1. Introduction
Slags are a mixture of silica, calcium oxide, magnesium oxide, aluminum oxide and iron oxide and classified into four types based on the production process used, i.e., blast furnace slag, basic oxygen furnace (BOF) slag, electric arc furnace slag, and ladle furnace slag [1, 2]. Annually, more than 400 million tons of the slags are produced [3]. BOF slag is formed during moldering of iron into steel and ladle slag is a by-product of steel refining in both basic oxygen and/or electric arc furnaces [4].
Mineral carbonation is the reaction of metal oxides such as Ca, Fe, and Mg in materials with CO2 to form metal carbonate (Eq. (1)), in which Ca− and Mg-oxides react with CO2 and produce CaCO3 and MgCO3 [5, 6]. The mineral carbonation is divided into gas-solid carbonation and direct aqueous carbonation. Compared to gas-solid carbonation, the aqueous carbonation can reduce the energy requirement for carbonation with high carbonation efficiency [6–9].
The carbonation can also occur depending on the content of Ca− and Mg-silicates such as calcium silicate (Ca2SiO3) and magnesium silicate (Mg2SiO4) in slags as shown in Eq. (2) and (3) [6, 10]. Bernal et al. [11, 12] investigated the natural carbonation of aged alkali-activated slag concretes. Several other researchers have worked the acceleration of aqueous carbonation of the steel-making slag [13], and the BOF slag using a rotating packed bed and the production of calcite (calcium carbonate, CaCO3) [7]. In addition, other parameters such as slag particle size, mineralogical characteristics, air pressure, CO2 concentration, temperature, and reaction time should be also considered to estimate the potential to the carbonation rate [8].
Many studies have been reported on the immobilization and stabilization of heavy metals with various kinds of binders such as cement, slag, lime and apatite [14–19]. Lee et al. [17] reported the good stabilization efficiency of Cd-, Pb− and Zn-contaminated soils with the furnace slag, decreasing the bioavailability of the heavy metals.
The leaching behavior of heavy metals may be related to the pH of slag. The pH buffering capacity of the slag related to hydroxide/ carbonate buffering may decrease by carbonation. Kim et al. [16] reported that pH in carbonated slag paste can decrease, and the solubility of some heavy metals can increase. Conversely, Capobianco et al. [15] reported that the increase of the buffering capacity was observed with the high calcite content of the carbonated slag. Liu et al. [18] reported that the carbonation improved the immobilization efficiencies of heavy metals. The effectiveness of slag in heavy metals immobilization in contaminated soil has been reported in several studies [17, 20]. The effect of carbonation on the leaching characteristics of heavy metals in contaminated soil has been reported [18]. Carbonated slag with CO2 sequestered is expected to be effective in the stabilization of heavy metals in contaminated soils. Therefore, the effect of carbonation on the immobilization characteristics of heavy metals should be further investigated.
In the present work, the objective of this study is to investigate the effect of three operational parameters (pressure, temperature and liquid/solid (L/S) ratio) on the efficiency of aqueous carbonation of BOF and ladle slags for CO2 sequestration and the effect of the carbonation on immobilization efficiencies of heavy metals in contaminated soils.
2. Materials and Methods
2.1. Chemicals
Sodium acetate (NaCH3COO, 99%), isopropyl alcohol (C3H7OH, 70%), ammonium acetate (C2H3O2NH4, 95%), hydrochloric acid (HCl, 35–37%), nitric acid (HNO3, 70%), acetic acid (CH3COOH, 95%), hydroxylamine hydrochloride (HONH2·HCl, 95%), and ethanol (C2H5OH) were purchased from Duksan Pure Chemical, Korea, and hydrogen peroxide (H2O2, 29–32%) was purchased from Alfa Aesar (MA, USA). Inductively coupled plasma multi-element standard solution IV was purchased from Merck, Germany.
2.2. Slags
BOF slag (POSCO Co., Pohang, Korea) and ladle slag (Hyundai Steel Co., Dangjin, Korea) were air-dried for 2 w, disaggregated, homogenized using a tumbler, sieved through a 595 μm mesh (sieve #30), and then stored in an airtight container before use.
Elemental compositions of raw and carbonated slags were analyzed using X-ray fluorescence (XRF, PW2400, Philips Electronic Instruments, Inc., the Netherlands), and mineral compositions were identified using X-ray diffraction (XRD, X’pert PRO MPD, PANalytical, the Netherlands). Sample morphologies were examined by ultra-high-resolution field-emission scanning electron microscopy (FE-SEM, S-4300, Hitachi, Japan), and surface areas and pore size distributions were characterized using a surface area and pore size analyzer (BET; Autosorb-iQ & Quadrasorb SI, Quantachrome, USA). The content of calcium carbonate in raw and carbonated slags was quantified by thermogravimetric analysis (TGA, Q500, TA Instruments, USA). A dried slag sample (50 mg) was heated from 25 to 1,000°C with a rate of 10°C/min under nitrogen atmosphere. The CO2 uptake was determined by the mass loss during heating from 500 to 800°C.
2.3. Contaminated Soils
Two different contaminated soils were used for immobilization experiments. Soil A (N 36°51′55″, E 129°2′4″) was collected from a abandoned mine located in Bonghwa-gun, Gyeongsang buk-do and soil B (N 36°4′49″, E 126°00′27″) was collected from the closed Janghang metal refinery in Seocheon-gun, Chungcheong nam-do. Soils were air-dried for two weeks, disaggregated, sieved through sieve No. 30 (595 μm), homogenized using a tumbler, and stored in a closed container prior to use as described elsewhere [14].
