1. Introduction
Diesel is a product of crude oil distillation [1] and contains rich light-weight hydrocarbons with the range of C8–C26 and polyaromatic hydrocarbons (PAHs) [2]. Diesel was reported as sea-water and soil contaminant [3]. The presence of diesel in sea-water and soil will cause serious effects to marine ecosystem [4] because the components of diesel are potentially carcinogenic and toxic for the environment [2]. Recent research reported that the biological process is the best practice for removing diesel spills in sea-water and soil. Some bacteria isolated from a diesel-contaminated area have the ability to produce enzymes to degrade and utilize diesel as carbon and energy source [5]. Vibrio alginolyticus (V. alginolyticus) is one bacterium isolated from diesel-contaminated sea-water. Isiodu et al. [6] reported that V. alginolyticus G19 could be isolated from crude oil contaminated brackish water and have the ability to degrade diesel with consortium bacteria up to 90%. Graziano et al. [7] also reported that Vibrio sp. PBN295 isolated from sea-water has the potential to produce natural biosurfactants. Currently, available information regarding biodegradation of diesel by V. alginolyticus is limited thus the research about this is very necessary to increase our knowledge.
Bioremediation processes are significantly affected by capabilities of microorganisms to overcome the bioavailability limitations in multiphase environmental scenarios and environmental factors, such as temperature, pH, nutrients and electron acceptor availability [3]. According to Sihag et al. [8], the pH optimum for diesel degradation by microorganisms is at range of pH 6.5 to 8. Palanisamy et al. [3] also reported that Acinetobacter baumannii could degrade diesel up to 99% with a pH of 7. Furthermore, temperature is also affected by diesel degradation. The temperature optimum for diesel degradation is in the mesothermic range of 20°C to 35°C [8].
The addition of surfactants on diesel biodegradation process can increase the efficiency of diesel degradation by bacterial isolates [3] because surfactant could increase the solubility of hydrophobic compounds in water [9]. Moreover, surfactants could increase the bioavailability of diesel on polluted medium [10]; thus, bacteria can utilize diesel as a carbon and energy source. Tween 80 is one of synthetic surfactants to be used on diesel degradation. The addition of Tween 80 on diesel degradation process can increase the efficiency of diesel degradation up to 90% for 15 d [9]. Palanisamy et al. [3] also reported that the addition of Tween 80 can increase the rate of bacterial growth in a diesel-polluted medium with efficiency of diesel degradation achieved at 99.9% for 5 d.
The bioavailability of macronutrient nitrogen (N) and phosphorus (P) on hydrocarbon-contaminated medium may affect the bioremediation process. According to Xia et al. [11], the addition of nitrogen and phosphorus on 15 mg/L of diesel contaminated media with 10 of N/P ratio can degrade diesel up to 75.8%, while a 20 of N/P ratio achieved 52.4%. Zahed et al. [12] also showed that an additional 10 of N/P ratio can degrade hydrocarbon compounds up to 95% with 100 mg/L of initial diesel concentration. Ghaly et al. [13] reported that the optimum N/P ratio on the diesel degradation process ranged from 2.4 to 10. Xia et al. [11] said that the addition of N and P as macronutrients can increase the percentage of diesel degradation in sea-water.
The salinity level can also affect the bioremediation process. Kee et al. [14] reported that the degradation of hydrocarbon compounds with more than 30‰ of salinity would run ineffectively. Mukherji and Vijay [15] also reported that the optimum salinity on hydrocarbon degradation process between 5‰ and 35‰. According to Kee et al. [14], at the salinity level between 10‰ and 30‰, microorganisms can degrade hydrocarbon compounds more than 50%, whereas at more than 30‰ of salinity the percentage of diesel degradation becomes less than 30%.
The statistical technique has been successfully applied in many fields [16]. The statistical experiment designs most widely used in optimization experiments were response surface designs. The designs provide information about direct effects, pairwise interaction effects and curvilinear variable effects. However, information on the optimization of biostimulation factors required for the enhanced biodegradation of diesel is limited.
This study was designed to optimize the diesel biodegradation using V. alginolyticus using response surface methodology (RSM). The RSM was conducted based on full factorial Box-Behnken design (BBD) with three factors, i.e. Tween 80 concentration, N/P ratio and salinity level. The selection of those factors was carried out based on biostimulation technique for biodegradation.
