Activated Carbon: A Review of Residual Precursors, Synthesis Processes, Characterization Techniques, and Applications in the Improvement of Biogas
Article information
Abstract
The energy growing demand and the international environmental policies contribute to the use of renewable energy sources. Among these sources, biogas has acquired great relevance due to its energetic similarity to fuels such as liquefied petroleum gas (LPG) and natural gas (NG). However, biogas needs to be upgraded by removing CO2 and trace gases to obtain biomethane (>85% CH4). This review identifies and classifies seven techniques used in biogas upgrading, reported in academic and scientific publications. A 13-year review period (2008–2021) was considered. Lineal regression was used to analyze the publications number per year. Membranes use represents the largest proportion of publications (33%), while activated carbon (AC) reaches only 22%. However, the use and application of AC obtained the best trend as a publication topic, with a maximum value of R2 = 0.7882. This review documents publish works on obtaining and applying AC in biogas enrichment processes. It includes a review of the characteristics and generation rates of various residual lignocellulosic materials used in the synthesis of AC, the synthesis processes, the characterization techniques, and the final adsorption capacities.
Abstract
Graphical Abstract
1. Introduction
The residues use such as activated carbon (AC) precursors is an environmentally sustainable and economically viable method. The carbon atom (C) has an electronic configuration 1s2 2s2 2p2 that gives it the possibility of bonding with itself, with many other elements and facilitating allotropic forms. There are three types of carbon’s orbital hybridization, sp3, sp2 and sp, equivalent to three basic structures as diamond, graphite and carbyne. Carbon has different presentations with multiple applications, including graphite, graphene, carbon nanotubes, carbon fibers, carbon black and AC. Graphite has a laminated carbon structure, as condensed rings which are joined by low-energy metal-type bonds. AC’s have a three-dimensional structure of carbon atoms in flat sheets, but with no crystallographic order in the third dimension, as there is cross-linking of the sheets, causing a greater surface area [1], the spaces between each sheet are called “pores” [2]. The microcrystalline structure of AC differs slightly from that of graphite since it has a spacing of 0.335 nm and in AC it varies from 0.34 – 0.35 nm [3]. AC is generally described as an amorphous form of carbon processed to have a high surface area, high internal porosity, and certain chemical properties on its surface, such as a wide range of oxygenated functional groups that allow it to absorb or react chemically with specific elements [4–6]. Actually AC has high demand due to its multiple applications, such as the removal of organic contaminants, ions, heavy metals and dyes in both liquid and gas phases, as well as in the manufacture of high-yield capacitors and adsorption refrigeration systems [7–8]. González-García et al. [5] publish the carbon weight percentage of most common applications in the removal of pollutants such as heavy metals (59.95), dyes (8.47), organic compounds (7.41), carbon dioxide (5.82), ammonia (4.16), methane (3.66), hydrogen sulfide (1.27), nitrogen dioxide (0.61); also its use in special applications such as catalysis (6.77), capacitors (1.48), sensors (0.50), lithium batteries (0.34), pharmacists (0.30), all these based on a review of 193,250 publications reported from 1985 to 2016.
The AC world market in 2018 was equivalent to 3,100 million dollars, with a projected growth of 4.2% by 2022, with a global demand of 457.60 thousand tons [9]. In Mexico there is a monetary deficit in AC trade, since 8,687 t are exported, equivalent to 32.98 million dollars and represents 58% of national coal exports; and 14,374 t are imported at a cost of 79.32 million dollars [10], 2.4 times more than the export cost. However, the AC can be obtained in mineral form or from virgin or residual lignocellulosic materials through a synthesis process. In this context, the use of agricultural and timber waste, including agroforestry waste, is one clean and efficient option for producing plant-based carbon. Materials with high carbon content such as lignite, peat, and other biomass resources are suitable for obtaining AC [7, 11–15] Furthermore, the use of agricultural wastes as precursors to other processes is considered a renewable and relatively affordable method [16], especially considering that agricultural production in the 1969–2015 period has tripled. In 2018, the global production of cereals was 2,609 million tons, yet, by 2050, it is estimated that 50% more food, forage, and biofuel will be required to sustain the growing population [17]. In addition, there are great losses in crop production during both production and processing, and these wastes could potentially be destined for other uses. In Africa, for example, it is estimated that 15% of cereals are lost during post-harvest operations [18]. During the different stages of agricultural production (harvest, post-harvest, washing, husking, pressing or extraction and packaging), different types of waste such as stems, branches, husks, fibers, seeds and bagasse are generated as a result of quality control or food safety. In Mexico, an increase of 27.8% in agricultural production is expected for 2030, for which the National Agricultural Plan has recommended the growing of 38 strategic crops along with 14 basic crops and 24 additional crops with market potential [19]. Notably, the expected increase in agricultural production in Mexico will also translate into an increase in agricultural and agro-industrial wastes. It is important to consider treatment options along with the possibility that these wastes be added value assigned for their potential applications, which could represent an extra income source for producers or processors. In addition, the abundance and availability of agricultural byproducts and some industrial and agro-industrial wastes mean that these could potentially be a good source of raw material in the AC production [13, 20–21]. However, the AC properties depend on numerous factors, such as the precursors type and characteristics, and the synthesis method and activation processes. Other factors influencing the precursors selection are purity, easy activation, minimum and economic preparation and pretreatment requirements, easy supply, and low cost.
Moreover, at the global level, energy demand grew by 2.2% compared to the average in the last 10 years (1.7%), with natural gas (NG) being the energy source with the highest growth (3% annual) due to the coal substitution to NG in China, the Middle East, and Europe [22]. The United States [23] projects that the demand for NG will increase to 5,349 billion cubic meters (3,752 MMcm) by 2040. Since 2016, following the Paris Agreement, the fossil fuels substitution for cleaner energy sources, the fuel efficiency improvement and the energy rational use were outlined as clear strategies for reducing greenhouse gas (GG) emissions and mitigating climate change [24]. NG is considered one of the least contaminating and most efficient primary energy sources. These characteristics have led to increase its use all over the world; however, it is still a non-renewable resource, that’s why some countries are in process of substituting this gas for renewable sources with similar potential in permanent and environmentally friendly production systems. Jury et al. [25] and Qian et al. [26] mention that, based on life-cycle analysis, it is difficult for biogas to compete with NG, although it is competitive in terms of its effect on climate change on natural resources and its reduction in the demand for fossil energy. Kacem, Pellerano, and Delebarre [27] mention that the biogas produced by the anaerobic digestion of manure, agricultural residues, wastewater, and landfills has the potential to replace up to 50% of the NG demand in the United Kingdom. However, to achieve this goal, systems or processes for the biogas enrichment are necessary to reduce up to 94% v/v the concentration of carbon dioxide (CO2) and thereby enhance the gas energy value to obtain a fuel similar to NG but with the benefit of being produced from renewable sources. Yousef et al. [28] mention that the biogas cleaning consists in the gases elimination and acid impurities, while the enrichment process is based on the separation of CO2.
