were compared in Fig. 3. The characteristic peak of the Keggin structure of PTA at 1,080 cm−1 (P-Oa), 987 cm−1 (W=O), 893 cm−1 (W-Ob-W), and 806 cm−1 (W-Oc-W) were well preserved in the Arg2PTA composite salt and only slightly moved compared to parent PTA [14, 15]. Peaks at 1,620–1,640 cm−1, 1,730–1,750 cm−1 and 1,440–1,629 cm−1 were correlate with the NH3+, −COOH, and the C-N stretching bands from amino acid molecules, respectively, which demonstrating the presence of amino acids in these hybrid ionic catalysts [9]. Additional vibration bands were observed at 3100–3400 cm−1 due to the N-H stretching originating from amino acids [16]. Some blue shifts of NH2 proved that the NH2 group of amino acids is protonated to NH3+. Therefore, these all indicated that the amino acid cation and the PTA anion are successfully combined through the electrostatic effect.
3.2.3. XRDNotwithstanding, the XRD spectrum of the Arg2PTA sample was slightly offset from the standard spectrum of the precursor acid, the diffraction peaks in the four ranges observed appeared at 2θ = 6–10°, 20–23°, 25–28°, and 33–36°, respectively, indicating that the AA/PTA remained the Keggin anion crystal structure of pure PTA [17]. As shown in Fig. 4, a peak detected at near 9.3° can correspond to the reflection of pure PTA at 10.7°, indicating that it has moved to higher lattice parameters after the organic-inorganic combination [18, 19]. All these results observed by XRD and FTIR indicated the successful introduction of amino acids into PTA.
3.2.4. XPSThe surface properties of the prepared Arg2PTA catalyst were examined by XPS spectroscopy. The binding energies of Arg2PTA at the C 1s (Fig. 5) level were 284.8 eV, 286.9 eV, and 289.4 eV, respectively, these values were usually ascribed to C-C bonding, C-O bonding, and C=O bonding [20]. The XPS survey spectrum of W 4f showed two different peaks at binding energies of 36.2 and 38.4 eV, representing W 4f7/2 and W 4f5/2 [21], proving that elements W exist as W6− [22]. The N 1s peak in the Arg2PTA catalyst confirmed the successful introduction of Arg. The high binding energy signal at 402.5 eV and the low binding energy signal at 397.4 eV corresponded to NH2 and protonated −NH3+, respectively [23]. The above analysis confirmed that the Arg2PTA composite catalyst is formed by electrostatic effect and demonstrated the existence of base active sites.
3.3. Influence of Catalytic Reaction ConditionsIn this paper, OA and methanol were used as raw materials, and Arg2PTA was used as the reaction catalyst to investigate the effects of catalyst loading, MeOH/OA molar ratio, reaction time, and reaction temperature on the esterification of OA into biodiesel.
3.3.1. Effect of catalyst loading
Fig. 6(a) showed the loading of the catalyst increased from 5% to 7%, the conversion yield gradually increased from 86.81% to 94.92%. Due to the increase in the loading of catalyst, the numbers of active catalytic sites has significantly increased, which can significantly improve the efficiency of esterification. However, a further increase in the loading of catalyst reduced the conversion yield to 79.62%. It is because the esterification reaction is a typical reversible reaction. Excessive addition of catalyst will cause a faster reaction balance, promoting the reverse reaction and ultimately leading to the conversion yield reduce [24].
3.3.2. Effect of Methanol/oleic acid molar ratioTheoretically, when the esterification reaction is completed, the molar ratio of MeOH to OA is 1:1, but the esterification reaction is an equilibrium reaction, requiring excess methanol to drive the reaction to produce higher methyl esters [25]. As shown in the Fig. 6(b), when the molar ratio was from 4 to 6, the conversion yield gradually increased. But when the MeOH/OA molar ratio increased to a certain extent, the relative concentration of catalyst and oleic acid in the reaction system decreased, even if the amount of methanol continued to increase, the conversion yield would also decrease [26]. That is, when the molar ratio was greater than 6, the conversion yield drops instead.
