1. Introduction
Uses of petroleum-based plastics have contributed to environmental problems such as production of non-biodegradable plastic wastes, exhaustion of limited petroleum resources, and emission of CO2 [1–5], and this triggered efforts for production of bio-based biodegradable plastics, in particular, polyhydroxyalkanoates (PHA) [3, 6]. PHA can be accumulated in bacterial cells under unbalanced nutrient conditions, for example, carbon-excess and nitrogen- or phosphorus-deficient conditions [2, 3, 7]. PHA exhibiting biodegradability and, in some cases, biocompatibility have a large potential for replacing existing petroleum-based plastics [2, 3]. However, bacterial PHA synthesis is often limited due to a relatively high production cost compared to petroleum-based plastics. In order to reduce the production cost, cheaper carbon sources have been used as raw materials, which almost take approximately 50% of the production cost [5, 6, 8], such as waste-waters, agricultural wastes, food wastes, and waste vegetable oils [3, 9–14]. Among agricultural wastes, rice straw, tequila bagasse, rice bran and starch have been studied as carbon sources for PHA production [8, 15–19].
Previous studies on bacterial PHA accumulation showed that bacterial accumulation of PHA is largely affected by nutrient conditions (e.g., carbon-to-nitrogen (C/N) ratios) [2, 3, 7]. For example, accumulation of poly-3-hydroxybutyrate (P(3HB)) in Bacillus megaterium was optimal (~70%) at the initial C/N ratio of 15:1 with sucrose as a C source and ammonium sulfate ((NH4)2SO4) as a N source [20]. Also, PHA accumulation in Ralstonia eutropha was maximal (~70%) at the initial C/N ratio of 80:1 with acetic acid, propionic acid, or butyric acid as a C source and ammonium chloride (NH4Cl) as a N source [21]. When activated sludge bacteria were used for PHA accumulation using glucose or fructose as a C source and (NH4)2SO4 as a N source, the maximum PHA content of 42% was achieved at the initial C/N ratio of 140:1 among the studied range of C/N ratios (20:1 to 140:1) [22]. These previous studies mainly used a single C source and a single N source to study the effect of the initial C/N ratio on PHA accumulation and either the maximum PHA content or the maximum PHA concentration that can be obtained at a particular C/N ratio has been reported. However, depending on the initial C/N ratios, the maximum PHA content and the maximum PHA concentration could be different and the changes in the PHA accumulation patterns could be different. But such aspects of bacterial PHA accumulation were not the main focus of the previous studies. When the changes in PHA accumulation pattern with time at different C/N ratios are better understood, more efficient bacterial PHA accumulation process can be designed depending on the initial C/N ratios of the organic wastes used as C and N sources.
Furthermore, since adjusting the C/N ratio of a given organic waste may not be practically economic, PHA accumulation could be enhanced by designing the PHA accumulation process based on the PHA accumulation pattern. However, previous studies are limited to reporting the maximum PHA production from a given organic waste that may contain both C and N sources [14, 16, 17]. For example, 32 g/L PHA or 70% PHA content could be obtained from food wastes using Alcaligenes latus DSM 1124 [14]. Also, 78 g/L PHA or 56% PHA content could be achieved using Haloferax mediterranei and rice bran, while 24 g/L PHA or 39% PHA content could be obtained using cornstarch [16]. Using hydrolyzed whey permeate, 5.5 g/L PHA could be obtained [17]. Thus, there is a need for studying the effect of various initial C/N ratios on the PHA accumulation pattern.
Rice straw is the most generated agricultural waste in South Korea and East Asia, thus, it is an attractive resource as raw materials for PHA production [23]. When rice straw is used for PHA accumulation, rice straw may be delignified to produce liquid hydrolysates, which contain C sources that can be easily utilized by PHA-accumulating bacteria. Rice straw used in this study consists of carbon (39 wt%), hydrogen (4.8 wt%), nitrogen (1.6 wt%), sulfur (0.67 wt%), and oxygen (54 wt%), suggesting the presence of both C and N sources in the liquid hydrolysates [24]. Therefore, this study used model rice straw hydrolysates prepared using glucose as a C source and NH4Cl as a N source to simulate rice straw hydrolysates having various initial C/N ratios, and investigated the effect of the initial C/N ratios on PHA accumulation patterns. The results can then be used to prepare strategies for PHA accumulation using rice straw hydrolysate.