This review documents and multiple organize publications related to the generation and chemical characteristics of lignocellulosic residues used as CA precursors. The generation and chemical characteristics of lignocellulosic residues used as CA precursors are documented. The synthesis processes were classified into physical and chemical processes. The types of processes and the final characteristics obtained by kind of precursor were summarized. The techniques generally used in the characterization of AC are described in a simplified way. Physicochemical and energetic characteristics of biogas are compared with conventional fuels. The conditions of the biogas enrichment techniques at an industrial level, the percentages of improvement and their final applications are described and compared. Techniques reported in academic and scientific publications related to biogas cleaning and enrichment were documented and organized. It was possible to observe the growth and weighting trends for each technique. Finally, the main reports related to the improvement of biogas, through the application of CA, are summarized.
2. Agricultural and Agro-industrial Waste
The plant biomass is generally composed of cellulose, hemicellulose, and lignin. Cellulose has the highest value in the market because of its multiple applications, such as in paper production. It is a linear polymer formed by glucose molecules [29]. Meanwhile, hemicellulose contains heteropolymers composed of xylose and mannose monomers. Finally, lignin is a complex, three-dimensional structure formed by the polymerization of phenylpropane with hydroxyl and methoxy substitutes. Someone widely used cellulose and hemicellulose in the production of paper, sugars, and biofuels [30–32]. Cellulose and many of its products are considered innocuous given their capacity to reincorporate into the environment by the action of decomposing microorganisms. However, large volumes of cellulosic materials currently generated in agricultural or industrial activities are wasted because their economic value is underestimated [33]. In addition, lignin is an abundant, low-cost resource in the market that is used as a renewable precursor of different added-value products, although less than 2% of the lignin produced annually is re-utilized.
Lignin is one of the main plants components and represents 15% to 40% of dry weight [34, 35]. Lignin provides rigidity and cell wall cohesion, confers impermeability to water, and forms a physical and chemical barrier against microbial attack [36, 37]. Currently, the most common application of lignocellulosic materials in agro-industrial processes is combustion for energy production. Although its use as fuel can save costs, it is necessary to add value. In this regard, it is important to advance in technology to increase the lignin potential as a versatile raw material [34]. Research has been performed on the conversion of lignin in synthetic resins and surfactants, although it would be interesting to explore its use in the fine chemical products production as a substitute for different substances usually obtained from petroleum, which would help to reduce the fossil fuels consumption of and decrease environmental contamination [34]. It is important in further studies to explore the lignin different applications, its fundamental chemistry and physicochemical transformation during different processes [35]. Lignin has been identified as the main component of the lignocellulosic biomass responsible for adsorption [38].
Agricultural or agro-industrial wastes are commonly used to generate heat and vapor as well as synthetic fuels, oils, methane (CH4), ethanol, biodiesel, methanol, and vegetal carbon. The waste conversion into other materials is a general strategy to support or to improve the production processes sustainability [39–41]. Several common agricultural crops and their associated wastes are listed in Table 1.
It is important to know waste yields or rates generated per production unit to quantify or estimate the potential use of residual biomass. Ajewole et al. [46] report that, from a metric ton of recently harvested cocoa pods, 240 kg of dry beans and 506 kg of shells or dry pods are obtained. Which would represent a 24% product yield per ton and around 50% waste per ton. White et al. [47] reports residual biomass generation rate of 496.9 kg ha−1 in sugarcane crops; extrapolating to 395 million t of residues, with respect to the global cultivation area. Other authors report that the amount of residual biomass in sugarcane cultivation represents 15 to 25% of production [48, 49]. In the case of plantain cultivation, a 220 t ha−1 waste generation is estimated [50]. Subramaniam et al. [51] calculate a residual biomass generation of 13.55 t ha−1 a−1 in the oil palm industry. The rice straw generation is equivalent to 45% (227.78 million tons) of the total rice production in the main producing countries [52]. In soybean processing (milk and tofu production) 1.2 kg are produced for each kilogram of product [53]. In the production of 1 kg of coffee generates 1 kg of residual coffee husk [54].
Some of the factors that enable the crop waste use feasibility are the yield, the moisture content, and easy degradation (for post-harvest crops). If these factors are discouraging (low quantity, high moisture content, and easy degradation), wastes are usually used as food for animals, burned (with or without energy recovery), or re-used in crop fields. Lower quantities wastes generated from agro-industrial processes are often used in fuel boilers. In Mexico, Secretary of Energy of Mexico (SENER) [55] reported that, 79 bioenergy plants that utilize agro-industrial and solid urban wastes were registered with an installed electrical generation capacity of 996 MW, and the electrical energy production was 1,884 GWh, 81% with sugar cane residues use and 13% with biogas at the end of 2017.
2.1. Composition of Agricultural and Agro-industrial Waste: Precursors of Activated Carbon
The lignocellulosic composition plays an important role in AC synthesis, mainly the lignin content. Since the complexity and high resistance make biomasses with high lignin content attractive for use in AC synthesis. Lignin is mainly composed of three elements: carbon (C), hydrogen (H) and oxygen (O) [34], although its complexity lies in the phenylpropane units, mainly syringyl, guaiacyl and p-hydroxy phenol [56], being complex amorphous polymers with three-dimensional spatial structure [57, 58], this allows lignocellulosic materials to be resistant to chemical and biological conversions [59]. Materials with high lignin content are more attractive for use in AC synthesis.