3.3.3. Effect of reaction timeWhen other conditions remain unchanged, prolonging the reaction time will help the chemical reaction proceed. Fig. 6(c) showed the effect of reaction time on the conversion yield of biodiesel. The conversion yield at the initial step increased with the extension of the reaction time, and the highest conversion yield of 94.92% was reached at 6 h. However, the reaction time further increased, the conversion yield decreased significantly, confirming that the esterification reaction is reversible and is more likely to occur over time. At the same time, the product of the esterification reaction may react with the water generated during the reaction, resulting in a decrease in the esterification yield [27].
3.3.4. Effect of reaction temperatureThe esterification reaction is endothermic. Increasing the reaction temperature can shift the reaction equilibrium to the right. The conversion yield firstly increased when the temperature increases from 55°C to 65°C (Fig. 6(d)). The reaction temperature continued to be increased, the conversion yield would decrease. However, the equilibrium constant of the reaction would be reduced more methanol were evaporated when the temperature was higher. Therefore, from the perspective of biodiesel conversion yield and reduction of energy consumption, the reaction temperature was selected as 65°C in this paper.
3.4. Optimization of Reaction conditions by Response Surface MethodologyUnder the condition of Arg2PTA as the catalyst, with the catalyst loading, MeOH/OA molar ratio, reaction time as independent variables, and biodiesel conversion yield as the response value, the response surface center combination test design were carried out. Among them, the reaction temperature had a minor influence on the methyl ester content. For the convenience and needs of research, the experiment determined the appropriate reaction temperature to be 65°C. The experimental factors and levels were shown in Table S1, and the test design and results were shown in Table S2. Each group of experiments was repeated 3 times, and the results were averaged.
Used design expert software to perform multiple regression analyses on the data in Table S2, and the results were shown in Table S3. The F-value of 5.41 and P-values less than 0.0500 indicate the model was significant. The P-value of the lack-of-fit term was 0.4195, which was not significant, indicating that the regression model was reliable. The influence of various factors on the response value of biodiesel conversion yield were fitted to obtain a quadratic polynomial regression equation:
Where Y is the conversion yield, A is catalyst loading (%), B is MeOH/OA molar ratio, and C is reaction time (h). The correlation coefficients R2 and Adj. R2 of the optimized model were 0.8743 and 0.7126, respectively, indicating that the two regression equation was relatively good and the optimized model had high feasibility. In addition, the lack of fit term of the model was 0.74, indicating that the design model was credible. The three-factor three-level interval design was reasonable, and the regression model obtained was reliable. The model could predict the influence of different factors on the conversion rate of OA. In this case C, B2 were significant model terms.
According to the regression equation, the optimal reaction conditions were obtained, the reaction time was 6 h, the MeOH/OA molar ratio was 5.9, and the catalyst loading was 8%. The maximum conversion yield under these conditions was 97.0%. Three verification experiments were carried out under these conditions, and the measured average conversion yield (96.54%) was consistent with the theoretical prediction value, indicating that the established model has good applicability.
3.5. Catalyst ReusabilityThe reusability of the catalyst is an essential indicator of the performance of the catalyst, because it reduces the cost of preparing biodiesel. In order to verify the stability and reusability of the prepared catalyst in the catalytic preparation of biodiesel, the reusability of the catalyst was investigated under the above optimized experimental conditions. After four consecutive cycles, the composite catalyst still showed an ideal conversion yield (76.44%). FT-IR was used to characterize and analyze the prepared catalyst after 4 cycles of recycling. The results showed that the four Keggin structural characteristic bands of PTA remain in the recovered catalyst.
3.6. Properties of Synthesised BiodieselThe characteristics of the biodiesel such as flash point, pour point, density, kinematic viscosity, acid value were evaluated and compared to reference [32, 33]. The results are listed in Table S4. According to the comparison, it revealed that the biodiesel in this study is similar to other biodiesel production.