2. Materials and Methods
2.1. Preparation of Bacteria
V. alginolyticus was isolated from a ship dismantling facility along the Tanjung Jati coast, Madura, Indonesia, and it was conducted based on our previous study [17]. Pure culture was maintained on nutrient agar slants at 4°C. The re-growth of the bacteria was conducted on NA in an incubator with a temperature of 37°C for 24 h and after that, the bacteria were transferred to an NB in a sterile Erlenmeyer flask. The bacteria were shaken vigorously in a shaker (Innova 2000, Eppendorf, Germany) at 150 rpm for 8 h. After that, the bacteria were harvested and ready to be used for diesel biodegradation studies. The diesel degradation studies were performed in a mineral salt medium (MSM) that contained diesel oil as the sole source of carbon. The MSM consisted of (NH4)2SO4 - 1 g/L; KH2PO4 - 1 g/L; K2HPO4 - 1 g/L; MgSO4.7H2O - 0.2 g/L; FeCl3 - 0.05 g/L; and CaCl2 - 0.02 g/L [2].
2.2. Diesel Biodegradation Studies
The biodegradation assay was carried out in a 250 mL Erlenmeyer flask (Pyrex, Germany) containing 100 mL of MSM and diesel as the carbon and energy source with bacteria. Ten per cent (v/v) of inoculum bacteria (OD600 = 1) were added into the MSM containing diesel. The diesel in the flask was amended with different amounts of Tween 80, N/P ratio and salinity level (Table 1), respectively. The diesel cultures were incubated for 14 d with agitation of 150 rpm on a rotary shaker (Memmert, Germany). The collected samples were extracted and analysed to check the total petroleum hydrocarbon degradation by gas chromatography. Assay was carried out in duplicate.
2.3. Analysis of Diesel Biodegradation
About 5 mL of sample from the biodegradation assay reactor was extracted twice with 20 mL of n-hexane (Fulltime, USA) as a solvent using separating funnels (Pyrex, Germany). Steps were conducted to remove cellular material [3]. Afterwards, the diesel residues were transferred into a vial and incubated in a water bath (Memmert, Germany) at 55°C for 3 d to remove n-hexane.
The diesel residues were analysed with a GCMS-QP2010S instrument (Shimadzu, Japan). A capillary column Rxi-5Sil MS (30 m × 0.25 mm × 0.25 μm) was used to separate hydrocarbons. About 0.8 μL of sample was injected into the GCMS. Helium was used as the carrier gas. The injector and detector temperatures were maintained at 310°C. The oven was programmed at an initial temperature of 50°C, which was held for 5 min. The temperature then was ramped at 5°C/min to 300°C and held for 15 min. The electron impact ionization was tuned at 70 eV. The degradation rate of diesel oil was determined according to the following Eq. (1):
where Rd, Ac and As are the degradation rate of diesel oil, the amount of diesel oil remaining in the control culture and the amount of diesel oil remaining in the sample culture [18].
2.4. Experiment Design and Data Analysis
The Box-Behnken factorial experimental design employed had three independent variables: Tween 80, N/P ratio and salinity level. Each independent variable was studied at three levels, with 17 experimental runs and one control (Table 2). The levels were selected based on the literature. Efficiency of diesel degradation for 14 d was considered an experimental response. Table 2 shows the coded and actual values of factors and levels used in the experimental design. Diesel contaminated water without biostimulation was also assayed as a control. The statistical software Design-Expert 6.0.8 (Stat-Ease Inc., Minneapolis, Minnesota, USA) was used to evaluate the analysis of variance (p < 0.05), determine the significance of each term in the fitted equations and estimate the goodness of fit in each case.
3. Results and Discussion
3.1. Diesel Biodegradation Studies
After performing 17 runs of the BBD and one control, the results of the statistical experiment were analysed with regard to the coded design matrix. The regression equation showed that the percentage of diesel biodegradation was an experimental function of test variables in coded units. Table 3 depicted that diesel contents had decreased in all reactors at 14 d. In control, the percentage of diesel biodegradation in natural biodegradation reached only 0.93%. It was indicated that the biological process of natural attenuation that may occur on the control by chemical reaction and evaporation. The addition of biostimulant on diesel contaminated medium will increase the efficiency of diesel biodegradation. The degradation of diesel contents in reactors containing biostimulants was much higher (Table 3) in the same period. These results indicated that the addition of biostimulant can increase the percentages of diesel biodegradation. Agarry and Ogunleye [16] and Olawale et al. [1] have reported similar observations. It showed that an increasing of diesel biodegradation with the addition of biostimulants such as nitrogen and phosphorus as macronutrients for bacterial growth.