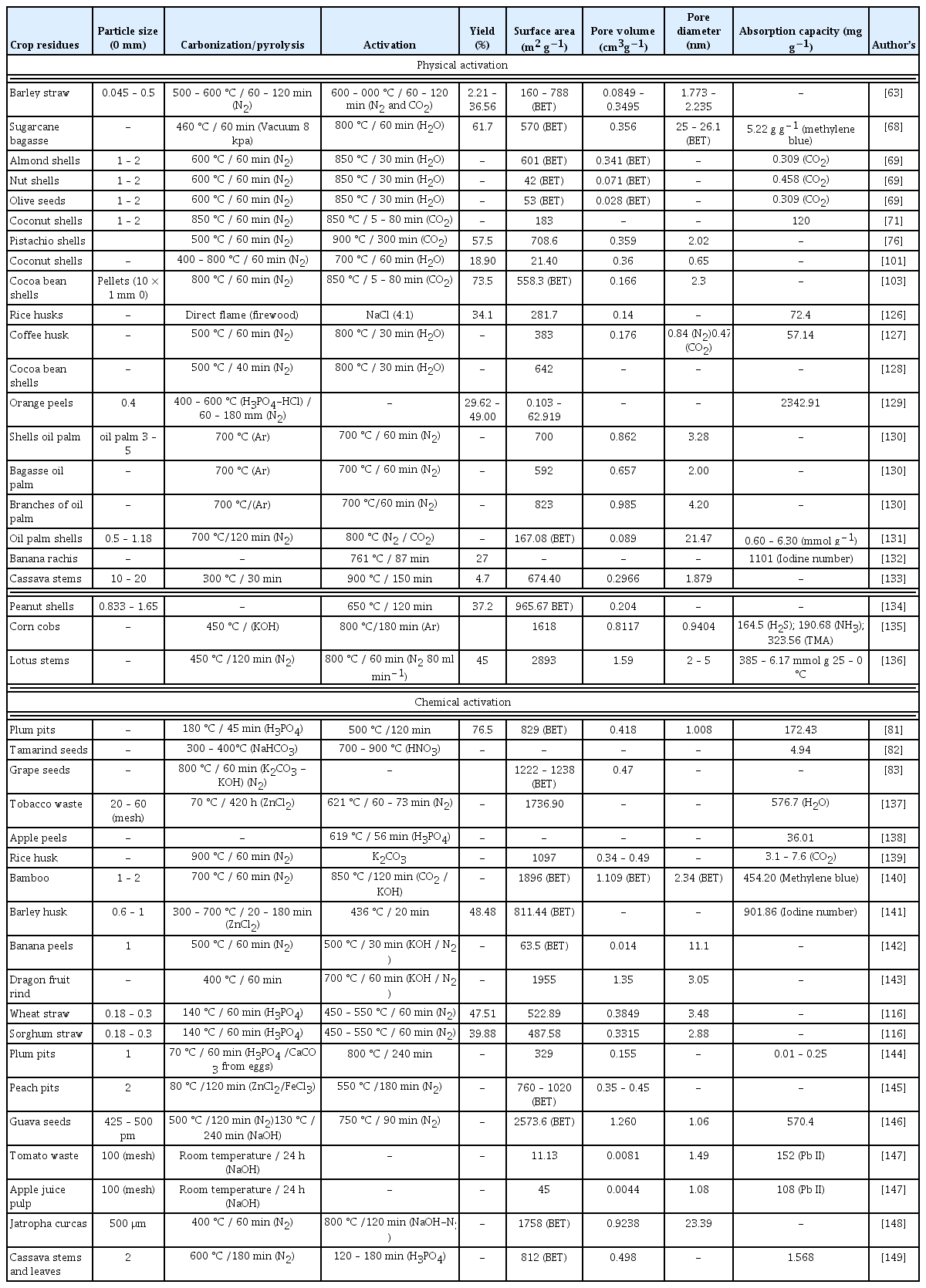
The composition or compounds present in lignocellulosic materials can be analyzed to evaluate their possibility of use or synthesis (Table 2). Two types of analysis are necessary for precursors: proximate analyses, such as humidity content, organic matter, ash [60], and fixed carbon (100% minus the sum of the previous contents); and ultimate analyses, including the elemental content of C, H, O, nitrogen (N) and sulphur (S). The proximate analyses enable the AC yield from synthesis processes to be calculated considering that a high amount of volatile material indicates carbonization losses, high content of fixed carbon indicates good yield, and a high moisture content indicates the need for drying processes due to the low yield of usable dry matter. For the elemental analysis, the use of automated equipment is currently the most employment technology for these analyses, although equipment such as LECO® elemental analyzer (CHNS 932) still requires the oxygen content to be calculated by subtracting the percentages of ash, C, H, N, and S, from 100%. For instance, the low presence of O in the carbonized material indicates the reduction of oxygenated functional groups, which has an impact in more alkaline materials or those with greater hydrophobicity [61, 62].
It is important to know the methods and the conditions under which these analyzes are carried out. For example, the proximal analysis can take as reference the ASTM D 2974 standard. It is important to report the specific use of times, temperatures, quantities, required pretreatment and type of material (soils, organic matter, organic waste, etc.). For the elemental analysis method, ASTM-D 5291 serves as a reference with the use of an elemental analyzer CHN-LECO 800, based on combustion in a furnace. For lignocellulosic determination, ASTM D 1106 is used to determine acid-insoluble lignin. The determination of cellulose is made based on its solubility in a sodium hydroxide (ASTM D 1696). however, prior to these lignocellulosic tests, it is necessary to carry out two extractive tests, one of them using the Soxhlet extraction apparatus (ASTM D 1107) and a mixture of solvents such as ethanol/toluene or acetone/hexane (2:1 ratio). The second extraction with hot water is extremely important (ASTM D 1110). The extraction stages are used for the removal of dyes, sugars, gums, tannins, and other substances soluble in both water and solvent. The determination of hemicellulose is generally carried out by difference of the sum of the concentration percentages of lignin and cellulose with respect to 100%. The lignin result must be adjusted with the ash content.
One relevant technique for the precursors characterization is thermogravimetric analysis (TGA). TGA is a thermal analysis technique, in which the change in the amount of mass of material is analyzed as a function of temperature and/or time, under a controlled atmosphere. The procedure is to expose small samples (less than 5 mg), to temperature increase between 5 to 20 °C min−1, until temperatures greater than 1000 °C or more are reach, using inert gases (N2, Ar, He), oxidants (air, O2) or reducers, meanwhile the equipment balance continuously determines the sample mass [87]. Miranda et al. [88] report the use of TGA on orange peels under heating velocities of 1, 5, 10, 20, and 40 °C min−1, range temperatures 25 to 1000 °C, and N2 flow of 60 ml min−1.
3. Synthesis of AC
Promdee et al. [89] highlighted that AC is produced in three stages: a) pre-activation, which involves the determination of the required quality and correct size; b) activation, or the process in which precursors are converted to AC; and c) post-activation, which consists of the output parameters, quality control and final AC characteristics. There are two general methods for the preparation of AC: physical and chemical processes. In physical treatments, the precursors are first carbonized and then activated under inert gas atmospheres such as water vapor (H2O), CO2, or nitrogenated compounds (N2 or NH3). In contrast, in chemical treatments, the precursors are first impregnated by a reagent and then submitted to a heating process in an inert atmosphere [90]. In particular, the reactive agents promote the formation and extension of cavities and reticulated structures [91, 92]. For this, some of the most utilized chemical agents are H3PO4, ZnCl2, KOH, NaOH, H2SO4, CaO, and HF.
3.1. Pyrolysis
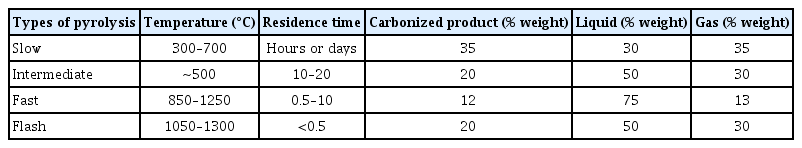
Pyrolysis is a thermochemical conversion method in which complex hydrocarbons are reduced to added-value products by heating in the presence of low levels or absence of oxygen. In this process, the biomass is heated from 300 to 800 °C, and three products are obtained: a carbonized solid (biochar), a liquid phase product, and a non-condensable gas-phase product, such as CO, CO2, CH4 or H2 [93, 94]. Pyrolysis can be classified into four types (Table 3) according to temperature, residence time, and heating velocity.
During thermal decomposition, biomass components undergo reactions such as dehydration, crosslinking, repolymerization and depolymerization, fragmentation, rearrangement, condensation, and carbonization [96–98]. Slow pyrolysis involves longer residence times and slower heating rates, producing similar composition products, and is carried out at near-atmospheric pressure conditions [96]. Slow pyrolysis is the most widely method used to obtain AC since it generates the largest amount of charred material. New methods using microwaves, for example, can be used to generate hot spots in carbon particles and bulk solids, representing a promising method to obtain AC under conditions from 0.91 to 2.45 GHz [99]. However, the carbonaceous product (biochar) obtained from the biomass thermal conversion under poor or zero oxygen conditions has a less efficient surface compared to AC, although biochar is less expensive to produce than AC [62].
