AbstractTo investigate the effect of seaweed (SW) addition on anaerobic co-digestion of food waste (FW) and sewage sludge (SS), batch experiments were conducted at various substrate concentrations (2.5, 5.0, 7.5, and 10.0 g volatile solids (VS)/L) and mixing ratios ((FW or SS):SW = 100:0, 75:25, 50:50, 25:75, and 0:100 on a VS basis). The effect of SW addition on FW digestion was negligible at low substrate concentration, while it was substantial at high substrate concentrations by balancing the rate of acidogenesis and methanogenesis. At 10 g VS/L, CH4 production yield was increased from 103 to 350 mL CH4/g VS by SW addition (FW:SW = 75:25). On the other hand, SW addition to SS enhanced the digestion performance at all substrate concentrations, by providing easily biodegradable organics, which promoted the hydrolysis of SS. khyd (hydrolysis constant) value was increased from 0.19 to 0.28 d−1 by SW addition. The calculation showed that the synergistic CH4 production increment by co-digesting with SW accounted for up to 24% and 20% of total amount of CH4 production in digesting FW and SS, respectively.
1. IntroductionDue to the high content of organic matters and moisture, organic solid wastes such as food waste (FW) and sewage sludge (SS) generate huge amount of odor, leachate, and greenhouse gases during their decomposition. Thus, they should be handled through appropriate treatment methods including anaerobic digestion (AD), which was proven as an environmentally friendly technology capable of recovering bioenergy in the form of methane (CH4) [1–3]. AD is a biological process, consisting of four steps of reaction: hydrolysis, acidogenesis, acetogenesis, and methanogenesis, and the balance among each step is known to be critical for the successful digestion performance [4].
The characteristics of FW might vary depending on the region and season, but it is generally characterized to have a high C/N ratio (20 to 50) and salinity concentration (2 to 5%), and insufficient trace element for the stable long-term operation [5, 6]. Also, FW contains a huge amount of easily biodegradable carbohydrates, causing the excessive hydrolysis and acidogenesis [7]. It could result in the accumulation of organic acids with pH drop under high-strength condition [8]. To avoid this failure, anaerobic digesters fed with FW should be operated at a low organic loading rate (OLR) range, from 1 to 4 g volatile solids (VS)/L/d, or long hydraulic retention time (HRT) up to 80 d [9, 10]. On the other hand, SS has a poor hydrolysis rate due to having microbial cells and extracellular polymers substances (EPS). The EPS are composed of various substances such as protein, carbohydrates, lipids, nucleic acids, and humic acids [11]. It forms a tight extracellular matrix to prevent the penetration of hydrolytic enzymes during AD [12]. To overcome this barrier, physical, thermal, and biological pretreatment methods have often been applied to SS, but their economic benefit is still questioned [13].
Extensive researches have been carried out on co-digestion, which can overcome some problems in the mono-digestion such as lack of trace elements, imbalanced C/N ratio, and high or low biodegradability [14–16]. For example, Zhang et al. [14] reported a 40% increase in a total CH4 production yield (MPY) compared to the mono-digestion of FW, by co-digesting with cattle manure at a FW/cattle manure ratio (on a VS basis) of 2. They mentioned the supply of buffer capacity and lowering C/N ratio, as the main cause of the increased performance. Higher amount of CH4 was produced by adding microalgae to SS, which was ascribed to the fact that the easily biodegradable portion in microalgae helped the degradation of SS [17].
Since 2000, the third-generation biomass has gained a lot of attention as a bioenergy feedstock, owing to its negligible lignin content, and higher CO2 uptake rate than terrestrial biomass. In particular, seaweed (SW) does not require land for its growth, and is easier to cultivate compared to the fresh-water algae. SW, for example brown algae, is, in general, composed of 30 to 40% carbohydrates, 35 to 40% of minerals, 15% of proteins, and 5% of lipids. It has a higher content of carbohydrates than SS, and its main carbohydrate unit is cellulose while it is starch in case of FW [18, 19]. Starch and cellulose are well known polysaccharides consisting glucose units, while their binding patterns are different with each other. In starch, each glucose is bound with α-linked connection, while each glucose is bound to β-linked connection in cellulose [20]. This structural feature can lead to the big differences in the biodegradability because β-linked is stronger than α-linked.
