Inactivation of various bacteriophages by different ultraviolet technologies: Development of a reliable virus indicator system for water reuse
Article information
Abstract
There is an urgent need to identify more reliable indicator systems for human pathogenic viruses in water reuse practice. In this study, we determined the response of different bacteriophages representing various bacteriophage groups to different ultraviolet (UV) technologies in real wastewater in order to identify more reliable bacteriophage indicator systems for UV disinfection in wastewater. Bacteriophage ϕX174, PRD1, and MS2 in two different real wastewaters were irradiated with several doses of both low pressure (LP) and medium pressure (MP) UV irradiation through bench-scale UV collimated apparatus. The inactivation rate of ϕX174 by both LP and MP UV was rapid and reached ~4 log10 within a UV dose of 20 mJ/cm2. However, the inactivation rates of bacteriophage PRD1 and MS2 were much slower than the one for ϕX174 and only ~1 log10 inactivation was achieved by the same UV dose of 20 mJ/cm2. Overall, the results of this study suggest that bacteriophage MS2 could be a reliable indicator for human pathogenic viruses for both LP and MP UV disinfection in wastewater treatment processes and water reuse practice.
1. Introduction
There has been an accelerating increase in water reuse due to growing world population, rapid urbanization, and increasing scarcity of water resources [1]. Along with the growing interest in water reuse, however, there are some public health and environmental issues in water reuse practice. In fact, it is well recognized that water reuse practice is associated with many human health and ecological risks due to a large variety of chemical and biological contaminants. One of the most serious human health risks from water reuse is the potential transmission of infectious disease by pathogenic viruses. As a matter of fact, there are more than 100 pathogenic viruses potentially in wastewater with sometimes in very high numbers (~5 × 103 plaque forming units (PFU)/100 L) [2].
On the other hand, ultraviolet (UV) irradiation has recently gained considerable attention as an alternative to conventional chemical disinfectants in water and wastewater treatment processes due to its considerable ability to inactivate highly chlorine-resistant Cryptosporidium parvum and Giardia lamblia [3]. However, it should be mentioned that important human pathogenic viruses are a lot more resistant (sometimes more than 100 times more resistant) than human pathogenic bacteria and protozoan parasites to UV irradiation [4–6].
Currently, coliforms (either total coliforms or fecal coliforms or E. coli) are used as indicator organisms for human pathogenic viruses in wastewater practice. Although coliforms have been considered useful indicator systems for classical bacterial pathogens such as Shigella spp., Salmonella spp., and Vibrio cholerae, they have been shown to be inadequate for the indicators for waterborne viruses because waterborne viruses are more persistent in the environment and also more resistant to water and wastewater treatment processes [7]. Therefore, there is an urgent need to identify more reliable indicator systems for human pathogenic viruses in water reuse practice. In this study, we determined the response of different bacteriophages representing various bacteriophage groups to UV irradiation in both buffered system and real wastewater in order to identify more reliable bacteriophage indicator systems for UV disinfection in wastewater.
2. Materials and Methods
2.1. Preparation and Assay of Bacteriophages
Table 1 shows the characteristics of the bacteriophages used in this study. They are different in terms of their virion and genomic size, composition of genomic material and protein capsid, and presence or absence of outer envelopes. These bacteriophages were propagated and assayed in their appropriate hosts (Table 1) by the double agar layer plaque technique [8], as previously described [9]. Briefly, for each bacteriophage, the top agar layer having confluent lysis of host cells was harvested by scraping into a small volume of phosphate buffered saline (PBS), and bacteriophages were extracted with an equal volume of chloroform. The supernatant was recovered by low speed (4,000 Xg) centrifugation for 30 min at 4°C and stored at −80°C until use.