The effect of different concentrations of Tween 80 was determined at same condition of N/P ratio and salinity level (number run of 1 and 15, number run of 3 and 9). The result showed that the addition of surfactant can enhance diesel degradation. The surfactants are used to decrease surface tension between water and diesel molecules [19]. In water-in-diesel emulsion, the polar group orients toward the water and the non-polar group orients toward the diesel and lowers the interfacial tension between diesel and water phases [20]. Similar to Agarry and Ogunleye [16], Palanisamy et al. [3], and Olawale et al. [1] showed that the addition of non-ionic surfactant for diesel biodegradation can increase diesel degradation. Moreover, V. alginolyticus can produce biosurfactants [21]. It was supported by Graziano et al. [7] where Vibrio sp. PBN295 has the potential to produce natural biosurfactants.
The effects of N/P ratio were shown in number run of 3 and 13, number run of 6 and 16. It showed the similar biodegradation conditions with different of N/P ratio. These results indicated that the higher N/P ratio extensively increased diesel biodegradation from 92.85% to 94.22% and from 85.24% to 88.31%. Similar to Agarry and Ogunleye [16], Palanisamy et al. [3] and Olawale et al. [1] showed that the addition of nitrogen and phosphorus in the biodegradation process can increase the rate and efficiency of biodegradation. Based on Borah and Yadav [22], the addition of nitrogen on the biodegradation process was used for the synthesis of amino acids and nucleic acids for rapid growth of microorganisms, whereas phosphorus was used for the synthesis of ATP and DNA. Xia et al. [11] also reported that the addition of N and P as macronutrients can increase the percentage of diesel degradation in sea-water.
Salinity levels can also affect the diesel degradation process. The number run of 1 and 5, number run of 6 and 11 had the same remediation conditions with different salinity levels. The removal of diesel on number run of 1 and 5, number run of 6 and 11 increased from 92.23% to 94.93% and 88.31% to 90.56%, respectively. It was shown that V. alginolyticus can degrade diesel in high salinity condition. Surendran et al. [23] also reported that V. alginolyticus was a halophilic bacterium that could live on media with high salinity about 15–35‰. Hu et al. [21] similarly reported that V. alginolyticus can produce biosurfactants with an optimum salinity of 20‰.
The gas chromatogram of diesel substrate retrieved from the inoculated medium on R5 after 14 d incubation is shown in Fig. 1. Fig. 1(a) showed that the diesel components contained hydrocarbons with range of C6–C44 and PAHs such as benzene and naphthalene. Ramasamy [2] reported that the diesel contains rich hydrocarbons ranged C8–C26 and PAHs. Bhuvaneswar et al. [24] also reported that the diesel contains a large amount of alkanes hydrocarbon chains from C10–C20. Hydrocarbons differ in their susceptibility to microbial attack and generally degrade in the following order of decreasing susceptibility: n-alkanes > branched alkanes > low molecular weight aromatics > cyclicalkanes > polyaromatic hydrocarbons [25]. Although the petroleum hydrocarbons compounds can be relatively easily degraded in a low salinity environment [25], little is known about their fate in moderate to high salinity conditions.
It was found that V. alginolyticus almost degraded hydrocarbons as shown in Fig. 1(b). Based on Fig. 1(b), the remaining hydrocarbons after 14 d incubation were α-methylphenethylamine, ethanol, acetone, pentane, methylcyclopentane and cyclohexane. It was indicated that V. alginolyticus could utilize diesel as a carbon source for metabolism and also convert it to CO2, H2O and a few intermediates compounds. Treated diesel substrate showed a decrease in the area of major peaks, indicating a breakdown of the main compounds; while new peaks leading to formation of short-chain compounds represented breakdown products or presumed metabolites [2].
The degradation pathways and enzymes involved in the aerobic metabolism of petroleum compounds for many non-halophiles. However, very little information exists about the pathway and enzymes involved in the biodegradation of hydrocarbons in high salinity condition. In non-halophiles, monooxygenases and dioxygenases initiate degradation of a wide variety of hydrocarbons under aerobic condition by the addition of oxygen atom to the terminal or subterminal carbon and converting them to a few central intermediates, such as catechols and protocatechuate, through convergent pathways. These intermediates are cleaved by ortho- and meta- cleavage dioxygenases into the Krebs cycle via beta-oxidation. The primary enzymes involved in the ortho-and meta- cleavage activity include catechol 1,2-dioxygenase and catechol 2,3-dioxygenase. The genes encoding these enzymes have been characterized for a variety of aerobic microorganisms, including several members of the genera Pesudomonas, Rhodococcus, Ralstonia and Mycobacterium, Acinetobacter [26, 27].