In the present work, we hypothesized that SW has a lower anaerobic biodegradability than FW, while higher biodegradability than SS, which in turn, a certain amount of SW addition to these organic solid wastes might enhance AD performance. To investigate this, batch anaerobic co-digestion experiments were performed at various substrate concentrations (2.5, 5.0, 7.5, 10.0 g VS/L) and mixing ratios ((FW or SS):SW = 100:0, 75:25, 50:50, 25:75, 0:100 on a VS basis). The synergistic increment by co-digestion with SW was calculated, and the reasons were revealed by monitoring pH and organic acids during digestion, and by applying first-order kinetic model.
2. Materials and Methods2.1. Seeding Inoculum and FeedstockThe granular sludge taken from the anaerobic digester in a wastewater treatment plant of local beer factory in Cheongju, South Korea, was used as an inoculum after grinding less than 2 mm of diameter. Prior to seeding, it was kept at 35°C for two weeks to remove residual biogas. FW was collected from the cafeteria in Inha University, where around 1,000 people eat two times of meal every day. It was also ground with blender having less than 2 mm of diameter. Laminaria japonica, which collected at Yeosu of South Korea, was chosen as a SW, and it was washed with tap water to remove salt, and dried at 105°C for 2 h. Then, the dried SW was pulverized with blender, and diluted with distilled water. The SS used in this study was the mixture of primary sludge and secondary sludge at the same volume ratio, taken from the local wastewater treatment plant. The characteristics of seeding inoculum and feedstock are shown in Table 1.
2.2. ExperimentThe co-digestion experiments were conducted by using 280 mL serum bottle, with a working volume of 100 mL. The seeding inoculum was injected into each serum bottle at a concentration of 7.5 g VS/L. Substrate concentrations of co-substrates (1. FW and SW, and 2. SS and SW) were set to 2.5, 5.0, 7.5, and 10.0 g VS/L at the mixing ratios of 100:0, 75:25, 50:50, 25:75, and 0:100 on a VS basis. The corresponding I/S (inoculum to substrate on a VS basis) ratios were 3.0, 1.5, 1.0, and 0.8 at each substrate concentration of 2.5, 5.0, 7.5, and 10.0 g VS/L, respectively. In addition, an experiment was conducted to produce CH4 from the inoculum itself, and all results presented exclude this CH4 production. The initial pH was adjusted between 7.8 and 8.0 with 2 N KOH solution, and then sufficiently replaced with nitrogen gas (99.999%) to provide anaerobic condition. Nutrients media including buffer were not added to clarify the role of SW in the co-digestion. The fermentation was carried out in an incubator (VS-8480SRN, VISION Co., Korea) set at 35°C and at the agitation speed of 150 rpm. All experiments were conducted duplicates, and the results were averaged. Gas and liquid samples were taken every 2–5 d to analyze biogas amount, CH4 content, pH, and organic acids.
2.3. AnalysisThe concentration of total solids (TS), VS, total nitrogen (TN), alkalinity (as g CaCO3/L), and total chemical oxygen demand (COD) were measured according to standard methods [21], while carbohydrate concentration was determined by the colorimetric method [22]. pH value was measured using a pH meter (HM-3R, DDK-TOA). The amount of biogas produced was measured using a gas syringe, and was converted to standard temperature and pressure (STP). CH4 gas content was measured by taking 0.5 mL of the gas phase in serum bottle with Gas-tight syringe (HAMILTON CO., USA) and using gas chromatography (GOWMAC series 580, USA) equipped with a thermal conductivity detector. The temperature of injector, column, and detector was set to 50, 80, and 70°C, respectively. The column was a 183 cm (Length) 0.32 cm (Diameter) stainless steel column with porapak Q (80/100 mesh). Nitrogen (N2, 99.999%) gas was used as the carrier gas and the flow rate maintained at 30 mL/min. The organic acids were analyzed by HPLC (Model VP, Shimadzu Co., Japan) after filtered the supernatant in the reactor with a 0.2 μm membrane.