2.2. UV Disinfection Experiments
2.2.1. UV irradiation systems and radiometry
Our UV irradiation systems and radiometry were described in a previous study [10]. Briefly, our bench-scale, collimated beam UV apparatus consist of either two 15-Watt germicidal lamps or a 400-Watt medium pressure (MP) UV lamp for low pressure (LP) and MP UV systems, respectively. The lamps were mounted on top of the UV irradiation apparatus and provided incident radiation perpendicular to the surface of the test suspension in 60 × 15 mm cell culture petri dishes. UV irradiance was measured with a calibrated International Light IL1700 radiometer (International Light Inc., Newburyport, MA). The delivered UV dose, accounting for the UV absorbance in the liquid and the depth of the suspension, was calculated based on the measurement of the irradiance incident on the petri dishes, a series of correction factors (petri factor, reflection factor, water factor, divergence factor, sensor factor (MP UV only), and germicidal factor (MP UV only)) as described in Bolton and Linden [11], and the exposure time in seconds.
2.2.2. UV disinfection protocol
UV disinfection experiments were performed as previously described [12]. Briefly, each bacteriophage was diluted in either PBS or wastewaters (wastewater A and B: secondary effluent from Uiwang and Dangjin wastewater treatment plants, respectively) to give a final concentration of ~106 PFU/mL. Small aliquots (usually, 5 mL) each in small cell culture (petri) dishes was irradiated with the aforementioned collimated beam type UV sources while stirring the samples slowly on a magnetic stir plate. After predetermined exposure times, samples were removed from the UV irradiation systems and were diluted serially for subsequent microbiological assays.
2.3. Data Presentation and Statistical Analysis
The titers of bacteriophages were calculated as PFU/mL. For each experiment, the concentrations of the control sample were computed and taken as No, the initial microorganism concentrations. For each test sample, the average concentrations of each bacteriophage were computed as Nd. The proportion of initial bacteriophage remaining at each test sample was computed by dividing the bacteriophage concentration at each test sample (Nd) by the initial bacteriophage concentration (No). These values were then log10-transformed (log10 (Nd/No)), and the values of replicate experiments were averaged. These mean data for log10 (Nd/No) was then paired with the data for UV dose (mJ/cm2) and plotted.
Statistical analysis on the extent and kinetics of bacteriophage inactivation was performed by using EXCEL (Microsoft, Redmond, WA) and SAS (SAS Institute Inc., Cary, NC). Linear regression analysis was used to calculate the IT values (UV intensity × exposure time) to achieve 2–4 log10 inactivation with EXCEL. Also, Analysis of Covariance (ANCOVA) was applied to determine the difference in the inactivation kinetics (slope difference) by different disinfectants with SAS GLM procedure.
3. Results
3.1. Inactivation of Bacteriophages in PBS
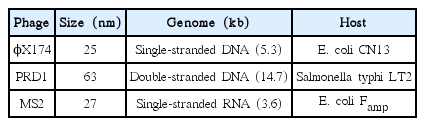
Fig. 1 shows the inactivation kinetics of bacteriophages by several different doses of monochromatic LP UV radiation in PBS (pH 7.4) at room temperature based on three independent experiments. The inactivation rate of ϕX174 by LP UV was rapid, approximately first-order, and reached~3.5 log10 within a UV dose of 20 mJ/cm2. However, the inactivation rates of bacteriophage PRD1 and MS2 by LP UV were much slower than the one for ϕX174 and only ~1.4 and ~1 log10 inactivation, respectively, was achieved by the same UV dose of 20 mJ/cm2.
Inactivation of bacteriophages by LP UV in PBS (pH 7.4) at room temperature (Each point is an average of three independent experiments and error bars are equal to one standard deviation).
Fig. 2 shows the inactivation kinetics of bacteriophages by several different doses of polychromatic MP UV radiation in PBS (pH 7.4) at room temperature based on three independent experiments. The inactivation rate of ϕX174 by MP UV was rapid, ~3.2 log10 within a UV dose of ~20 mJ/cm2, which is similar to that by LP UV. Like LP UV, the inactivation rates of bacteriophage PRD1 and MS2 by MP UV were slower than the one for ϕX174. However, the inactivation rates of bacteriophage PRD1 and MS2 by MP UV were similar and somewhat faster than the ones by LP UV, ~2 log10 with a UV dose of ~20 mJ/cm2.