A few recent studies have shown that the degradation of hydrocarbons at high salinity condition occurs using enzymes described for many non-halophiles [25]. Dalvi et al. [28] have analysed the draft genome sequence of the extremely halophilic benzene and toluene degrading Arhodomonas sp. strain Seminole. These proteins share 44–77% sequence identity with proteins previously described in non-halophilic organisms. The results indicate that benzene is converted to phenol and then to catechol in two steps by monooxygenase-like enzymes closely related to phenol hydroxylases. Thus, formed catechol undergoes ring cleavage via the meta pathway by 2,3-CAT to form 2-hydroxymuconic semialdehyde, which subsequently enters the tricarboxylic acid cycle. To corroborate these predicted enzymes that benzene is converted first to phenol and then to catechol prior to ring cleavage by 2,3-CAT, Dalvi et al. [28] also grew a closely related species Arhodomonas sp. strain. Rozelon deuterated benzene and deuterated phenol was detected by GC-MS as the initial intermediate of benzene degradation. A 2-D gel electrophoresis and tandem mass spectrometry has identified the phenol hydroxylase-like and 2,3-CAT in the cell extract of strain Rozel grown on benzene as the sole carbon source.
3.2. Polynomial Regression Model and Statistical Analysis
The experimental data were fitted to a second order polynomial regression model containing 3 linear, 3 quadratic and 3 interaction terms using the same experimental design software to derive the regression equation for diesel removal as stated in Eq. (2):
where β0 is the value of the fixed response at the centre point of the design; β1, β2, β3 are linear coefficients; β12, β13, β23 are quadratic coefficients and the interaction effect coefficients regression terms, respectively; A, B and C are the levels of independent variables.
The significance of each coefficient in the equation was determined by F-test and P-values. F-test showed that all the factors and interactions considered in the experimental design were statistically significant i.e. p < 0.05, at 95% confidence level. The regression equation obtained after analysis of variance gave the level of diesel biodegradation as a function of the different variables: Tween 80, N/P ratio and salinity level. All terms regardless of their significance are included in the following Eq. (3):
where A is Tween 80 concentration, B is N/P ratio and C is salinity level. The regression equation and determination coefficient (R2) were determined to test the fit of the model. The model F-value of 6,268.85 infers the model was significant. The low probability value (< 0.0001) indicated that the model was significant. Coefficient determination (R2 = 0.9936) are a measure of goodness of fit to the model indicated a high degree of correlation between the observed value and predicted values. The determination coefficient suggested that more than 99.36% of the variance is attributable to the variables and indicated a high significance of the model. Thus, 0.64% of the total variance cannot be explained by the model.
The fitted model was considered adequate if the F-test is significant (p < 0.05). The Analysis of Variance (ANOVA), as shown in Table 4, demonstrated that the model was highly significant, as was apparent from the very low probability (p < 0.0001) of the F-test and insignificant result from the Lack of Fit model (p = 0.0713). The Lack of Fit test was implemented by comparing the variability of the current model residuals to the variability between observations at replicate settings of the factors. The Lack of Fit F-value of 5.26 implied the Lack of Fit is not significant relative to the pure error. There was a 7.13% chance that a Lack of Fit F-value this large could occur due to noise.
The Lack of Fit was designed to determine whether the selected model was adequate to describe the observed data or whether a more complicated model should be used. The predicted R2 value of 0.9164 was in reasonable agreement with the adjusted R2 value of 0.9854. Adequate precision measures the signal-to-noise ratio. A ratio > 4 was desirable. The ratio of 29.041 obtained in this research indicated an adequate signal. This model can be used to navigate the design space.