2.4. Synergistic EffectIn this study, the amount of CH4 production at mono-digestion of each substrate was used to determine the increased CH4 production at co-digestion. The amount of CH4 production increase by the synergistic effect was calculated by defining as follows (Eq. (1)):
where ‘Ma’ was the actual amount of CH4 production, while ‘A’ and ‘B’, were the amount of CH4 production from the mono-digestion of FW or SS, and SW, respectively. ‘C1’ and ‘C2’ indicates the mixing ratios in co-digestion of FW or SS and SW, respectively. In other words, the value of ‘((A × C1) + (B × C2))’ indicates the expected amount of CH4 production in the co-digestion of FW or SS with SW.
2.5. Kinetic Model AppliedFirst-order kinetic model has been widely applied in AD of organic matter, in particular, SS degradation [23, 24]. The amount of cumulative CH4 production in the anaerobic co-digestion of SS and SW was fitted using the first-order kinetic model as shown below (Eq. (2)):
where ‘M(t)’ is the cumulative CH4 production at time (mL CH4); ‘Fd’ is maximum CH4 production potential (mL CH4); ‘khyd’ is hydrolysis rate constant (d−1); and ‘t’ is digestion time (d).
3. Results and Discussion3.1. Comparison of BiodegradabilityThe biodegradability under anaerobic condition of individual feedstock can be determined at a low substrate concentration (2.5 g VS/L), under which both substrate and product inhibition on AD are negligible [25]. CH4 production began as soon as the digestion started, indicating that the seeding inoculum was suitable for degrading all types of feedstock (Fig. 1). The MPY increased up to 10 d, and reached 425, 344, and 169 mL CH4/g VS in cases of FW, SW, and SS, respectively. These amounts correspond to the CH4 conversion efficiency (MCE) (actual CH4 production/theoretical CH4 production, on a COD basis of 1 g COD = 350 mL CH4) of 89, 76, and 31%, respectively. This difference in the AD performance might be linked to the characteristics of main composition of individual feedstock. SS has less biodegradability than other feedstocks, since it is mainly composed of diverse microorganisms and organic and inorganic compounds clumped together in a polymeric network by EPS [13], while FW and SW have a high content of carbohydrates that are easier to degrade. However, the main carbohydrates in FW consist of starch, whereas the carbohydrates of SW (brown algae) are mostly composed of cellulose [18, 19, 26]. As previously mentioned, each glucose is bound to the next glucose by either 1,4-α or 1,6-α glycosidic bonds (α-linked) in starch, while each glucose is bound to the next glucose by 1,4-β glycosidic bonds (β-linked) in cellulose [20]. The fact that β-linked is stronger than α-linked is why AD performance of SW was less than FW. As hypothesized, the biodegradability of SW was found to be between FW and SS, which could be a suitable co-digesting material for enhanced AD.
3.2. Co-digestion of FW and SW3.2.1. Performance and synergistic effectThe amount of CH4 production, MPY, and MCE are arranged in Table 2. Biogas production was completed within 14 d, and hydrogen production was negligible during whole experimental period (Fig. S1). The MPY of FW (FW:SW = 100:0) decreased as substrate concentration increased. In particular, there was a huge drop at 10 g VS/L, exhibiting only 103 mL CH4/g VS. In cases of low substrate concentration range (2.5, 5.0 g VS/L) in FW digestion, SW addition did not show any improvement. It rather decreased the MPY, probably due to the less biodegradable characteristics of SW compared to FW. However, in cases of high substrate concentration range (7.5 and 10.0 g VS/L) of FW digestion, the MPY significantly increased, by up to 1.2 and 3.5 times at 7.5 and 10.0 g VS/L, respectively. In terms of MCE, SW addition increased MCE from 60% to 74% and 22% to 76% at 7.5 and 10.0 g VS/L, respectively. By applying Eq. (1), it was found that the ‘Synergistic CH4’ was negligible at low concentration, while it was increased at high concentration with SW addition increase. In particular, 85 mL CH4/g VS of ‘Synergistic CH4’ was produced at a substrate concentration of 10.0 g VS/L and FW:SW = 25:75, accounting for the 24% of total amount of CH4 produced. To explain the reasons for the synergistic effect, some analytical results are shown in the next chapter.