Chen et al. [100] describe described and classify the carbonization process into four temperature ranges. In the first stage (25–150°C), the physical desorption of approximately 12% of the absorbed water occurs with a small degree of change in order; second stage (150–240°C), the dehydration of the =H and -OH fragments occurs on an intramolecular scale; third stage (240–400°C), thermal rupture of the glycosidic bond in other C-O bonds and some C=C, through a reaction of free radicals, forming large amounts of tar, H2O, carbon monoxide (CO) and CO2; fourth stage (>700°C), a graphite-like structure is obtained, with disordered layers that tend to align with increasing temperature.
3.2. Activation
The porosity and selectivity are great interesting characteristics in the production and use of AC. These characteristics dependent on the disorganization level in the monocrystalline structure, therefore, they depend on the precursor type, synthesis process and activation method used [101]. Pallarés et al. [63] highlighted that the precursor carbonization (300–800 °C) causes the less stable bonds rupture, liberating the precursor volatile fraction (gases and tars), resulting in carbonaceous residues enriched with aromatic carbon rings (plant carbon) with low adsorption capacity due to the tars repolymerization and their deposition on the material surface, filling in the formed pores. Therefore, a post-activation step is necessary to remove tar deposits and increase both porosity and adsorption capacity. For this reason, the temperature, time, and agents used for activation are essential for developing and enhancing the microporosity of AC [102].
The activation process generally requires high temperatures (700–1000 °C) and an activating agent presence. During this stage, several reactions occur, such as the tar deposits removal and the opening and new pores formation [63]. There is debate that higher temperatures favor CA adsorption by increasing its porosity [102]. Some authors indicate that a larger pore volume is obtained at low combustion temperatures and larger pores at higher combustion temperatures [103]. Pallarés et al. [63] highlights that, in the activation, the pores become larger with longer times, however, the new pores formation and the existing pores depth are severely reduced. Consequently, with prolonged times, more meso and macropores develop, as well as surface area (BET method, Brunauer, Emmett and Teller), however, the pore volumes decrease [104]. The formation and increase of porosity in the morphology of ACs directly influences the functionality and application of certain types of AC. Qambrani et al. [96] explain that the micropores are responsible for the surface area and the high absorption capacity; Mesopores are important in liquid-solid adsorption processes, and macropores influence aeration, solid-gas adsorption, and hydrology.
Physical activation is carried out primarily with steam and gas to produce significant physical changes in surface area, pore volume, and pore structures or to affect surface chemical properties, including surface functional groups, the surface, hydrophobicity and polarity [105]. The use of CO2 and H2O is endothermic in nature, which allows better control of the process. Ahmad et al. [103] point out that the use of CO2 favors the microporosity formation during the first stages of activation. Pallares et al. [63] concluded that the use of CO2 increases the microporosity by 43% and that the application of high temperature water (800 °C) reduced the BET surface area (−3%) and the micropores volume (−5.4 %), increased the total pore volume (9.6%) due to enlargement of the mesopores, and increased the activation time from 1 to 2 h, which led to a reduction in microporosity due to the enlargement and collapse of the mesopores. Table 4 presents a summary of the different synthesis methods organized by physical and chemical activation techniques.
Both temperature and time are vital to optimize AC functionalities, especially when there are prepared for special needs or applications. Therefore, the activation process not only influences the AC morphological modification but also affects the chemical surface. During each stage of the synthesis, the formation of important compounds that promote better adsorption capacities occurs. but they are also affected by time and temperature conditions. An important step is the cleaning of the synthesized ACs. In the use of chemical activators, the AC must be washed with water or alcohol, but in physical activations, it is recommended to apply a vacuum devolatilization process, at relatively low temperatures, from 100 to 250° C, at −1 ATM. This procedure removes impurities in the materials, causing pore saturation and, in some cases, reducing the adsorption capacity of the materials.
4. Surface Chemistry
The AC surface has a great advantage, which is the ability to attract and make simple bonds with certain types of substances or compounds due to its electrostatic power. On the AC surface, the adsorption mechanism can be by physisorption and/or chemisorption. These adsorption mechanisms usually favor the contaminants’ removal in liquid or gas phases, into a solid phase. The AC modified surface increases the selectivity and the efficiency of certain compounds’ immobilization. On the AC surface, the presence of functional groups is related to the presence of heteroatoms (C and H atoms) and O, N, S, and P [106]. These functional groups develop from the chemical interaction between the AC network (the external surface or at the edge of the basal plane) and the radicals supplied by the chemical agent used for synthesis [107]. The delocalized heteroatoms and electrons determine the acid or basic AC surface character [108]. Finally, the surface chemistry influences the AC adsorption capacity of, as shown below.
4.1. Acidic Surfaces
The surface complexe formation such as carbon-oxygen, carbon-hydrogen, carbon-nitrogen, carbon-sulfur, and carbon-halogen has been demonstrated by X-ray diffraction analysis [3]. Oxygen groups come from a wide variety of groups such as carboxylic, hydroxyl, carbonyl, lactone, phenol, pyrone, chromene, quinone, and ether [106, 109–111] Oxygen complexes provide electron-rich properties, which can enhance the interaction between the carbon absorbers surface and CO2, due to the introduction of several polar groups, such as hydroxyl (OH) and carboxyl (COOH) groups [112]. The carbon-oxygen complex has various classifications such as acid, base or neutral [108]. There is an effect of the synthesis temperature and activation in the formation of carboxylic groups considered strong acids at low temperatures, while phenolic groups are considered weak acid groups at high temperatures. The temperature effect on carbonization and activation is linked to the presence or absence of functional groups. Bansal and Goyal [3] indicate that the surface oxygen groups developed in the carbonization stages are desorbed by high temperatures in the activation stage. For example, CO2 is released by the decomposition of carboxylic and lactone groups in the temperature range of 350 to 750°C; CO due to decomposition of quinones and phenolic groups in the temperature range of 500 to 950°C; H2O from the decomposition of carboxyl, phenols in the temperature range from 200 to 600°C.
The AC precursors chemical characteristics influence the formation of oxygen functional groups, as well as the oxidation mechanisms. In gas phase synthesis, an increase in hydroxyl and carbonyl functional surface groups has been shown, while in the liquid phase, carboxylic, quinone and phenolic groups increase [113, 114]. The gaseous agents that promote oxygen groups are air, O2, CO2, and H2O [106]. The liquid agents are HNO3 and H2SO4 [111–114], these particularly favor the formation of carboxylic, lactone, and phenolic hydroxyl groups [109, 115]. Functional groups containing oxygen have a higher affinity for alkaline gases [107]. Chemisorption by oxygen complexes achieves the decomposition of oxidizing gases (O3, NO2, CO2) and various aqueous solutions (FeCl3, K2S2O8, (NH4)2S2O8, NaClO, KMnO4, K2Cr2O7, Na2S2O3, H2O2, HNO3), mainly by O2 chemisorption [3]. Therefore, the oxidation mechanisms are important in the AC synthesis.
4.2. Alkaline Surfaces
In general, the basic surface AC characteristics result mainly from delocalized beta electrons in the graphene layers [116, 117], which can act as Lewis bases [118]. However, nitrogen functional groups are associated with basic functionalities on the AC surface. The reaction of compounds such as NH3, HNO3, and amines, or by activation with nitrogen-containing precursors [106, 108, 120]. Another formation route is when the carbon surface is freed from its oxygen groups by thermal treatment in vacuum or in inert atmospheres at high temperatures (1000°C) and cooled to room temperature, with contact with oxygen gas [3].