2. Materials and Methods
2.1. Bacterial Culture Conditions
Cupriavidus necator (ATCC 17697T) was obtained from the DSMZ. Two-stage sequential batch tests consisting of a growth stage and an accumulation stage were conducted to produce PHA. During the growth stage, C. necator was grown in a nutrient broth (8 g/L) for 24 h at 180 rpm and 30°C. After 24 h, cells centrifuged at 7,000 g for 5 min were resuspended (O.D. of 1.6 at 600 nm) in a mineral medium containing (in g/L) KH2PO4 2.3, Na2HPO4 2.3, MgSO4×7 H2O 0.5, NaHCO3 0.5, CaCl2×2 H2O 2.3, Fe(NH4) 2.3, and 5 mL of a trace element solution ((in g/L) ZnSO4×7 H2O 0.1, MnCl2×4 H2O 0.03, H3BO3 0.30, CoCl2×6 H2O 0.20, CuCl2×2 H2O 0.01, NiCl2×6 H2O 0.02, Na2MoO4×2 H2O 0.03). Different C/N ratio conditions (i.e., different degrees N-deficient conditions and N-free condition) were prepared by adjusting the amount of glucose and NH4Cl in the media. For different degrees of N-deficient conditions, media with C/N ratio of 3.6:1 with 2 g/L glucose and 1 g/L NH4Cl, C/N ratio of 36:1 with 2 g/L glucose and 0.1 g/L NH4Cl, C/N ratio of 360:1 with 2 g/L glucose and 0.01 g/L NH4Cl, and C/N ratio of 36:1 with 20 g/L glucose and 1 g/L NH4Cl were prepared. For N-free conditions, only 2 g/L glucose without NH4Cl and only 20 g/L glucose without NH4Cl were used to prepare media. The PHA accumulation experiments were conducted in triplicate.
2.2. Sampling and PHA Extraction
During the PHA accumulation stage, samples were taken at predetermined times (0, 6, 12, 24, 48, 72, and 96 h). At each sampling time, 8 mL culture was centrifuged at 7,000 g for 5 min and, after discarding supernatant, cell pellets were dried at 60°C for 24 h to measure dried cell weight (DCW). The dried cell pellets were subject to simultaneous PHA extraction and butanolysis in a mixture of butanol (1.5 mL) and 37% HCl (0.5 mL) at 100°C for 8 h in an oil bath. The butanolyzed PHA were extracted with hexane (2.5 mL) for analysis [25].
2.3. Rice Straw Pretreatment
Rice straw was provided by the Rural Development Administration in South Korea. Rice straw that was chopped and sieved through a 2 mm sieve was dried at 60°C for 24 h. The prepared rice straw was placed in 6% sulfuric acid (H2SO4) at a solid-to-liquid (S/L) ratio of 1:10, and the mixture was autoclaved at 121°C for 1 h. The autoclaved mixture was cooled down to room temperature, and centrifuged at 7,000 g for 5 min to separate supernatant. The supernatant was neutralized with 5 M NaOH to pH 6.5, and filtered through a 0.2 μm syringe filter to obtain the rice straw hydrolysates.
2.4. Rice straw Hydrolysate Analysis
Total organic carbon (TOC) and total nitrogen (TN) of the rice straw hydrolysates were analyzed using a TOC analyzer (Shimadzu, Japan) and the Humas water test kit (Humas, Korea), respectively. The hydrolysate composition was analyzed with high-performance liquid chromatography (HPLC; Dionex Ultimate3000, USA) equipped with an Aminex 87H column and a refractive index detector (ERC; RefractoMAX520, Japan). The HPLC column was eluted with 0.01 N H2SO4 at 0.5 mL/min at an oven temperature of 40°C.
2.5. PHA Analysis
The PHA extracts were analyzed by using gas chromatography (GC, 6100; Young Lin Instrument Co., South Korea) equipped with a HP-1 column (30 m × 0.32 mm I.D. × 1.00 μm) and a flame ionization detector. The temperature profile is as follows: 70°C for 5 min followed by 20°C/min rise to 240°C, which was held for 1 min. The helium flow rate was 3 mL/min and the temperatures of the injection and detection ports were both 250°C. Poly(3-hydroxybutyric acid-co-3-hydroxyvaleric acid) (P(3HB)- co-(3HV)) with poly(3-hydroxyvalerate) (P(3HV)) content of 12 wt % (Sigma Aldrich) was used to prepare standard solutions. The concentrations of poly(3-hydroxybutyrate) (P(3HB)) were divided by the DCW and multiplied by 100 to obtain P(3HB) content (%).