3.2.2. Reasons of synergistic effectThe change of pH during digestion and cumulative organic acids concentration are shown in Fig. 2 and Table 3, respectively. When FW was digested alone at 2.5 g VS/L, the cumulative organic acids concentration was negligible, and the pH remained high near 7.0. The SW addition showed little effect on the change of pH and organic acids accumulation at 2.5 g VS/L. On the other hand, in the mono-digestion of FW at 10.0 g VS/L, organic acids were accumulated to over 7,000 mg COD/L, and pH rapidly dropped to about 5.5, which did not recover above 6.5 until the end of digestion. This indicates that the drop of AD performance at high substrate concentrations of FW was due to the excessive production of organic acids, while the rate of methanogenesis process could not follow at high substrate concentration. Organic wastes, which contain many readily biodegradable organic matters such as FW, cause process failure due to quick hydrolysis [27, 28]. However, in case of SW mono-digestion, there seemed to be a balance between the rate of acidogenesis and methanogenesis even at 10 g VS/L. Compared to FW digestion, the cumulative organic acids concentration was lower, and pH drop was less. As mentioned earlier, this might ascribe to the difference in the biodegradable characteristics between FW and SW that they have similar anaerobic biodegradability (in terms of MCE or MPY), but different rate. In addition, although alkalinity concentration of raw SW was negligible, there might have been a production of buffering materials from SW like microalgae. In a previous work of Yun et al. [29], there was an increased concentration of alkalinity during anaerobic acidogenesis of microalgae, which was attributed to the release of buffering materials caused by cell hydrolysis. Therefore, it can be concluded that SW addition to FW under high organic loading is beneficial to gain high CH4 production with maintaining high MPY and MCE. However, other factors such as supplementing of trace elements like Ni, Co, Fe, and Se rich in SW [5, 30], and the dilution of salinity might also have contributed to the enhanced digestion performance [6, 31]. These effects can be critical in the continuous operation, but chances are low in the batch-type operation. The seeding inoculum (digester sludge) generally contains enough amounts of trace elements, which would be depleted in the long-term operation [32].
3.3. Co-digestion of SS and SW3.3.1. Performance and synergistic effectIn co-digestion of SS and SW, biogas production was completed within 8 and 14 d, and hydrogen production was negligible during whole experimental period (Fig. S2). Compared to FW and SW, the MPY of SS was lower, ranging 170–180 mL CH4/g VS (Table 4). However, the inhibition at high substrate concentration was negligible, probably indicating that initial steps (hydrolysis or acidogenesis) rather than the final step (methanogenesis) in AD were the rate-limiting step in using SS as a feedstock. Under all substrate concentrations, SW addition to SS substantially increased the digestion performance. For example, MPY and MCE at 10.0 g VS/L increased from 180 to 308 mL CH4/g VS, and from 33 to 64%, respectively. However, it could not be a surprising result since SW had a higher biodegradability than SS as seen in Fig. 1.
Therefore, it is very important to see the synergistic effect in co-digestion of SS by employing Eq. (1). It was found that the ‘Synergistic CH4’ increased with SW addition increase at all substrate concentrations. In particular, 51 mL of ‘Synergistic CH4’ was produced at 10.0 g VS/L and SS:SW = 75:25, accounting for the 20% of the total amount of CH4 produced. Assuming that MPY of SW was equal in all cases, this meant that SW addition triggered the anaerobic biodegradation of SS. To explain the reasons for the synergistic effect, some analytical results are shown in the next chapter.