The functional groups associated with the AC basicity are amid, imide, lactam, pyrolic, and pyridine [35, 38, 91]. Other groups, such as chromene [120], ketone [119], and pyrone [117] (functionalities containing oxygen), contribute to the AC basicity. The AC basic property increases the CO2 adsorption capacity [118, 121–124] and H2S [125]. AC with basic properties is related to the removal or the equilibrium reaction between basic and acidic species.
5. AC Characterization Techniques
It is important to characterize changes on the surfaces and in the AC structures and to identify the presence and new compounds formation giving characteristics as adsorption and absorption’s capacity and selectivity. Different techniques can be used depending on the availability of technology; however, they can be classified into two types, physical and chemical techniques.
The physical techniques consider the structure and AC morphology according to its surface area, size, and pore volume. Below, the main techniques for the characterization of AC are described.
5.1. Scanning Electron Microscope
Scanning electron microscope (SEM) is one of the most utilized methods for visualizing the surface morphology of different materials. The images are obtained by scanning with a very narrow electron beam through a sample, measuring the scattered electrons and mapping them in space, achieving magnifications of 10 – 50 kX, and resolutions of 50 – 100 nm [150]. Depending on its electron source, SEM can be classified into thermal and field emission, and depending on the chamber conditions, at high vacuum or ambient pressures. When materials with high electron absorption capacity are available, it is necessary to cover the samples with thin films of metals such as platinum or gold, achieving resolutions of up to 5 nm [124]. Ronix et al. [151] mention the use of SEM in carbonized samples of coffee husks, managing to observe the development of porosity with the formation of cavities of different sizes and their distribution on the surfaces, in comparison with samples of precursors, which present a rigid morphology. and compact, with no apparent cavities and no pore structure.
This technique enables the surfaces and pore structures of AC to be characterized. However, to clearly view the surface morphology of materials with high electron absorption capacity, they must be first coated with gold [124]. Although there are techniques with higher resolution power such as scanning probe microscopy (SPM) or atomic force microscopy (AFM), which have been applied in the characterization of black carbon, they managed to observe structures at a scale of 5 – 10 nm (SPM) and 1–2 nm (AFM) [152].
5.2. Adsorption Capacity
Among the common techniques to determine the AC adsorption capacity, there is the iodine number method and the methylene blue method. Both methods determine the carbons’ relative activation level based on the quantity of adsorbed iodine or methylene blue solution in milligrams per gram of carbon [153, 154]. In some cases, the use of techniques such as ultraviolet rays (UV) to determine the absorbance allows more accurate readings of the initial and final concentrations. Adsorption isotherms are another commonly utilized technique for representing the interaction between the adsorbate and adsorbent in addition to the distribution of the adsorbate between the liquid phase and solid phase under equilibrium conditions. In particular, the isotherms of Langmuir and Freundlich are frequently used. The first is based on the affinity of the binding sites and is used to identify the saturation point, or moment, in which all active centers are occupied (monolayer). Meanwhile, the second is based on an empirical model that describes non-ideal multilayer adsorption on the heterogeneous surface of an adsorbent [154]. Chemical techniques are considered qualitative or quantitative according to the available procedures and applications. Qualitative techniques are based on the identification of the chemical species present, whereas the quantitative techniques are used to determine the number of chemical species present on the carbon surface [107].
The BET method utilizes the multilayer adsorption framework under two principals: The first assumes that the surface is energetically homogeneous, assuming that there is no energy variation in adsorption across a single layer, whereas the second assumes that the adsorption-desorption velocities are similar according to a model of kinetic equilibrium. This method utilizes the multilayer adsorption framework under two principles: The first assumes that the surface is energetically homogeneous, assuming that there is no energy variation in adsorption across a single layer, where the second assumes that the adsorption-desorption velocities are similar according to a model of kinetic equilibrium. The BET equation describes the full course of the isotherm (Eq. 1), including the areas of monomolecular adsorption, polymolecular adsorption, and capillary condensation.
q : adsorption capacity (mg adsorbate adsorbed per g adsorbent)
B : Constant expressing the interaction of energy with the surface.
qmax : solute adsorbed per unit weight of adsorbent in forming a complete monolayer on the surface (Ms/Mc
Ss : Solute saturation coefficient (Ms L3−1)
Se : equilibrium adsorbate concentration, (Ms/L3)
The BET theory starts from inferring that only the first adsorbed layer is strongly attracted to the surface, the second layer is essentially adsorbed by the first layer, and finally, the adsorption propagates to result in multilayer adsorption, although it is not necessary. That a layer is completely formed before the beginning of subsequent layers and Van der Waals forces would be responsible for adsorption [2]. The technique used in the analyzers generally begins with degassing at 25°C, from 12 to 24 h, subsequently, a saturation of the material with nitrogen gas (N2) is carried out, as a sorbent at temperatures of 77 K and desaturation pressures of <660 mmHg [91]. Invariably, the BET technique has been the most widely used in the characterization of adsorption capacities.
5.3. Raman Spectroscopy
Raman spectroscopy (RS) is a technique to identify and analyze the structural footprint of functional groups on AC surfaces by measuring the photon scattering energy. In this technique, AC is exposed to monochromatic light from a laser. Although some beams are elastically scattered, RS depends on the inelastic scattering of photons. Samples may be then observed through revolution or in their different rotational or vibrational states [155, 156].
The energy gradient between the incident and reflected beam is measured in terms of the wave number (cm−1) [157], which corresponds with the molecular vibration within the adsorbent, enabling the chemical species in a sample to be identified [158–160]. This technique presents advantages because of the minimal spectral manipulation, simple data interpretation, high spectral recording speed, high spatial resolution, and relative ease of preparation. Also, it allows the simultaneous and clear surface species identification without interference from water and serves as a complementary method to infrared spectroscopy [161, 162]. However, degradation effects may affect the sample, including the fluorescence and sub-development, possibly leading to an incomplete understanding of the provided information [107].
5.4. X-ray Photoelectron Spectroscopy
X-ray photoelectron spectroscopy (XPS) is one of the most utilized techniques in the quantification and characterization of the elemental composition, chemical state, and oxidation of elements and radicals of the surface structures of AC. It is also called electronic spectroscopy for chemical analysis [106, 163–165]. In this technique, the study material is subjected to radiation by x-rays, and the electrons liberated from the material are then measured, analyzing the upper (0 nm) to lower portions (10 nm) of the material and their kinetic energies [107]. Susi et al. [165] described the method in three stages: (1) Photons from x-rays are absorbed, and their energies are transferred to a nucleus electron. (2) Select atomic species are excited, emitting photoelectrons, and resulting in an empty core state. (3) Emitted photoelectrons are transported to the surface of the material before escaping the void where the energy content is measured with the help of an electron analyzer. When using XPS in the identification, concentration, and binding of doping groups, measurements of the samples prepared for analysis should be considered, specifically the intrinsic photoemission response and presence of relevant spectra. These determinations can reduce the risk of making incorrect conclusions about the material or its characterization, especially in the assignment of linking energies to the respective atomic configurations during the composition analysis [107].