The coefficient of variation (CV) as the ratio of the standard error of estimate to the mean value of the observed response is a measure of reproducibility of the model. Generally a model can be considered reasonably reproducible if its CV is not greater than 10%. Hence, the low variation coefficient value (CV = 2.98%) obtained indicated a high precision and reliability of the experiments. The coefficient of the model and the corresponding P-values are presented in Table 5. The significance of regression coefficients was considered, ignoring those with an insignificant effect on the response at a significance level of 95 per cent. The P-values of the regression coefficients suggest that among the test variables, linear, quadratic and interaction effects of Tween 80, N/P ratio and salinity level are highly significant. The insignificant effects (factors and interactions) with P-values higher than 0.05 were ignored. In this study, A, B, C, A2, B2, C2, AB, AC and BC are significant model terms.
Thus, statistical analysis of all the experimental data showed that Tween 80, N/P ratio, and salinity level had a significant effect on diesel degradation during the study. Moreover, it was observed that Tween 80 exerted more pronounced linear effect (higher coefficient values) on diesel biodegradation. Diesel biodegradation was mostly and positively influenced by Tween 80 concentration followed by salinity level and N/P ratio.
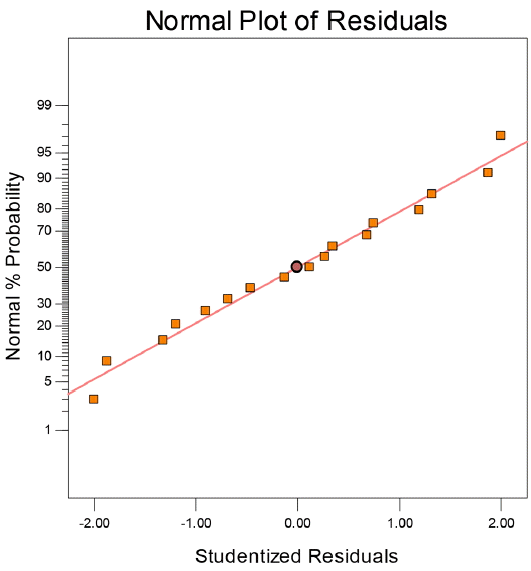
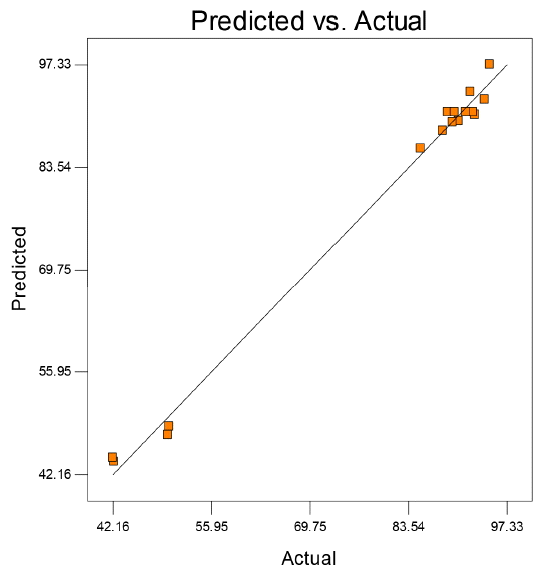
Fig. 2 showed the studentized residuals and normal percentage probability plot. Residual showed the difference between the observed value of a response measurement and the value that is fitted under the theorized model. Small residual values indicated that model prediction was accurate. The predicted versus actual plot of diesel oil biodegradation were shown in Fig. 3. Actual values were determined for a particular run and the predicted values were calculated from the approximating function used for the model.