3.7. Catalytic Mechanism of the Prepared CatalystIn the traditional concept, the Brønsted acidity of the catalyst is positively correlated with the activity of the esterification reaction. However, PTA is a beneficial solid acid with only moderate catalytic activity in this study, probably due to the low surface area (about 5 m2g−1), so the number of surface acid sites is minimal [28]. Therefore, the resistance of mass transfer may hinder pure PTA, and the three available proton acids H+ cannot be fully utilized in the esterification process. The Keggin unit of the PTA molecule can crystallize into a secondary structure together with water molecules [29]. The secondary structure is the most stable under environmental conditions. Each Kegging unit contains six water molecules, so the acidic protons of PTA is represented as H5O2+ and is located on the terminal O atom of the Keggin structure, while the polyanion PW12O303− is connected by H5O2+ [30, 31]. Fig. S2 shows the simplified mechanism of the reaction catalyzed by the acidic proton H5O2+. The side chain of basic amino acids also contains other basic functional groups. Taking lysine as an example, the structure of lysine contains basic amino groups. Both of them can be protonated in an acidic medium to generate double-positive ions, so the incorporation of AA as an alkaline cationto serve as a spacer to promote segregation of PW polyanion increases the acidic surface sites, improve catalytic activity in the esterification reaction [21].
4. ConclusionsThe novel acid-base bi-functional catalyst (Arg2PTA) has been successfully synthesized for the conversion of the OA into biodiesel. The effects of different process parameters on conversion yield were investigated by practical experiment and model design experiment. Through the mathematical regression model, the conversion yield of biodiesel could reach 97.0% under the optimal process conditions. In addition, the catalyst exhibited good recyclability after 4 cycles of reused. This stable heterogeneous catalyst provides a new and promising catalyst choice for preparing biodiesel from low-quality waste oil.
AcknowledgmentThis work was financially supported by the Open Project of Beijing Key Laboratory for Enze Biomass and Fine Chemicals, Hubei Key Laboratory of Novel Reactor and Green Chemical Technology (No. NRG202106), and the Open Project of Engineering Research Center of Phosphorus Resources Development and Utilization of Ministry of Education (No. LKF2021008).
NotesAuthor Contributions Q.Z. (Graduate Student) conducted all the experiments and wrote the manuscript. X.D. (Ph.D.) revised the manuscript. S.T. (Graduate Student) revised the manuscript. C.W. (Professor) provided resources. W.W. (Associate Professor) provided resources. W.F. (Associate Professor) wrote and revised the manuscript. T.W. (Professor) provided funding and resources. References1. Zhang D, Duan M, Yao X, Fu Y, Zu Y. Preparation of a novel cellulose-based immobilized heteropoly acid system and its application on the biodiesel production. Fuel. 2016;172:293–300.
2. Rajak U, Nashine P, Singh TS, Verma TN. Numerical investigation of performance, combustion and emission characteristics of various biofuels. Energ Convers Manage. 2018;156:235–252.
3. Christopher LP, Kumar H, Zambare VP. Enzymatic biodiesel: Challenges and opportunities. Appl Energ. 2014;119(15)497–520.
4. Morinaga H, Nakajima L, Nishioka Y, et al. Polymer synthesis by solid-acid catalyst based on heteropolyacid ammonium salt. Polym Bull. 2016;73(2)435–448.
5. Zhang Q, Wei F, Li Q, et al. Mesoporous Ag1(NH4)2PW12O40 heteropolyacids as effective catalysts for the esterification of oleic acid to biodiesel. RSC Adv. 2017;7(81)51090–51095.
6. Jeon Y, Chi WS, Hwang J, Kim DH, Shul YG. Core-shell nanostructured heteropoly acid-functionalized metal-organic frameworks: Bifunctional heterogeneous catalyst for efficient biodiesel production. ApplCatal B-Environ. 2018;242:51–59.
7. Cardoso AL, Augusti R, Silva M. Investigation on the esterification of fatty acids catalyzed by the H3PW12O40 heteropolyacid. J Am Oil Chem Soc. 2008;85(6)555–560.
8. Shanmugam S, Viswanathan B, Varadarajan TK. Esterification by solid acid catalysts-a comparison. J Mol Catal A-Chem. 2004;223(1–2)143–147.
9. Zhao Q, Wang H, Zheng H, et al. Acid-base bifunctional HPA nanocatalysts promoting heterogeneous transesterification and esterification reactions. Catal Sci Technol. 2013;3(9)2204–2209.
10. Imaz I, Marta RM, An J, et al. Metal-biomolecule frameworks (MBioFs). Chem Commun. 2011;47(26)7287–7302.
11. Kortz U, Savelieff MG, Ghali F, Khalil LM, Sinno DI. Heteropolymolybdates of AsIII, SbIII, BiIII, SeIV, and TeIV functionalized by amino acids. Angew Chem Int Edit. 2002;41(21)4070–4073.