Tongpoothorn et al. [166] performed XRD analysis on AC obtained from Jatropha Curcas husks. They exhibit broad peaks and the absence of a sharp peak, revealing a predominantly amorphous structure. In addition, they mention that the frequency of broad peaks around 26° and 43° shows signs of the formation of a crystalline carbonaceous structure, equivalent to an alignment of graphene layers. Keiluweit et al. [167] classified the structural changes in cellulose (grass) and lignin (wood), in CA synthesis at different temperatures. The XRD data emerge that from 300 to 400 °C the cellulose crystallinity of chars disintegrates and randomizes, while turbostratic crystallites evolve at char temperatures above 400 °C. There are some variants, such as X-ray photoelectron spectroscopy (EFRX) and analysis of structures near the X-ray absorption edge (ECBARX), which have contributed to improving the quality of the analyses.
5.5. Fourier Transformed Infrared Spectroscopy
Fourier transformed infrared spectroscopy (FTIR) consists of passing a light beam at different frequencies on a sample to measure the absorbance or transmittance of infrared light [168, 169]. The obtained spectra is graphically represented as the infrared density as either transmittance or absorbance (cm−1) against the wave number. This technique has several advantages: It is highly sensitive, has a good signal-noise relationship, produces high-quality spectra, has a non-destructive focus, can detect species at low concentrations, and can be used to analyze multiple gas species at the same time. However, it is only suitable for compounds with a dipole moment, as compounds without a dipole moment do not absorb infrared rays and, therefore, cannot be characterized by FTIR [170, 171]. In the quantitative evaluation and infrared spectra interpretation, errors may be generated since absorption bands do not always represent a specific functional group, but may be formed as a result of the overlapping of several bands representing different groups. However, obtaining infrared spectra from AC is often a challenge due to irregular light scattering caused by the large adsorbent particle size, poor transmission, or problems associated with sample preparation [107, 172].
5.6. Boehm Titration
Boehm titration (BT) is a wet characterization technique. It is an acid-base titration method that was originally developed to identify and quantify the acidic oxygen functional groups on the surface of modified and AC carbonized according to their different acid strengths according to the acidity constant, Ka [173, 174]. The basic principle is that strong acids potentially react with all types of bases, whereas weak acids only donate protons to the conjugated bases of acids with higher pKa values; therefore, functional groups with different acidity can be measured against suitable bases of different strength [107]. The technique classifies acid groups according to three ranges of pKa strength, 5.0–6.4, 6.4–10.3, and 10.3–13.0, which correspond with the carboxylic, lactone, and phenolic groups, respectively. In the titration of AC with three different bases (NaOH, Na2CO3, and NaHCO3), each functional group will react with the bases that have a conjugated acid with greater pKa [175, 176]. During titration, it is assumed that NaOH easily reacts with the carboxylic, phenolic, and lactonic functional groups and that Na2CO3 reacts with carboxylic and lactonic groups. Meanwhile, NaHCO3 only reacts with carboxylic groups [107, 175, 177–179]. Also, the basic functional groups can be identified and quantified by reaction with hydrochloric acid based on the quantity of HCl consumed during the evaluation compared to the original AC sample [178, 180, 181].
6. Biogas Enrichment and Upgrading Processes
Biogas by itself does not have the energy capacity that allows it to compete directly with fuels such as LP gas or natural gas (Table 5, part A). However, the importance of upgrading and enrichment processes is aimed at increasing or improving these characteristics. Another important factor is the substrates used to obtain biogas, since it has different characteristics in their composition, for example, substrates from farms, sewage sludge and landfill. The particular characteristics of each substrate result in different ranges in the biogas constituent compounds (Table 5, Part B). These differences in the biogas “quality” is a key piece for the selection and implementation of upgrading and enrichment systems and processes, with the aim of achieving the necessary criteria for the use of biogas or biomethane, in combined heat and power systems or in its injection to grid gas.
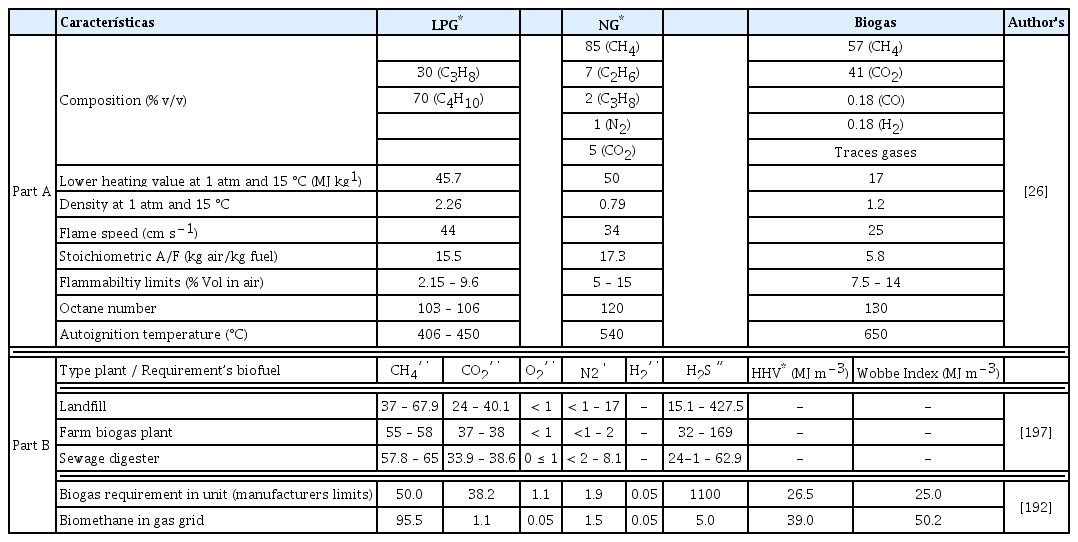
Characteristics of Gaseous Fuels (Part A); Characteristics of Biogas by Source and the Requirements for Its Use (Part B).
The heat capacity of the presented fuels has important differences. However, analyzing the specific heats of each hydrocarbon, CH4 has the highest specific heat (1,708 kJ kg−1), followed by C4H10 (1,551 kJ kg−1), C3H8 (1,480 kJ kg−1), and C2H6 (kJ kg−1). However, the presence of CO2 in biogas reduces its heat capacity total, thence, the need to remove the CO2 immersed in the biogas, to reach similar values to NG.
6.1. International Enrichment and Upgrading of Biogas, and AC Application
The case of Denmark is one of the most important examples of the biogas use in local heat and energy generation plants (combined cycle). The government has aimed to increase the manure use, currently at 10%, to 50% by 2025, which would correspond with an increase in energy production of 7 to 17 PJ. The use of manure represents a good strategy because of its constant production throughout the year, especially considering that its storage is expensive [182]. It has been shown, that biogas produced from manure is a good substitute for NG when quality is improved.