3.3. Interaction among Factors That Influence Diesel Biodegradation
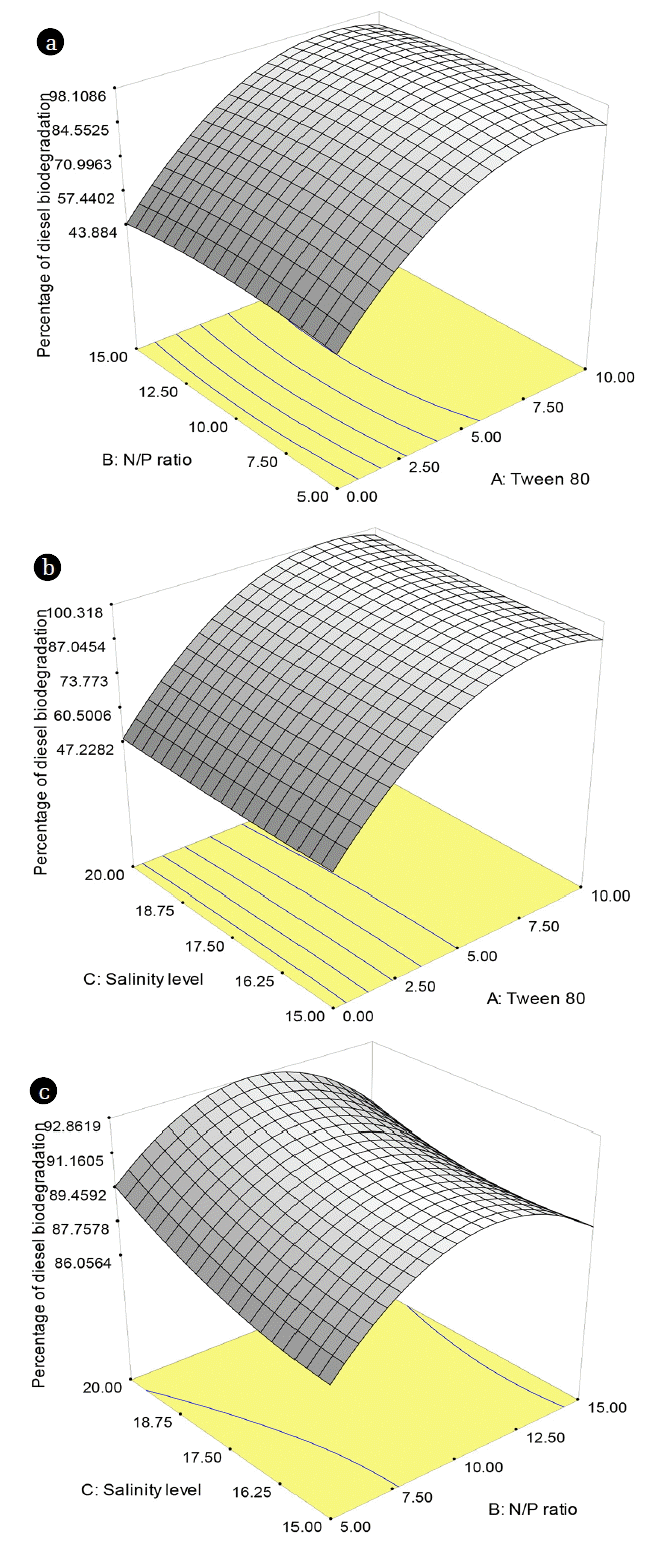
The interaction between factors affecting diesel biodegradation process showed in Fig. 4 can assist in visualizing the additional effect of Tween 80, N/P ratio, and salinity level to percentage of diesel degradation. The interaction between Tween 80 and N/P ratio with 17.5‰ of salinity depicted in Fig. 4(a). According to Fig. 4(a), the higher percentage of diesel degradation by V. alginolyticus was attained with increased in Tween 80 concentration and N/P ratio. At salinity of 17.5‰, the highest percentage of diesel degradation was 94.22% with 10 mg/L of Tween 80 concentration and 15 of N/P ratio. Sodium chloride was used to for the ion exchange and osmotic processes on metabolisms of the microorganism. However, high salinity can also cause bacterial cells to be lysis. It was shown that V. alginolyticus can degrade diesel in high salinity condition. Hu et al. [21] reported that V. alginolyticus can produce biosurfactants with 20‰ of optimum salinity. In addition, Graziano et al. [7] reported that Vibrio sp. PBN295 isolated from sea-water has the potential to produce natural biosurfactants.
Fig. 4(b) showed the interaction between Tween 80 and salinity level with 10 of N/P ratio. Based on Fig. 4(b) showed that the higher concentration of Tween 80 and salinity level, the efficiency of diesel degradation was also relatively higher. The highest percentage of diesel degradation influenced by Tween 80 concentration and salinity level was 94.93% with 10 mg/L of Tween 80 concentration, 10 of N/P ratio, and 20‰ of salinity. Xia et al. [11] reported that the addition 10 and 20 of N/P ratio on biodegradation process can degrade diesel up to 75.8% and 52.4%, respectively with 15 mg/L of initial diesel concentration. Zahed et al. [12] also reported that the addition 10 of N/P ratio on biodegradation process can degrade hydrocarbon compounds up to 95% with 100 mg/L of initial concentration. The addition of nitrogen to the biodegradation process was used for synthesis of amino acids and nucleic acids for rapid growth of microorganisms, whereas phosphorus was used for synthesis of ATP and DNA.