3.3.2. Reasons of synergistic effectThe amount of cumulative CH4 production in the anaerobic co-digestion of SS and SW was well fitted (R2 > 0.98) using first-order kinetic model (Eq. (2)). As shown in Table 5, Fd and khyd increased with SW addition increase, at both low (2.5 g VS/L) and high concentration (10.0 g VS/L). In particular, khyd is an important indicator reflecting hydrolysis rate and substrate biodegradability [23, 24]. khyd value in the co-digestion of SS and SW at 10.0 g VS/L and the mixing ratio of SS:SW = 75:25 was increased about 37% (0.19 to 0.26) compared to AD of SS alone (10.0 g VS/L, SS:SW = 100:0). In addition, as shown in Fig. 3, further and faster pH drop with SW addition confirmed the promoting effect on the hydrolysis and acidogenesis of SS [10]. However, when SW was digested alone, it showed lower khyd value comparted to the mixed cases, probably due to the too much pH drop below 6.5. Like our study, many studies have been reported to increase the AD efficiency by improving the hydrolysis rate of SS through co-digestion of substrates with higher biodegradability than SS. For example, Zhang et al. [33] reported the increase of MPY to 82% in the co-digestion at SS:pig manure = 1:2, with khyd value increase from 0.062 to 0.094. Zhen et al. [34] also found that the addition of aquatic plant (Egeria densa) increased the MPY by 19% and khyd value from 0.175 to 0.194 as compared to digestion of SS alone. To reveal the reason of increased CH4 production by FW addition on SS digestion, Yun et al. [35] observed the increased activity of protease when FW was added. Therefore, it can be concluded that SW addition could provide easily biodegradable organics to anaerobic consortium, which promoted the hydrolysis of SS, and accordingly, enhanced the AD performance.
4. ConclusionsThe anaerobic biodegradability of SW was found to be between FW and SS, which acted as a suitable co-digesting material for enhanced AD of FW and SS. At low substrate concentration of FW digestion, SW addition showed a negligible effect on AD performance, while it synergistically enhanced the performance by balancing acidogenesis and methanogenesis at high substrate concentrations. At 10 g VS/L and FW:SW = 25:75, MCE increased from 22 to 76% by SW addition. On the other hand, SW addition to SS synergistically enhanced the digestion performance at all substrate concentrations, by providing easily biodegradable organics to anaerobic consortium, which promoted the hydrolysis of SS.
AcknowledgmentsThis work is supported by the “R&D Center for Reduction of Non-CO2 Greenhouse gases” (2017002410003) funded by Korea Ministry of Environment (MOE) as “Global Top Environment R&D Program”.
References1. Li K, Liu R, Sun C. Comparison of anaerobic digestion characteristics and kinetics of four livestock manures with different substrate concentrations. Bioresour Technol. 2015;198:133–140.
2. Xie T, Xie S, Sivakumar M, Nghiem LD. Relationship between the synergistic/antagonistic effect of anaerobic co-digestion and organic loading. Int Biodeterior Biodegrad. 2017;124:155–161.
3. Zhao J, Liu Y, Wang D, et al. Potential impact of salinity on methane production from food waste anaerobic digestion. Waste Manage. 2017a;67:308–314.
4. Feng Q, Lin Y. Integrated processes of anaerobic digestion and pyrolysis for higher bioenergy recovery from lignocellulosic biomass: A brief review. Renew Sust Energ Rev. 2017;77:1272–1287.
5. Zhang L, Jahng D. Long-term anaerobic digestion of food waste stabilized by trace elements. Waste Manage. 2012;32:1509–1515.
6. Zhao J, Liu Y, Wang D, et al. Potential impact of salinity on methane production from food waste anaerobic digestion. Waste Manage. 2017b;67:308–314.