12. Li H, Govind KS, Kotni R, et al. Direct catalytic transformation of carbohydrates into 5-ethoxymethylfurfural with acid-base bifunctional hybrid nanospheres. Energ Convers Manage. 2014;88:1245–1251.
13. Sereda TJ, Mant CT, Quinn AM, Hodges RS. Effect of the α-amino group on peptide retention behaviour in reversed-phase chromatography. Determination of the pKa values of the α-amino group of 19 different N-terminal amino acid residues. J Chromatogr A. 1993;646(1)17–30.
14. Qi Z, Huang Z, Wang H, et al. In situ bridging encapsulation of a carboxyl-functionalized phosphotungstic acid ionic liquid in UiO-66: A remarkable catalyst for oxidative desulfurization. Chem Eng Sci. 2020;225:115814–115818.
15. Zhang F, Jin Y, Shi J, et al. Polyoxometalates confined in the mesoporous cages of metal-organic framework MIL-100(Fe): Efficient heterogeneous catalysts for esterification and acetalization reactions. Chem Eng J. 2015;269:236–244.
16. Santos FM, Brandão P, Félix V, et al. Synthesis and structural characterization of Keggin polyoxometalate compounds with argininium(2+) cations. J Mol Struct. 2010;963(2–3)267–273.
17. Kim HJ, Jeon Y, Park J, Shul Y. Heterocycle-modified 12-tungstophosphoric acid as heterogeneous catalyst for epoxidation of propylene with hydrogen peroxide. J Mol Catal A-Chem. 2013;378:232–237.
18. Gong S, Lu J, Wang H, Liu L, Zhang Q. Biodiesel production via esterification of oleic acid catalyzed by picolinic acid modified 12-tungstophosphoric acid. Appl Energ. 2014;134:283–289.
19. Ito T, Inumaru K, Misono M. Structure of porous aggregates of the ammonium salt of dodecatungstophosphoric acid, (NH4)3PW12O40: Unidirectionally oriented self-assembly of nanocrystallites. J Phys Chem B. 1998;110(48)9958–9963.
20. Shchukarev AV, Korolkov DV. XPS study of group IA carbonates. Cent Eur J Chem. 2004;2(2)347–362.
21. Han X, Chen K, Yan W, et al. Amino acid-functionalized heteropolyacids as efficient and recyclable catalysts for esterification of palmitic acid to biodiesel. Fuel. 2016;165:115–122.
22. Akhtar MS, Cheralathan KK, Chun J, Yang OB. Composite electrolyte of heteropolyacid (HPA) and polyethylene oxide (PEO) for solid-state dye-sensitized solar cell. Electrochim Acta. 2008;53(22)6623–6628.
23. Zhang L, Jin Q, Huang J, et al. Modification of palygorskite surface by organofunctionalization for application in immobilization of H3PW12O40
. Appl Surf Sci. 2010;256(20)5911–5917.
24. Li R, Chen L, Yan Z. Synthesis of trimethylolpropane esters of oleic acid using a Multi-SO3H-Functionalized ionic liquid as an efficient catalyst. J Am Oil Chem Soc. 2012;89(4)705–711.
25. Li J, Wang X, Zhu W, Cao F. Zn1.2H0.6PW12O40 Nanotubes with double acid sites as heterogeneous catalysts for the production of biodiesel from waste cooking oil. Chem Sus Chem. 2009;2(2)177–183.
26. Zhang F, Fang Z, Wang YT. Biodiesel production directly from oils with high acid value by magnetic Na2SiO3@Fe3O4/C catalyst and ultrasound. Fuel. 2015;150(15)370–377.
27. Amani H, Ahmad Z, Hameed BH. Synthesis of fatty acid methyl esters via the methanolysis of palm oil over Ca3.5xZr0.5yAlxO3 mixed oxide catalyst. Renew Energ. 2014;66:680–685.
28. Okuhara T, Watanabe H, Nishimura T, Inumaru K, Misono M. Microstructure of cesium hydrogen salts of 12-Tungstophosphoric acid relevant to novel acid catalysis. Chem Mater. 2000;12(8)2230–2238.