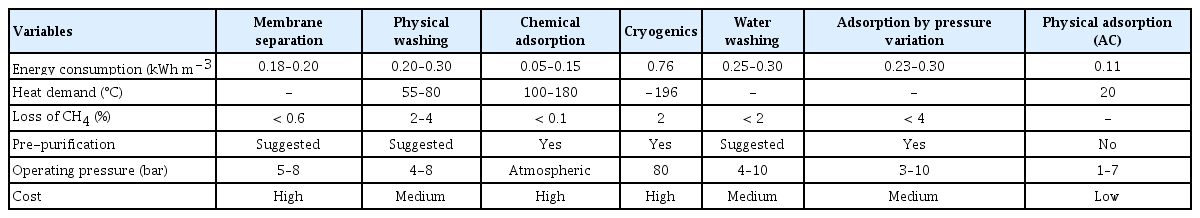
The International Energy Agency [183], with 37 member countries lists up to 2019, 602 biogas upgrading and enrichment plants. 70% of these plants are in countries such as Germany (203*), the United Kingdom (96*), Sweden (69*) and Netherlands (54*). The main uses of biogas upgrading, and enrichment plants are injection and conduction in the gas red (271*), vehicle gas and fuel (25*), CHP, electricity, and heat. The methane increase reaches ranges of 88 – 99% v/v. The main processes used are membrane (184*), scrubber (176*), chemical scrubber (103*), adsorption by pressure variation (79*) and organic physical scrubber (17*), catalytic methanation (1*), biological methanation (1*). Therefore, multiple biogas enrichment techniques have been implemented, with characteristics and requirements as shown in Table 6.
Historically, adsorption processes using different media have been developed under two operating schemes, a liquid-gas or solid-gas contact, at both industrial and commercial scales. The combination of these two interfaces is common, for example, packed bed adsorption columns, spray columns, bubble columns, rotating beds, mini-channels, and contact membranes, to mention several. One of the additional advantages of using AC for CO2 capture compared to other technologies is its low cost. Farooq et al. [184] reported that AC has a lower cost in terms of capital and operation cost in comparison to washing with water, which has costs of 1.5 and 0.9 p kWh−1, respectively. In addition, the large amount of wastewater that is usually generated can be reduced. Therefore, this method represents a more sustainable form of biomethane production.
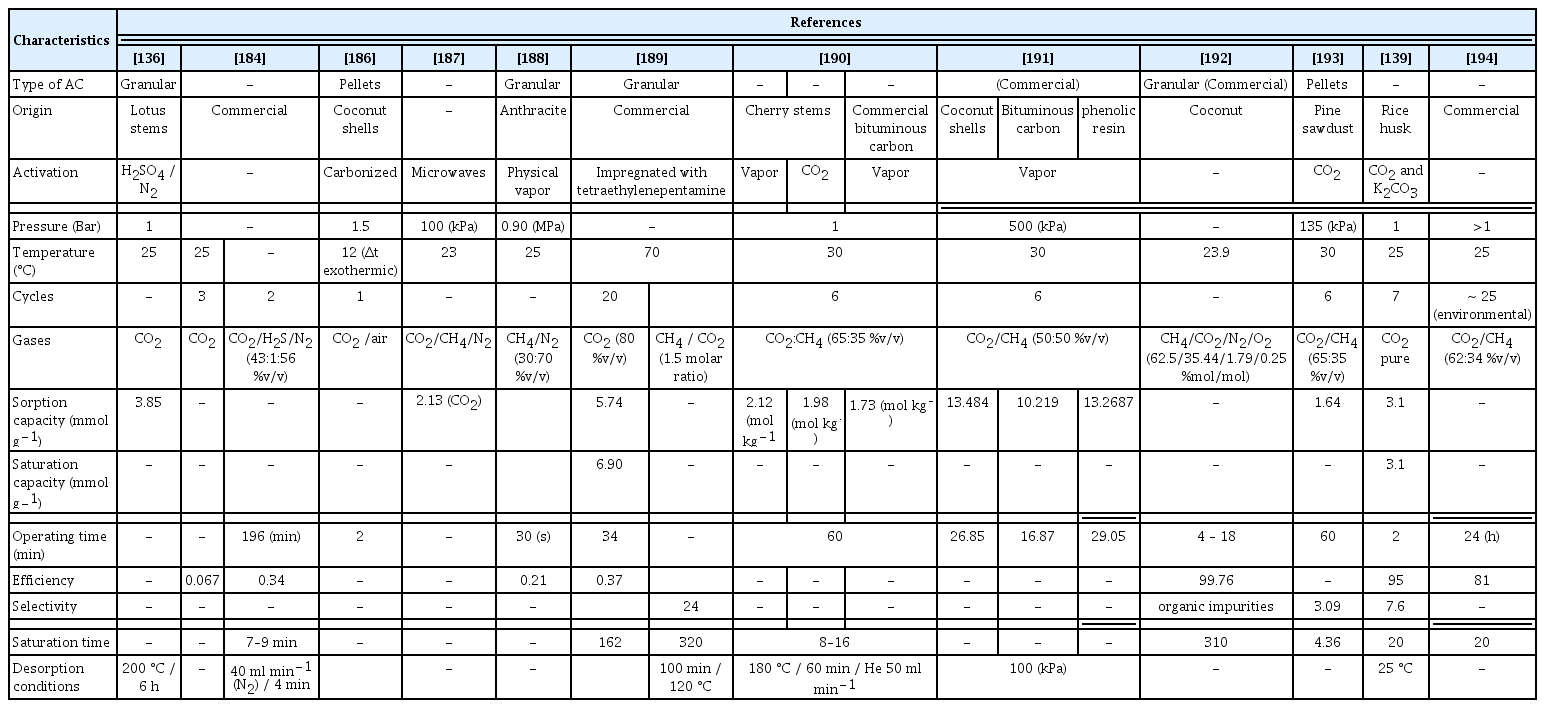
The most important variables in the use of adsorption columns in the capture of CO2 are column height and diameter as well as the packing material, elevated surface area of the packing material, resistance to wear under operating conditions, low energy consumption, and easy regeneration. In Table 7, a summary of the diverse applications of AC in the capture of CO2 is provided, along with their key characteristics.
6.2. Art State of Topics Upgrading and Enrichment from Biogas
To know the state of the art, research articles published by Elsevier® and EBSCO® host were reviewed. Through the corresponding online databases, articles on topics related to biogas upgrading and enrichment were searched during the 2008–2021 period, comparing the number of articles for each method (dependent variable) with respect to period time (independent variable). 164 articles were found and organized by six techniques, as shown in Fig. 1.