7. Kumar G, Sivagurunathan P, Park JH, Kim SH. Anaerobic digestion of food waste to methane at various organic loading rates (OLRs) and hydraulic retention times (HRTs): Thermophilic vs. mesophilic regimes. Environ Eng Res. 2016;21:69–73.
8. Wang P, Wang HT, Qiu YQ, Ren LH, Jiang B. Microbial characteristics in anaerobic digestion process of food waste for methane production – A review. Bioresour Technol. 2017b;248:29–36.
9. Cecchi F, Traverso P, Pavan P, Bolzonella D, Innocenti L. Characteristics of the OFMSW and behavior of the anaerobic digestion processMata-Alvarez J, editorBiomethanization of the organic fraction of municipal solid waste. UK: IWA; 2003. p. 141–180.
10. Banks CJ, Chesshire M, Heaven S, Arnold R. Anaerobic digestion of source-segregated domestic food waste: Performance assessment by mass and energy balance. Bioresour Technol. 2011;102:612–620.
11. Yang Q, Luo K, Li XM, et al. Enhanced efficiency of biological excess sludge hydrolysis under anaerobic digestion by additional enzymes. Bioresour Technol. 2010;101:2924–2930.
12. Sun J, Guo L, Li Q, et al. Structural and functional properties of organic matters in extracellular polymeric substances (EPS) and dissolved organic matters (DOM) after heat pretreatment with waste sludge. Bioresour Technol. 2010;219:614–623.
13. Dhar BR, Nakhla G, Ray MB. Techno-economic evaluation of ultrasound and thermal pretreatments for enhanced anaerobic digestion of municipal waste activated sludge. Waste Manage. 2010;32:542–549.
14. Zhang C, Xiao G, Peng L, Su H, Tan T. The anaerobic co-digestion of food waste and cattle manure. Bioresour Technol. 2013;129:170–176.
15. Grosser A, Neczaj E, Singh BR, Almås ÅR, Brattebø H, Kacprzak M. Anaerobic digestion of sewage sludge with grease trap sludge and municipal solid waste as co-substrates. Environ Res. 2017;155:249–260.
16. Shin JD, Han SS, Eom KC, Sung SW, Park SW, Kim HO. Predicting methane production potential of anaerobic co-digestion of swine manure and food waste. Environ Eng Res. 2008;13:93–97.
17. Olsson J, Feng XM, Ascue J, et al. Co-digestion of cultivated microalgae and sewage sludge from municipal waste water treatment. Bioresour Technol. 2014;171:203–210.
18. Ohkouchi Y, Inoue Y. Direct production of L(+)-lactic acid from starch and food wastes using Lactobacillus manihotivorans LMG18011. Bioresour Technol. 2006;97:1554–1562.
19. Chen J, Bai J, Li H, Chang C, Fang S. Prospects for bioethanol production from macroalgae. Trend Renew Energ. 2015a;1:185–197.
20. Johnson AW. Invitation to organic chemistry. Massachusetts: Jones & Bartlett Learning; 1998.
21. APHA. Standard methods for the examination of water and wastewater 2. 2nd edWashington D.C: American Public Health Association; 2012.
22. Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colormetric method for determination of sugars and related substances. Anal Chem. 1956;28:350–356.
23. Xie S, Hai FI, Zhan X, et al. Anaerobic co-digestion: A critical review of mathematical modelling for performance optimization. Bioresour Technol. 2016;222:498–512.
24. Lee EY, Cumberbatch J, Wang M, Zhang Q. Kinetic parameter estimation model for anaerobic co-digestion of waste activated sludge and microalgae. Bioresour Technol. 2017;228:9–17.
25. El-Mashad HM, Zhang R. Biogas production from co-digestion of dairy manure and food waste. Bioresour Technol. 2010;101:4021–4048.
26. Chen H, Zhou D, Luo G, Zhang S, Chen J. Macroalgae for biofuels production: Progress and perspectives. Renew Sust Energ Rev. 2015b;47:427–437.