29. Yang J, Janik MJ, Ma D, et al. Location, acid strength, and mobility of the acidic protons in keggin 12-H3PW12O40: A combined solid-state NMR spectroscopy and DFT quantum chemical calculation study. J Am Chem Soc. 2005;127(51)18274–18280.
30. Silva M, Xavier R, Santos L. H3PMo12O40 heteropolyacid: A versatile and efficient bifunctional catalyst for the oxidation and esterification reactions. Environ Ben Catal. 2013;7(18)225–244.
31. Kozhevnikov IV, Sinnema A, Jansen RJJ, Van BH.
17O NMR determination of proton sites in solid heteropolyacid H3PW12O40
. Catal Lett. 1994;4(3)92–93.
32. Ks A, He B, Bha C, Ne A. AC/CuFe2O4@CaO as a novel nanocatalyst to produce biodiesel from chicken fat. Renew Energ. 2020;147:25–34.
Fig. 1Conversion yield of biodiesel during the esterification of OA and methanol on various catalysts. (A = Arg1PTA, B = Arg2PTA, C = Lys1PTA, D = Lys2PTA, E = His1PTA, F = His2PTA, G = PTA, and H = AA3PTA). 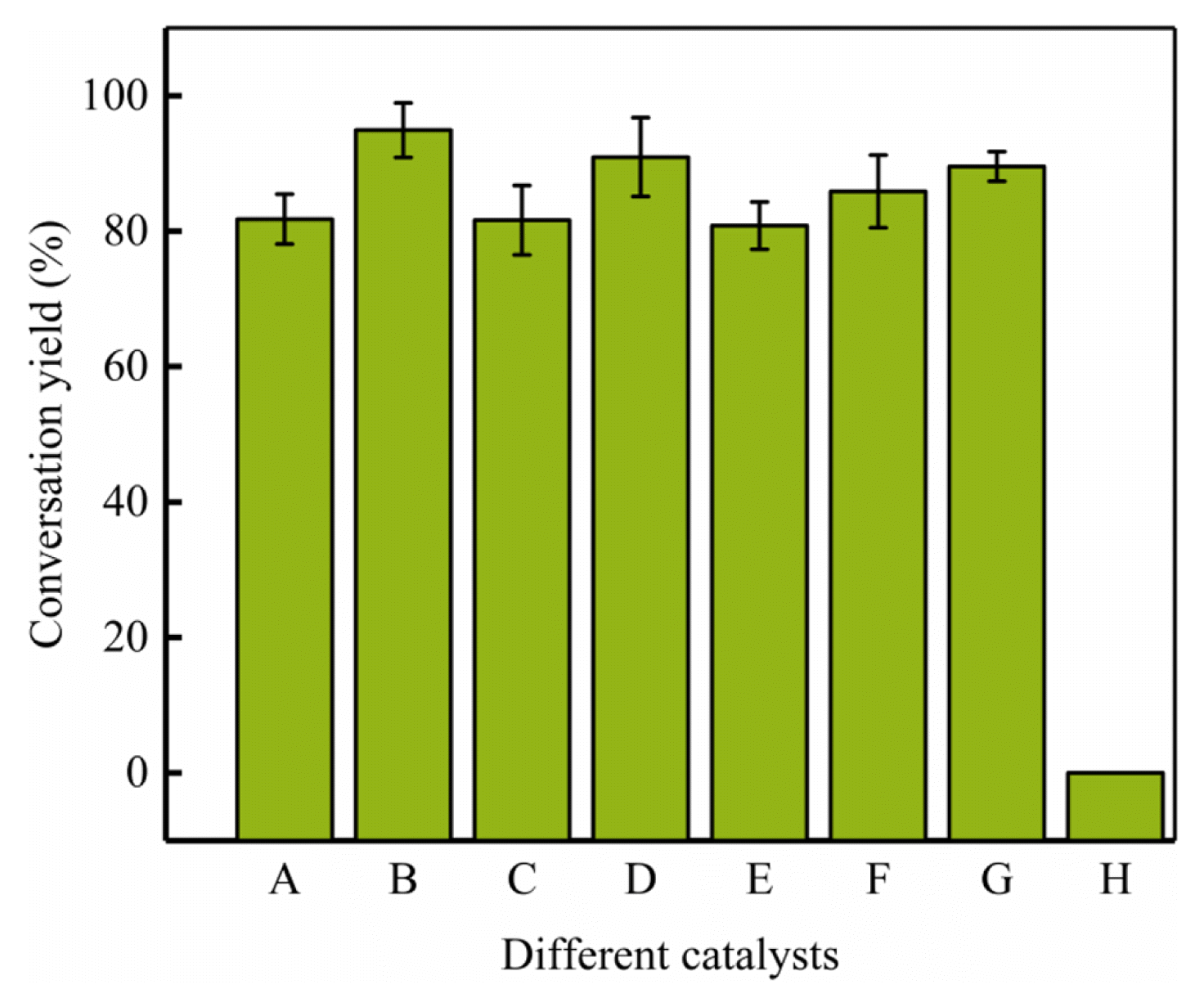
Fig. 6Effects of reaction conditions on the biodiesel conversion yield. (a) Catalyst loading, (b) MeOH/OA molar