Fig. 4(c) showed the interaction between N/P ratio and salinity level with 5 mg/L of Tween 80 concentration. Based on Fig. 4(c), the higher N/P ratio and salinity level, the efficiency of diesel degradation was also relatively high. However, more than 10 of N/P ratio, the efficiency of diesel degradation was tend to decrease. Agarry and Ogunleye [16] said that excessive nutrient concentrations can lead to eutrophication process in the aquatic environment. At 5 mg/L of Tween 80 concentrations, the highest percentage of diesel degradation was 91.57% with 10 of the N/P ratio and 17.5‰ of salinity. This predicted due to better bioavailability of substrate for the intrinsic microorganisms [16]. Agarry and Ogunleye [16] also reported that the addition 10 mg/L of surfactant can degrade diesel with efficiency more than 80%. However, the maximum concentration of Tween 80 is 10 mg/L. Based on Li and Chen [29] showed that at 200 mg/L of surfactant, the efficiency of diesel degradation was reached 80%, while at 1,000 mg/L of surfactant, the percentage of diesel degradation was less than 30%. At high surfactant concentrations can kill bacterial cells. It was caused that the surfactant molecule can dissolve the lipid contained in the cell membrane. In addition, it can cause lysis of cells due to the cell membranes structural were changed [29].
3.4. Optimization of Diesel Biodegradation
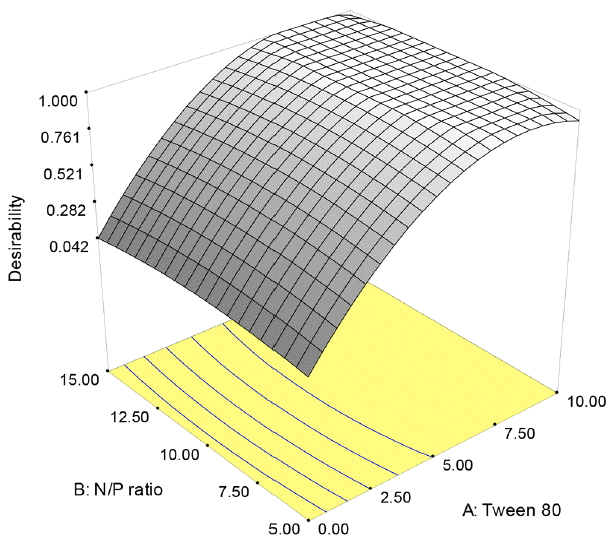
The numerical optimization technique based on the desirability function was carried out to determine the optimum conditions for diesel biodegradation by V. alginolyticus. To determine the optimum conditions in the BBD, the factors (Tween 80, N/P ratio, and salinity level) were set in range, while the percentage of diesel biodegradation was set on maximize. The predicted optimum values of Tween 80 concentration, N/P ratio and salinity level based on the model were obtained 9.33 mg/L, 9.04 and 19.47‰, respectively, to achieve 98.20% of diesel degradation while the desirability value was 1.00 for the experiment, as can be seen in Fig. 5.
Agarry and Ogunleye [16] similarly reported that optimization of diesel biodegradation process on soil by bacteria with BBD. The result showed that the optimum value of NPK, Tween 80, and H2O2 to achieve 83,79% of diesel degradation was obtained 4.25 gram, 10.22 mg/L and 0.46 g/L, respectively, while desirability value was 1.00 for the experiment. Zahed et al. [12] also used RSM for optimization of diesel biodegradation process. The result showed that the efficiency of diesel degradation achieved 94.9% while the optimum value of N/P ratio was 10 and the desirability value was 1.00 for experiment. Based on the description above, it can be concluded that RSM method with BBD can be used for diesel biodegradation process by isolated bacteria.
4. Conclusions
This study was designed to determine the diesel biodegradation and its optimization using RSM. The results indicated that biostimulation on diesel biodegradation by V. alginolyticus resulted in the enhancement of diesel degradation. RSM with BBD is a reliable and powerful tool for modelling and optimizing diesel biodegradation processes. The second order quadratic regression model for diesel degradation by V. alginolyticus with a coefficient of determination (R2 = 0.9936) was obtained with a statistically significant (p < 0.0001). The predicted optimum values of Tween 80 concentration, N/P ratio and salinity level based on the model obtained 9.33 mg/L, 9.04 and 19.47‰, respectively, to achieve 98.20% of diesel degradation.