3. Results and Discussion
3.1. Effect of C/N Ratios at a Fixed Carbon Concentration on PHA Accumulation Pattern
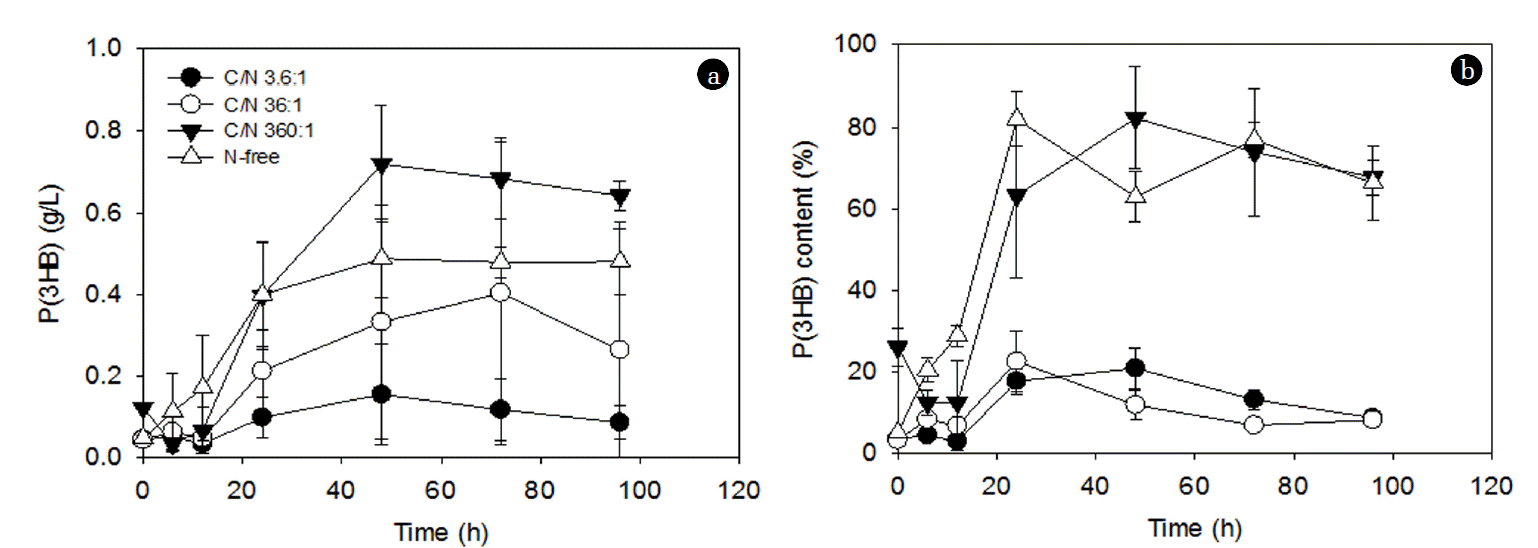
The effect of C/N ratios at a fixed glucose concentration (2 g/L) on PHA accumulation is shown in Fig. 1. The accumulated PHA in this study was P(3HB). Regardless of the C/N ratios, the accumulation stage had a lag phase of approximately 12 h before initiation of PHA accumulation, except the N-free condition (Fig. 1). The lag phase before initiation of PHA accumulation (Fig. 1) may imply that the cultures having both C and N sources need some time to enter into the N-deficient condition, which triggers PHA accumulation [22, 26, 27]. Based on the average values, the PHA concentration increased with time reaching the maximum after 48–72 h depending on the C/N ratios (Fig. 1(a)). On the other hand, the PHA content reached the maximum after 24–48 h depending on the C/N ratios (Fig. 1(b)). After the maximum PHA accumulation, the PHA concentrations or the PHA contents were maintained at a similar level or decreased with time (Fig. 1).
The PHA accumulation generally increased with increasing C/N ratio at the fixed glucose concentration (i.e., increasing degrees of N-deficient conditions) (Fig. 1(a)). Similarly, previous studies observed that N-deficient conditions in the presence of excess carbon sources promoted higher PHA accumulation [22, 26, 27]. For example, P(3HB) productivity by C. necator increased from 0.08 to 0.17 g/L/h with decreasing (NH4)2SO4 concentration from 3.00 to 0.75 g/L at a fixed C concentration of 80 g/L using corn syrup, which mainly consists of glucose [28]. Up to 24 h, the N-free condition seemed to be better for PHA accumulation than the C/N ratio of 360:1 condition. When the PHA concentrations after 48 h accumulation were compared, the PHA accumulation increased from 0.15 g/L to 0.72 g/L with increasing C/N ratios from 3.6:1 to 360:1; however, further increase in the C/N ratio (i.e., N-free condition) resulted in a lower PHA concentration (Fig. 1(a)). This suggests that, depending on the PHA accumulation period, the higher degrees of N-deficient condition (e.g., C/N ratio of 360:1) may be preferred over the N-free condition for PHA accumulation.