ratio, (c) Reaction time, and (d) Reaction temperature. Note that while varying each experimental variable, other parameters were kept constant at their predicted optimal values. 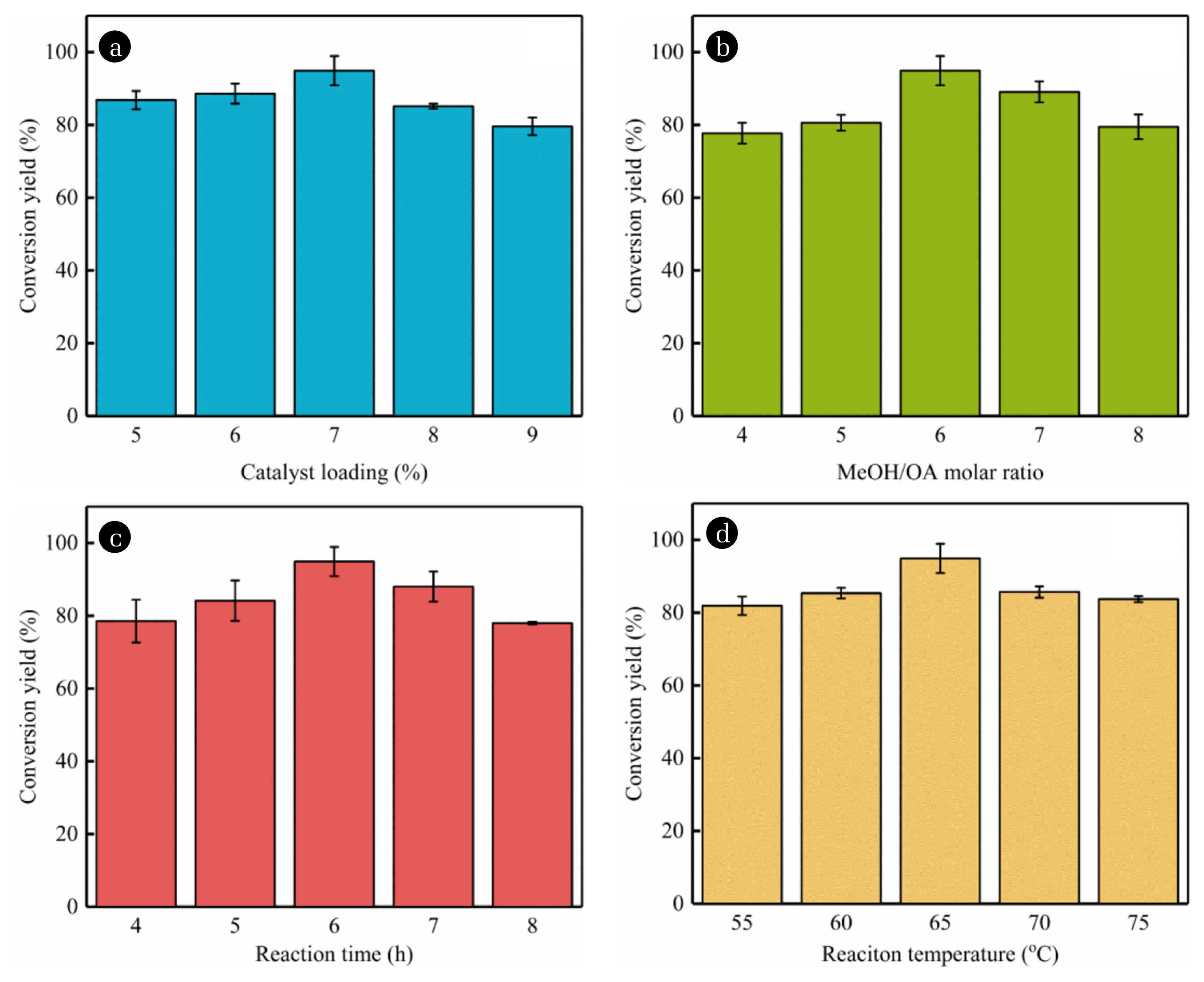
Fig. 7(a) Stability of the prepared catalyst during esterification of OA with MeOH, and (b) FTIR spectrums of fresh Arg2PTA and revive Arg2PTA catalysts. 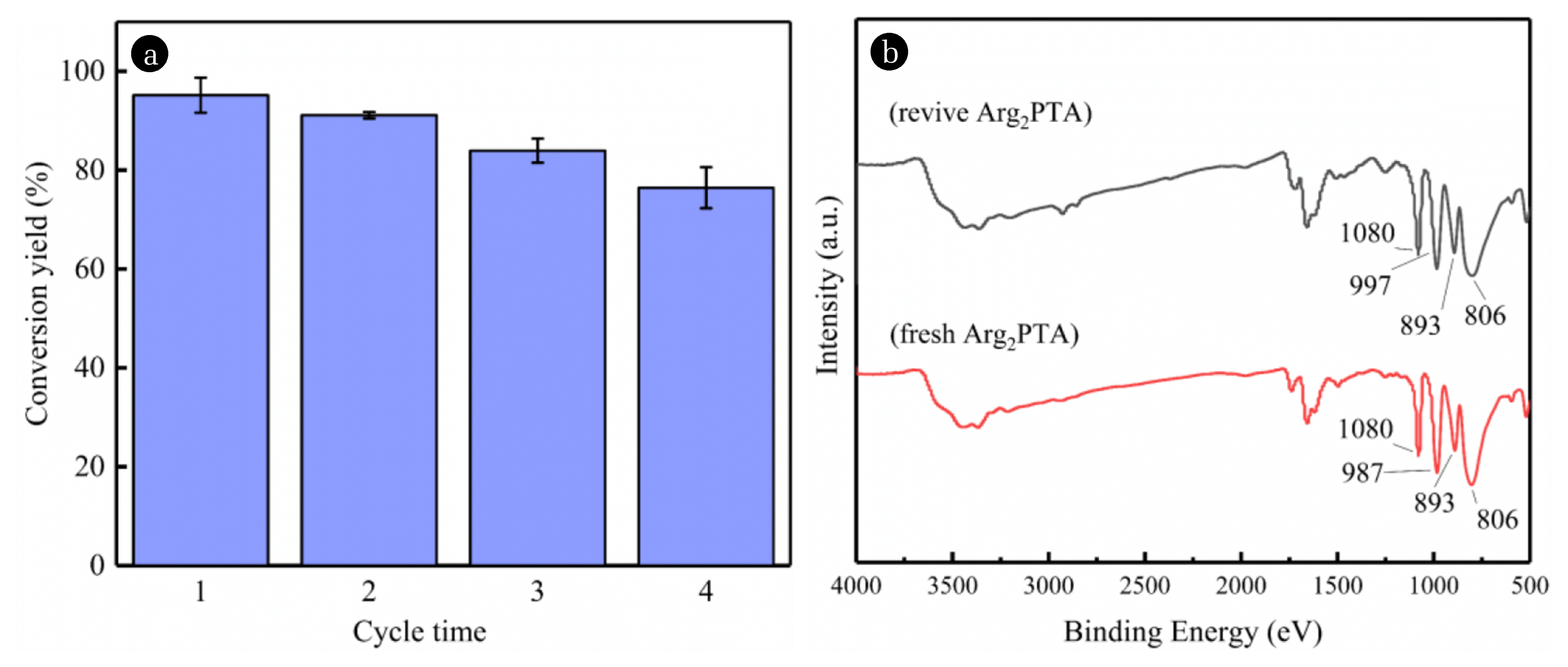
Table 1Acid and Basic Strength Simultaneously Detected for These Materials
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||