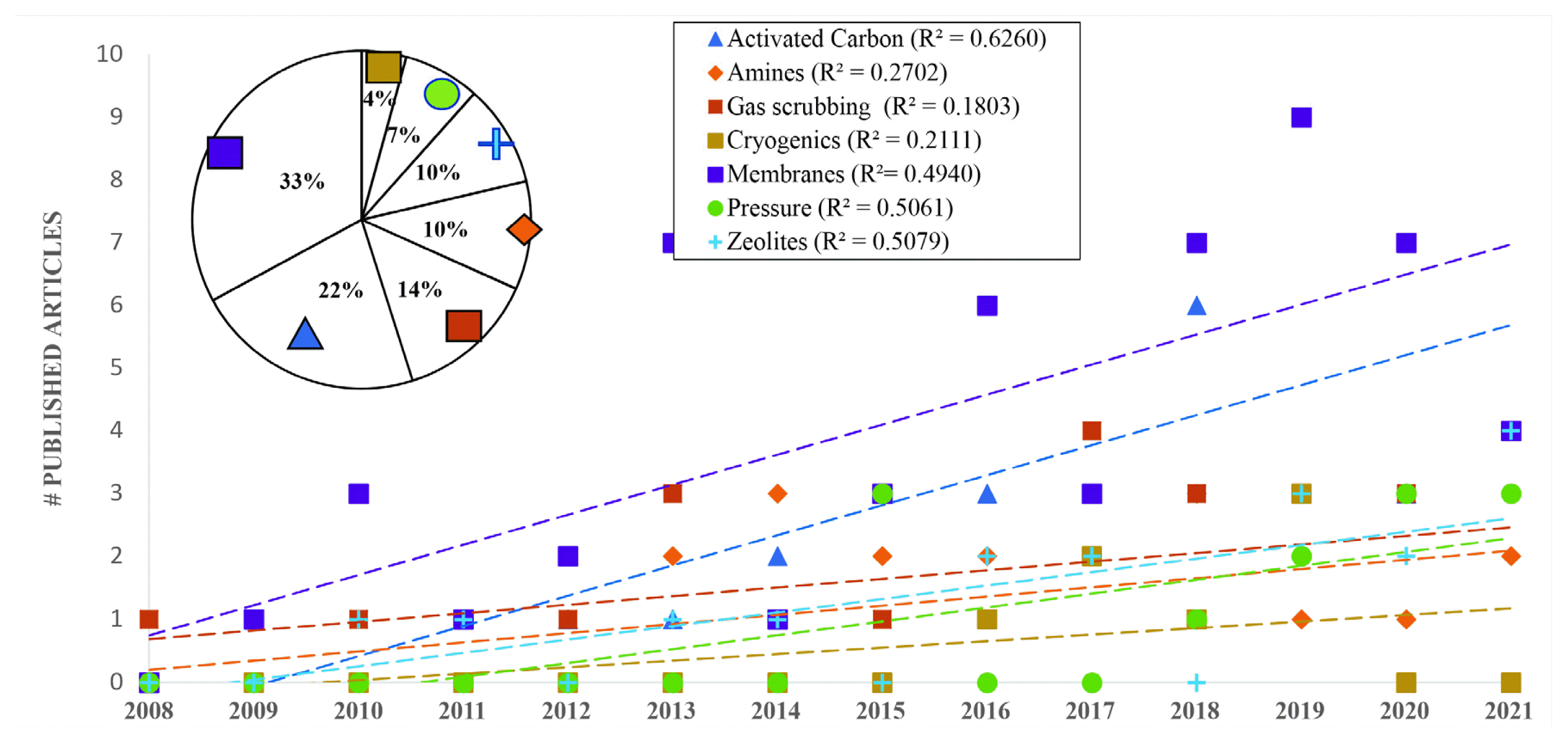
Tendencies in techniques for the upgrading and enrichment of biogas in published scientific articles
Most articles were related to membranes (32.93%), AC (21.95%), gas scrubbing (13.41%), and zeolites (10.37%). Linear regression was performed to determine the trend in the number of publications per year. As a result, the technique or use of AC obtained the greatest tendency with a value of R2 = 0.6260, indicating continual growth in the study of biogas for the topics of interest. However, a decrease in publications is observed in the years 2020 and 2021. Discarding these two years, the trends present a better fit, for example AC increase in R2 = 0.7882, membranes in R2 = 0.5641 and gas scrubbing in R2 = 0.4149.
Absorption and adsorption processes have been frequently implemented in the cleaning and enrichment of biogas. Although absorption processes have a high separation capacity, they also have a high cost because of the high energy requirement of solvent regeneration in addition to requiring large quantities of water [195]. An alternative approach is adsorption in which contaminants are eliminated from mixtures by solid, porous absorbents such as AC. In the case of AC, the use of low temperatures is most functional for the capture of CO2 because its removal capacity decreases as temperature increases. In addition, AC can be regenerated by varying the temperature or pressure or vacuuming [125, 196].
7. Conclusion
AC synthesis is an affordable and economically viable option, for handling large volumes of lignocellulosic waste, generated in the southeastern region of Mexico. This option would represent an alternative income for agricultural producers and agro-industries, helping to balance the trade balance of AC in the country. Certain synthesis and characterization techniques are standardized, favoring production on a commercial scale. The multiplicity of applications that AC has is a commercial market guarantee.
The need to produce Biomethane as an alternative to NG promotes the development of more efficient and economic cleaning and enrichment technologies. The use of AC as a biogas improvement technique closes a cycle of simultaneous use of residual biomass.
The positive trend in research on the use of AC in biogas upgrading is important. Although the membrane process is the most widely used industrially, would be related to the greater number of publications on this technique. However, the abundance, low cost, good yields, and capacities of lignocellulosic precursors help to increase the use of CA in biogas upgrading processes.
List of Nomenclature
%
Percent
%v/v
Percent volume on volume
~
Relative or approximate
®
Register mark
°C
Degrees centigrade
°C min−1
Degrees centigrade per minute
bar
Pressure in bar
cm−1
Wave number
cm s−1
Centimeter per second
cm3g−1
cubic centimeter per gram
GHz
Gigahertz
GWh
Gigawatt per hour
h
Hours
Ka
Acidity constant
kPa
Pressure in kilopascal
kg ha−1
Kilogram per hectare
kg air/kg fuel
Kilogram of air, per kilogram of fuel
kWh m−3
kilowatt hour per cubic meter
m2 g−1
Square meters per gram
mg g−1
Milligrams per gram
min
Minute
ml min−1
Milliliters per minute
mm
Millimeters
MMcm
Billions of cubic meters
mmol g−1
Millimole per gram
mol kg−1
Mole per kilogram
MPa
Pressure in megapascal
MJ m−3
Mega Joule per cubic meter
MJ kg−1
Mega Joule per kilogram
MW
Megawatt
nm
Nanometers
Ø
Diameter
p kWh−1
Cost per kWh
PJ
Peta Joule
pKa
Dissociation constant
ppm
Part per million
R2
Regression value
s
Second
t ha−1
Tons per hectare
t ha−1 a−1
Tons per hectare per annual
x
Multiplication symbol
Δt
Temperature delta
Acknowledgments
We appreciate the support of the professor from the University of Louisiana helping with the manuscript redaction.
Notes
Conflict-of-Interest Statement
The authors declare that there is no conflict of interest in the development of the topic and the compilation of information
Autor’s Contribution
J.S.O. (Ph D. Student). Contribution: Review bibliographic of synthesis and characterization of AC. https://orcid.org/0000-0001-6786-0521
J.R.L. (Head of pilot plant 3 - treatment of air and solid waste). Contribution: Review bibliographic of lignocellulosic waste. https://orcid.org/0000-0002-6770-5596
D.S.G. (Head of the nanotechnology laboratory). Contribution: Review characterization technical. http://orcid.org/0000-0001-9953-9142
R.H. (Professor and Department Head of Chemical Engineering). Contribution: Review application of upgrading and enrichment of biogas.
M.Z. (Professor and Department Head of Chemical Engineering). Contribution: Review application of upgrading and enrichment of biogas. https://orcid.org/0000-0002-7608-076X
A.E.E.M. (Professor) Contribution: Review of general technics and writing). https://orcid.org/0000-0003-1319-5380