Furthermore, during the first 48 h, the PHA accumulation rate increased from 0.0034 (g/L/h) to 0.015 (g/L/h) with increasing C/N ratio from 3.6:1 to 360:1, while the PHA accumulation rate under the N-free condition (0.012 (g/L/h)) was similar to that at the C/N ratio of 360:1. Although the PHA accumulation rates under the N-free condition and at the C/N ratio of 360:1 were similar, the total amount of PHA that can be obtained is likely to be greater at the C/N ratio of 360:1 as new cells are likely to be generated (Fig. 1). The stagnation of PHA accumulation at later times (after 48 h) (Fig. 1(b)) may partially be explained by exhaustion of the carbon source (i.e., glucose) in the media, which is likely to create conditions that no longer have excess carbon sources for PHA accumulation, and this could possibly lead to endogenous PHA usage for maintaining bacterial metabolism [29].
3.2. Effect of C/N Ratios at a Fixed N Concentration on PHA Accumulation Pattern
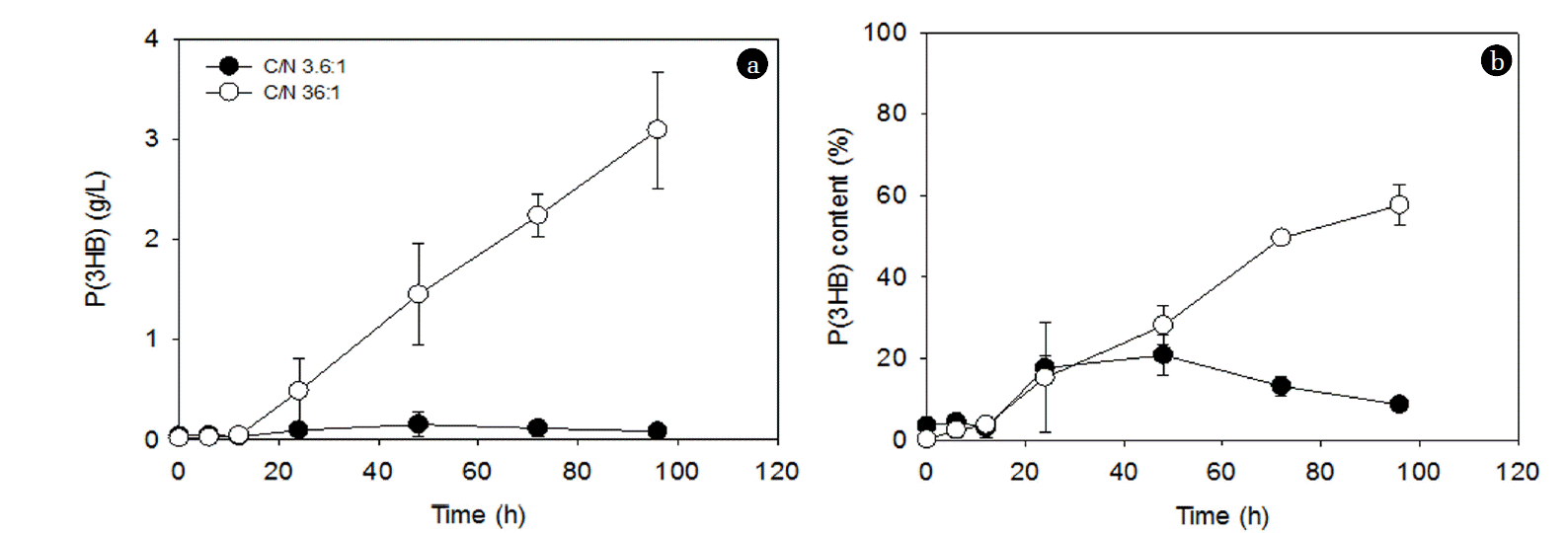
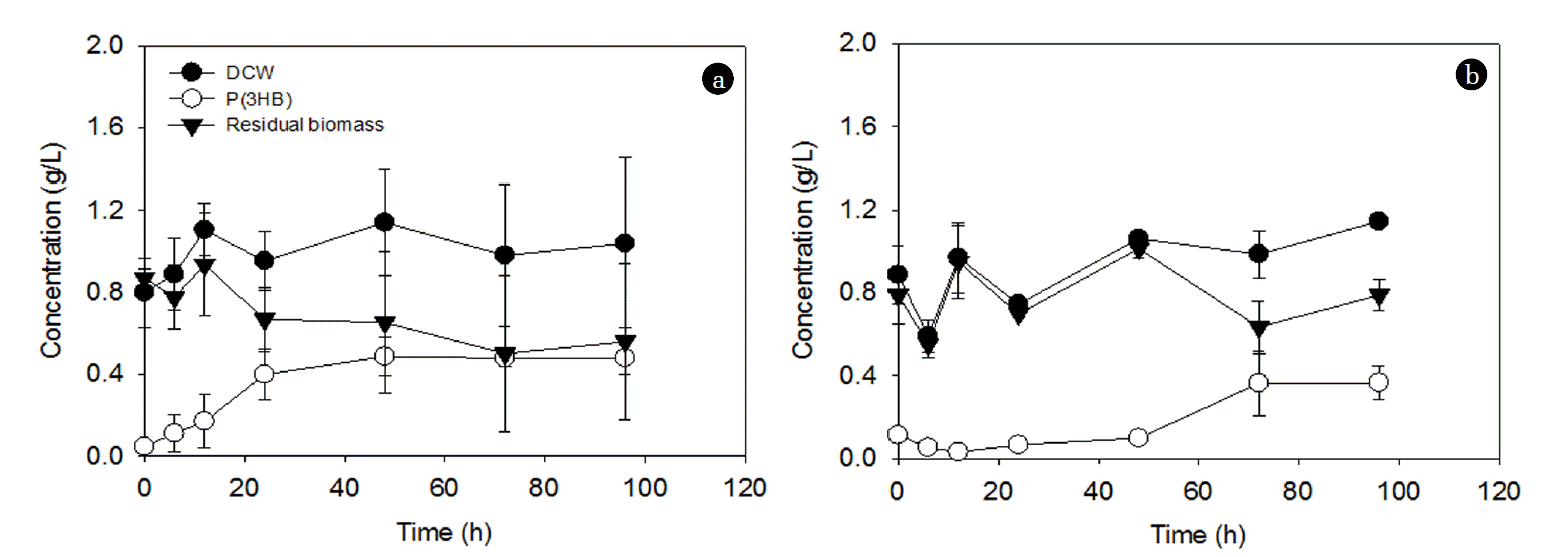
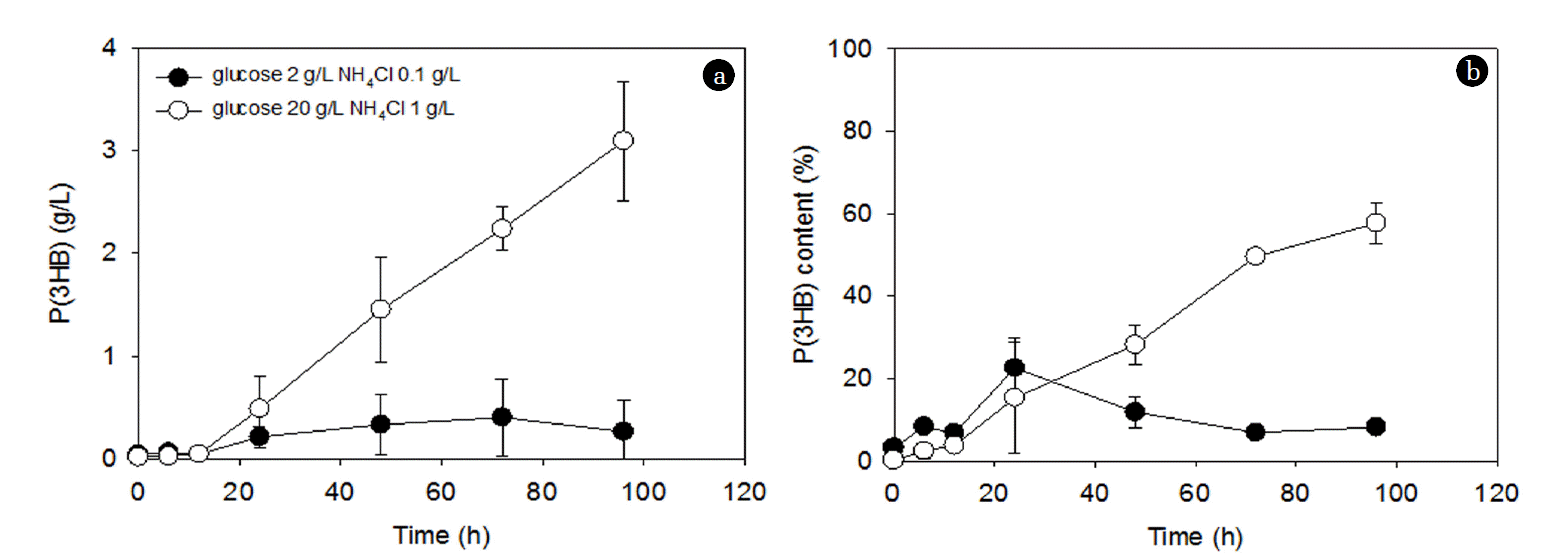
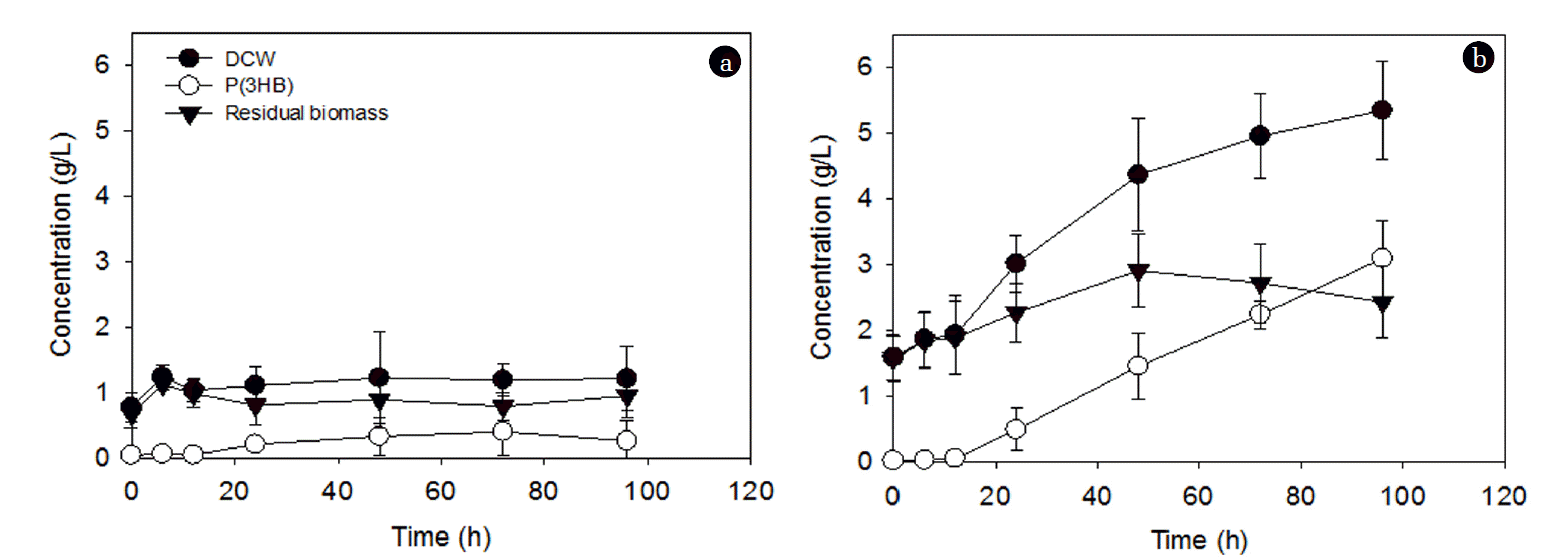
The effect of C/N ratios at a fixed NH4Cl concentration (1 g/L) on PHA accumulation is shown in Fig. 2. After an initial lag phase, the P(3HB) concentration continued to increase with time at the C/N ratio of 36:1, while it was almost negligible at the C/N ratio of 3.6:1 (Fig. 2(a)). Contrastingly, the PHA content was statistically similar at both C/N ratios up to 48 h; however, after that, it gradually decreased at the C/N ratio of 3.6:1, while it kept increasing to approximately 60% at the C/N ratio of 36:1 (Fig. 2(b)). The changes in P(3HB), DCW, and residual biomass for these two conditions are shown in Fig. 3. The residual biomass was calculated by subtracting the PHA mass from the DCW. The DCW and residual biomass were similar at the C/N ratio of 3.6:1 (Fig. 3(a)), while the difference between the DCW and the residual biomass increased with time at the C/N ratio of 36:1 (Fig. 3(b)). This suggests that the increase in the residual biomass over 96 h at the C/N ratio of 3.6:1 may be largely due to the production of new cells rather than PHA accumulation (Fig. 3(a)). This may also explain the decrease in the PHA content after 48 h accumulation (Fig. 2(b)). On the other hand, at the C/N ratio of 36:1, the residual biomass increased up to 48 h and then stayed at a similar level afterward (Fig. 3(b)). The increase in the residual biomass up to 48 h is likely to be attributed to both cell production and PHA accumulation and the similar level of the residual biomass after 48 h indicates that the increase in DCW is largely due to PHA accumulation (Fig. 3(b)). This could explain the significant increase in PHA accumulation after 48 h (Fig. 2).