3.2. Inactivation of Bacteriophages in Real Wastewaters
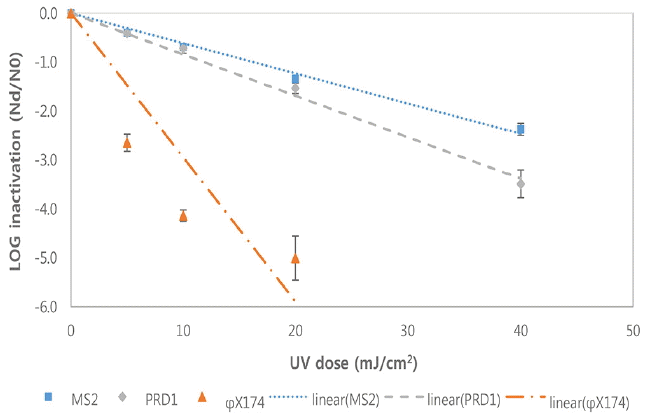
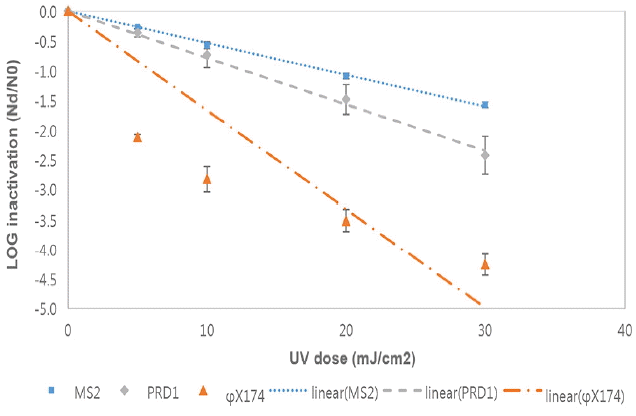
Fig. 3 and Fig. 4 show the inactivation kinetics of bacteriophages by several different doses of monochromatic LP UV radiation in two different real wastewaters at room temperature based on three independent experiments. The inactivation rates of the bacteriophages in real wastewaters were similar to the ones in PBS. That is, the inactivation of ϕX174 by LP UV was rapid and reached~4 log10 within a UV dose of 20 mJ/cm2. However, the inactivation rates of bacteriophage PRD1 and MS2 were much slower than the one for ϕX174 and only ~1 log10 inactivation was achieved by the same UV dose of 20 mJ/cm2.
Inactivation of bacteriophages by LP UV in a wastewater A at room temperature(Each point is an average of three independent experiments and error bars are equal to one standard deviation).
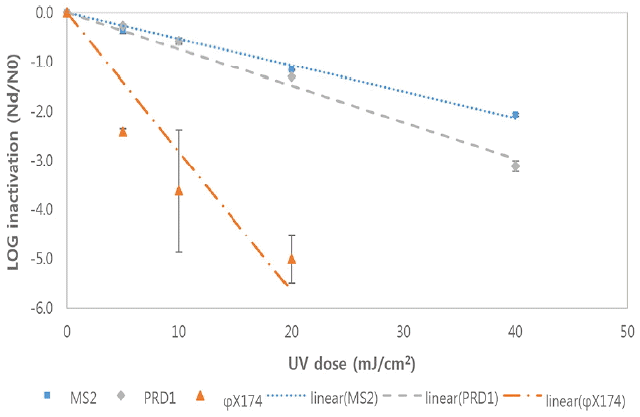
Inactivation of bacteriophages by LP UV in a wastewater B at room temperature(Each point is an average of three independent experiments and error bars are equal to one standard deviation).
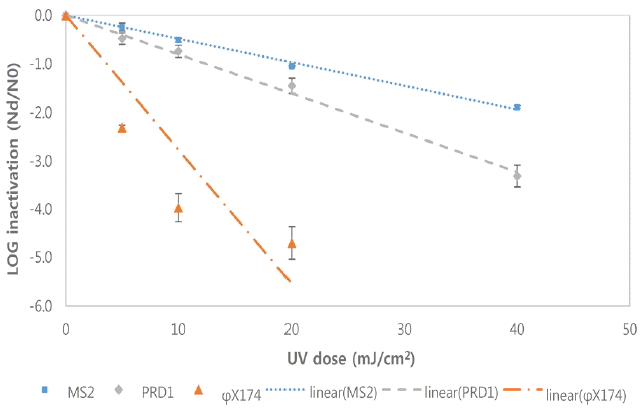
Fig. 5 and Fig. 6 show the inactivation kinetics of bacteriophages by several different doses of polychromatic MP UV radiation in two different real wastewaters at room temperature based on three independent experiments. Interestingly, the inactivation rates of the bacteriophages in real wastewater by MP UV were much slower than the ones in PBS. In fact, the inactivation rates of the bacteriophages in real wastewater by MP UV were very similar to the ones by LP UV (Fig. 3, 4).