27. Haider MR, Zeshan , Yousaf S, Malik RN, Visvanathan C. Effect of mixing ratio of food waste and rice husk co-digestion and substrate to inoculum ratio on biogas production. Bioresour Technol. 2015;190:451–457.
28. Li Q, Li H, Wang G, Wang X. Effects of loading rate and temperature on anaerobic co-digestion of food waste and waste activated sludge in a high frequency feeding system, looking in particular at stability and efficiency. Bioresour Technol. 2017;237:231–239.
29. Yun YM, Jung KW, Kim DH, Oh YK, Shin HS. Microalgal biomass as a feedstock for bio-hydrogen production. Int J Hydrogen Energ. 2012;37:15533–15539.
30. Karlsson A, Einarsson P, Schnurer A, Sundberg C, Ejlertsson J, Svensson BH. Impact of trace element addition on degradation efficiency of volatile fatty acids, oleic acid and phenyl acetate and on microbial populations in a biogas digester. J Biosci Bioeng. 2012;114:446–452.
31. Drennan MF, DiStefano TD. High solids co-digestion of food and landscape waste and the potential for ammonia toxicity. Waste Manage. 2014;34:1289–1298.
32. Kim DH, Kim SH, Kim HW, Kim MS, Shin HS. Sewage sludge addition to food waste synergistically enhances hydrogen fermentation performance. Bioresour Technol. 2011;102:8501–8506.