Interestingly, when NH4Cl was absent, P(3HB) concentrations were initially greater at a lower glucose concentration (i.e., 2 g/L, a lower C/N ratio) than at a higher glucose concentration (i.e., 20 g/L, a higher C/N ratio), although the P(3HB) concentrations were statistically similar after 72 h (Fig. 4(a)). At 20 g/L glucose, the PHA concentration increased continually over 72 h, while the PHA concentration increased only up to 48 h at 2 g/L glucose (Fig. 4(a)). On the other hand, the P(3HB) contents were significantly higher at 2 g/L glucose concentration throughout the 96 h accumulation period, which gradually decreased after 24 h (Fig. 4(b)), and this can be attributed to the similar DCW over the 96 h accumulation period, which suggests negligible cell production (Fig. 5(a)). The residual biomass decreased, while the P(3HB) concentration increased with time throughout the 96 h accumulation period in the presence of only 2 g/L glucose (Fig. 5(a)). At 20 g/L glucose, the increase in the P(3HB) was observed after 48 h (Fig. 5(b)). In other words, the lag phase until the initiation of PHA accumulation was longer in the presence of 20 g/L glucose than in the presence of 2 g/L glucose (Fig. 5). The similar levels of DCW in the presence of only 2 or 20 g/L glucose indicate that new cell production was not involved in the absent of NH4Cl as N is required for new cell production. This is supported by the approximately ten times greater PHA accumulation in the presence of NH4Cl (3.1 g/L P(3HB) after 96 h) (Fig. 2(a)) than in the absence of NH4Cl (0.37 g/L P(3HB) after 96 h) (Fig. 4(a)) at 20 g/L glucose. Similarly, approximately four times greater PHA accumulation by Pseudomonas sp. LDC-5 was observed in the presence of (NH4)2SO4 (3 g/L of PHA) than in the negligible presence of N (0.8 g/L of PHA) [30]. Overall, in the absence of N, the similar PHA concentrations may be achievable by using different PHA accumulation periods at different C source concentrations.
3.3. Effect of C and N Concentrations at a Fixed C/N Ratio on PHA Accumulation Pattern
At a fixed C/N ratio of 36:1, different concentrations of glucose and NH4Cl resulted in different P(3HB) accumulation (Fig. 6). At lower concentrations of both glucose (2 g/L) and NH4Cl (0.1 g/L), the changes in the P(3HB) concentration were insignificant with time (Fig. 6(a)), and the P(3HB) content reached the maximum (23%) after 24 h and decreased afterward (Fig. 6(b)). On the other hand, at higher concentrations of both glucose (20 g/L) and NH4Cl (1 g/L), the P(3HB) concentration and content increased significantly with time (Fig. 6). The insignificant changes in the P(3HB) concentration at lower concentrations of C and N are in agreement with a slight decrease in the residual biomass, which was accompanied by a slight increase in P(3HB) concentration (Fig. 7(a)). The increase in P(3HB) at higher concentrations of C and N was accompanied by significant increases in both residual biomass and P(3HB) (Fig. 7(b)), which indicate both cell production and P(3HB) accumulation. The results suggest that, with relatively lower amounts of C and N, the PHA accumulation period needs to be reduced to achieve similar PHA accumulation to that with relatively higher amounts of C and N.