Inactivation of bacteriophages by MP UV in a wastewater A at room temperature (Each point is an average of three independent experiments and error bars are equal to one standard deviation).
4. Discussion
The inactivation of bacteriophages in PBS by LP UV from this study is similar to those in previous study [5, 13, 14]. The results of this study showed that bacteriophage ϕX174 was the most sensitive one to LP UV and bacteriophage MS2 was the most resistant one to LP UV, which is consistent with the ones in previous studies [5, 13, 14]. Compared to LP UV, there is little study on the inactivation of bacteriophage by MP UV. The results of this study showed that the inactivation of bacteriophages by MP UV was more extensive than LP UV, which is consistent with the one in a recent study [15].
It should be mentioned that our results showed that the inactivation of bacteriophage PRD1 by LP UV was less than that of MS2, but the one by MP UV was actually similar to that of MS2, which is comparable with the recent study [15]. The results of the recent study [15] also showed that there was a relatively large difference between the inactivation of bacteriophage PRD1 and MS2 by LP UV, but the difference was a lot smaller when they were irradiated with MP UV. They suggested that it is possibly due to the more extensive repair of UV damage by bacteriophage PRD1 when it was irradiated with LP UV than MP UV [15]. LP UV lamp generates a single wavelength at 254 nm and its primary damage on microorganisms is formation of pyrimidine dimers [16]. On the other hand, MP UV lamp generates a wider range of wavelengths and some of these wavelengths (100–1,000 nm) might cause some additional damage, not only to DNA but also to various proteins in UV-irradiated microorganism. Therefore, it is possible that the additional damages to DNA from MP UV – such as (6–4) photoproduct and other photoproducts (pyrimidine hydrates, mixed purine-pyrimidine, thymine glycols, and so on), DNA cross-links and strand breaks, and sensitized photoreaction products [16] – make it difficult for the UV-irradiated PRD1 to repair and results in permanent inactivation of PRD1.
The results of this study showed that the inactivation rates of the bacteriophage in real wastewater by LP UV were similar to (or only slightly slower than) the ones in PBS. Although there are lot of suspended matters in wastewater that may absorb or scatter UV irradiation, the correction of UV irradiance by the UV absorbance of the sample and a series of correction factors (as described in the Material and Method) appears to be appropriate for LP UV, which emits only one wavelength(254 nm). On the other hand, the inactivation rates of the bacteriophages in real wastewaters by MP UV were much slower than the ones in PBS and it was actually similar to the ones by LP UV. It appears that the effect of additional wavelengths from MP UV might be attenuated in real wastewaters possibly due to absorption and scattering of the multiple wavelengths by suspended matters in wastewater, which would reduce the efficacy of MP UV against bacteriophages in wastewater.
Overall, the results of this study indicate that bacteriophage MS2 could be a reliable indicator for human pathogenic viruses for UV disinfection in wastewater treatment processes. First, it is found relatively large numbers in wastewater. Second, it is easy, simple, and inexpensive to quantify this bacteriophage. Third, and most importantly, it is relatively persistent in the environment and also very resistant to water and wastewater treatment processes, especially various wastewater disinfection processes.
5. Conclusions
In this study, we determined the response of different bacteriophages to both traditional LP UV and alternative MP UV irradiations in real wastewaters in order to identify more reliable bacteriophage indicator systems for UV disinfection in wastewater. The results of this study showed that bacteriophage MS2 was the most resistant one to both LP and MP UV disinfection in wastewaters among the bacteriophages tested. It appears that bacteriophage MS2 could be a reliable indicator for human pathogenic viruses for both LP and MP UV disinfection in water reuse practice.
Acknowledgements
This research was financially supported by the National Research Foundation of Korea (NRF-2013R1A1A2064741).