33. Zhang W, Wei Q, Wu S, et al. Batch anaerobic co-digestion of pig manure with dewatered sewage sludge under mesophilic conditions. Appl Energ. 2014;128:175–183.
Fig. 1Cumulative CH4 production yield in the anaerobic digestion of food waste (FW), sewage sludge (SS), and seaweed (SW) at 2.5 g VS/L. 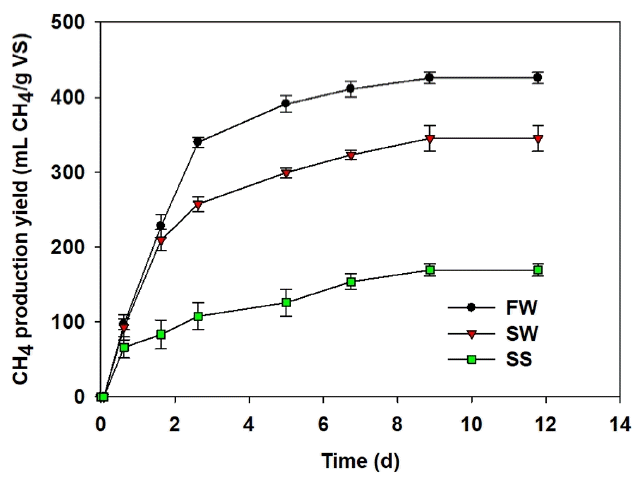
Fig. 2The variation of pH in anaerobic co-digestion of food waste (FW) and seaweed (SW) at (a) 2.5 g VS/L, and (b) 10.0 g VS/L. 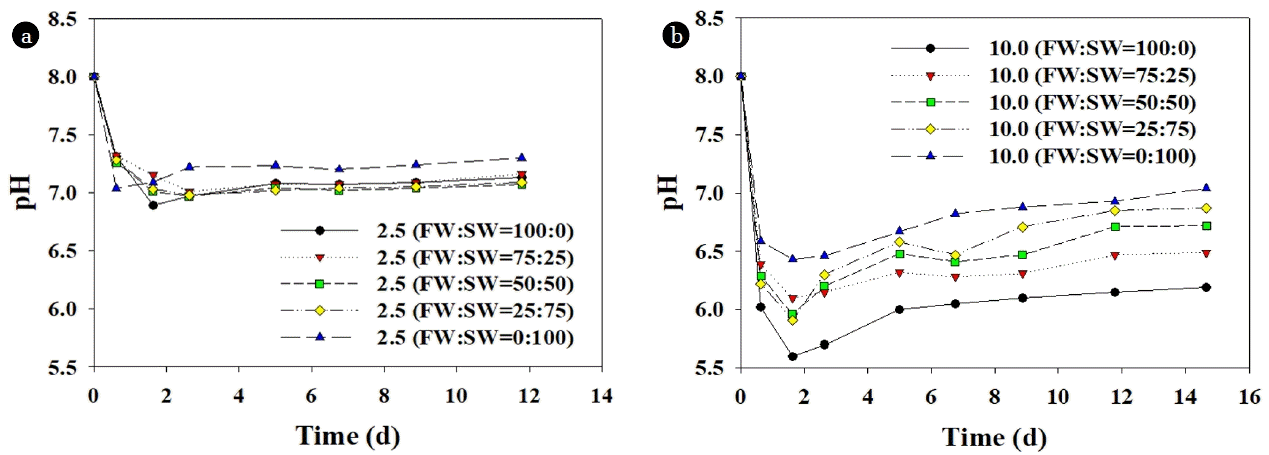
Fig. 3The variation of pH in anaerobic co-digestion of sewage sludge (SS) and seaweed (SW) at (a) 2.5 g VS/L, and (b) 10.0 g VS/L. 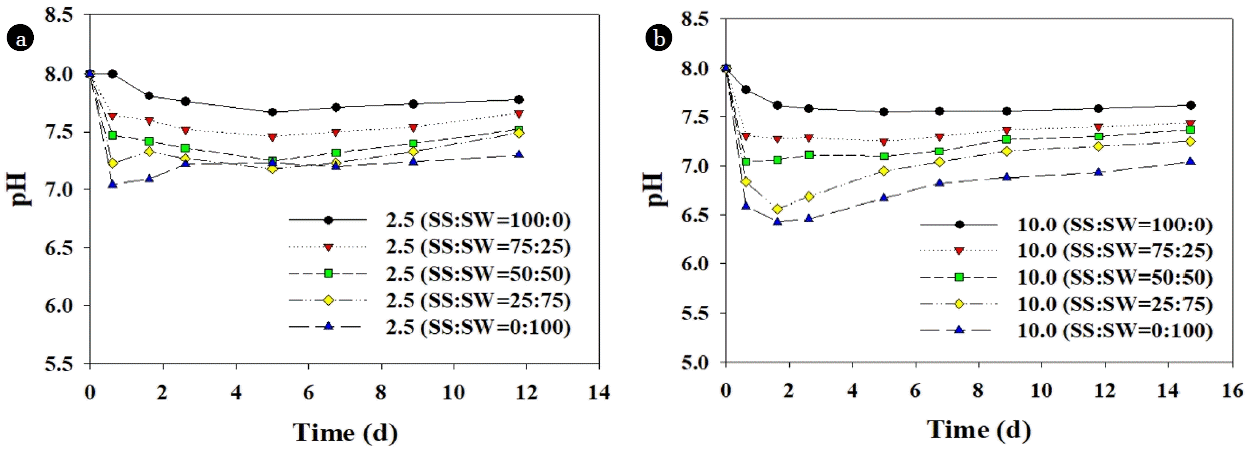
Table 1Characteristics of Seeding Sludge, Food Waste, Sewage Sludge, and Seaweed
Table 2Batch Performances in the Anaerobic Co-digestion of Food Waste (FW) and Seaweed (SW) at various substrate Concentrations and Mixing Ratios
Table 3Cumulative Organic Acids Concentration in the Broth in Co-digestion of Food Waste (FW) and Seaweed (SW) at 2.5 and 10.0 g VS/L (Samples were taken after 12 d and 14 d of operation.)
Table 4Performances in the Anaerobic Co-digestion of Sewage Sludge (SS) and Seaweed (SW) at Various Substrate Concentrations and Mixing Ratios
Table 5Estimated Parameters Obtained through the First-order Kinetic Model
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||