3.4. Implication on PHA Accumulation using Rice Straw Hydrolysates
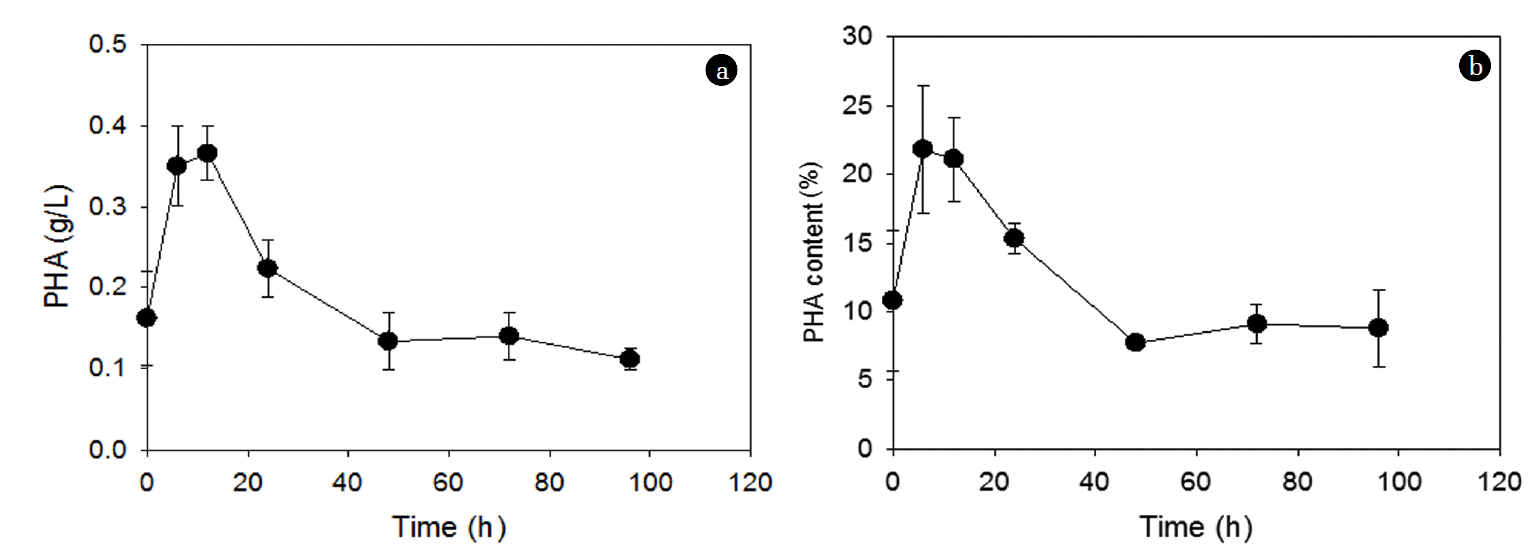
The C/N ratio of the rice straw hydrolysates obtained in this study was estimated to be approximately 270:1, based on the TOC and TN of 19,700 and 72 mg/L, respectively, of rice straw hydrolysates. Table 1 shows the composition of the rice straw hydrolysate obtained in this study. Sugars (e.g., glucose, xylose, arabinose) constituted approximately 85% of the hydrolysates obtained in this study. There are also other carbon sources such as organic acids (e.g., formic acid, levulinic acid). Among the sugars in the hydrolysate, C. necator may selectively utilize glucose but not xylose or arabinose [31]. Thus, with C. necator, the C/N ratio of the rice straw hydrolysate may be adjusted to approximately 160:1 based on the glucose content. Although the estimated C/N ratio of the hydrolysate is between the 36:1 and 360:1 where the PHA accumulation is increasing with increasing C/N ratio, the hydrolysate also contains other constituents such as furfurals and 5-HMF (Table 1) that may accumulate inside the cells and inhibit utilization of C sources (e.g., glucose) by C. necator, and hence, PHA accumulation [31]. Using the rice straw hydrolysate, the PHA concentration of 0.36±0.0033 g/L and PHA content of 21±3.1% were obtained after 12 h accumulation at the hydrolysate-to-media ratio of 1:9 (v/v) (Fig. 8). The highest PHA concentration and content were obtained after 12 h and both decreased with time. Considering that the most PHA accumulation was generally achieved between 12 h and 24 h after the initial lag phase of about 12 h using glucose and NH4Cl, the use of the 12 h accumulation period to obtain the maximum PHA accumulation using the hydrolysate seems to be reasonable (Fig. 8).
4. Conclusions
This study showed that, depending on C/N ratios, the PHA accumulation patterns, the time to reach the maximum PHA concentration and content, are different. Although PHA accumulation is generally greater under higher degrees of N-deficient conditions (e.g., C/N ratio of 360:1) than under lower degrees of N-deficient conditions (e.g., C/N ratios of 3.6:1 and 36:1), similar PHA accumulation may be obtained by using different accumulation periods. During the PHA accumulation period, the most PHA accumulation was observed during the first 12 h after the initiation of PHA accumulation, regardless of the initial C/N ratios and the C and N concentrations. The results suggest that it may not be necessary to adjust the initial C/N ratio of the organic wastes used as raw materials for PHA accumulation, but the PHA accumulation period may be adjusted. Therefore, when organic wastes such as rice straw hydrolysate are used as C and N sources, an appropriate accumulation period may be used for PHA accumulation based on the PHA accumulation patterns observed at various C/N ratios and C and N concentrations.