Heavy metals were extracted from soil samples by using microwave-assisted acid digestion with aqua regia solution to estimate background concentrations [21]. The concentrations of the extracted heavy metals were measured by an inductively coupled plasma optical emission spectroscopy (ICP-OES, Optima 2100 DV, Perkin Elmer Inc., USA).
The physicochemical properties of soils are summarized in Table S1. Soil A was identified as neutral (pH 6.75), while soil B was moderately acidic (pH 5.7). The cation exchange capacity of soils A and B were 17.05 and 11.95 meq/100 g and the organic carbon contents were 0.77 and 0.44%, respectively. The background heavy metals concentrations in soil B were higher than soil A. Especially soil B was heavily contaminated with Pb (122,200 mg/kg) and Zn (17,520 mg/kg). Other heavy metals such as Cd, Cr, and Ni were not detected.
2.4. Aqueous Carbonation
The carbonation was conducted by preparing 100 g slags into a closed stainless steel reactor equipped with u-shape impeller and pure CO2 gas (PS Chem. Co., Gimhae, Korea). Gas in aluminum supply vessel is supplied into the reactor for a constant pressure condition by a differential pressure between reactor and supply vessel. The volumes of the reactor and the supply vessel were 256.4 and 571.3 cm3, respectively. The temperatures in the reactor and supply vessel were controlled by immersing them in a water: ethanol (7:3, v/v) bath. The temperatures and pressures in the reactor and supply vessel were recorded automatically every 30 s using a data acquisition system (Converter RS485-RS232C, Sejinyoung Tech, Korea).
The aqueous carbonation experiments were carried out under several conditions such as i) different pressures (1, 5, and 10 bar) at 25°C with an L/S ratio of 5 mL/g-dry, ii) different temperatures (1, 25, and 40°C) with 10 bar and L/S ratio of 5 mL/g-dry, and iii) different L/S ratios (1, 2, and 5 mL/g-dry) with 40°C and 10 bar, respectively.
After carbonation, the mixtures of slag and water were separated by centrifugation at 3,000 rpm and dried in an oven at 60°C. The following equation was used to calculate the CO2 uptake of slag [22]:
where nCO2(t) is the mole number of CO2 reduced in the slag-water mixture (mol), P0 and P(t) are pressures (kPa) at time 0 and t, respectively, R is the gas constant (8,314 cm3 kPa/mol/ K), T0 and T(t) are temperatures (K) at time 0 and t, respectively, z0 and z(t) are real-gas compressibility factors at time 0 and t, respectively, and VR is the reactor volume (571.3 cm3).
The carbonated CO2 concentration in slag at time t (qCO2 (t), mmol CO2/g slag) is calculated from nCO2 (t ) (mol) in Eq. (4) multiplied by thousand (1,000 mmol/mol) divided by slag weight (100 g). The qCO2(t) data were fitted to a pseudo-first-order kinetic model (PFOKM) and a pseudo-second-order kinetic model (PSOKM) to estimate the maximum carbonation capacity (mmol/g) [10, 23–25], as described below:
where qCO2,m 1 in Eq. (5) and qCO2,m2 in Eq. (6) are the maximum carbonated CO2 capacity in slag at equilibrium (mmol/g) for PFOKM and PSOKM, respectively, and k1 (1/min) and k2 (g/mmol/min) are the rate constants of carbonation for PFOKM and PSOKM, respectively. The 1/(k2qCO2.m2 ) in the denominator of Eq. (6) is defined as the half-carbonation time (t1/2, min) with the initial rate of CO2 uptake (r0, mmol/g/min) defined as:
Model parameters were estimated by nonlinear regression using a commercial software package, TableCurve 2D® (Version 5.1, SYSTAT Software, Inc.).
2.5. Acid Neutralizing Capacity (ANC) of Slags
ANC is the ability of a sample to maintain a certain pH during acid treatment [26] The ANC was used to assess the effects of carbonation [27]. ANC tests were performed to support the CO2 carbonation efficiencies for both raw and carbonated slags. Briefly, 1 g slag sample was mixed with 100 mL of distilled water in an Erlenmeyer flask, and 1 M HNO3 (0.5 mL) was added to the mixture every 24 h until pH was reached to be 8.2. Subsequently, the flasks were sealed to prevent any further contact of the solution with atmospheric CO2, and the pH was checked by a pH meter (ORION 3-Star, Thermo ScientificTM, USA).
2.6. Immobilization of Heavy Metals with Raw and Carbonated Slags
Immobilization of heavy metals in the contaminated soils with the raw and carbonated slags was conducted. Subsamples of slags (0.1, 0.3, or 1.0 g) were thoroughly mixed with 1.0 g of soil for 20 s using a vortex mixer (M37610-33, Thermo ScientificTM, USA). The mixtures were diluted with distilled water to 50 wt.% and allowed to stand for 24 h at room temperature [28] before the leaching test.
2.7. Analysis of Extractable Heavy Metals Concentrations in Soils
To measure the extractable concentrations of heavy metals in the soils with or without immobilization treatment with raw or carbonated slags, the toxicity characteristic leaching procedure (TCLP, USEPA, Method 1311) [29] and the Standards, Measurements and Testing Programme (SM&T, formerly BCR) [30, 31] were applied.
For TCLP, 1.0 g soil sample was extracted for 18 h with 20 mL of the acetic acid (0.57%, v/v). The mixture was centrifuged for 20-min at 1,500 rpm and the supernatant was filtered through a 0.2-μm syringe filter (Whatman, cellulose nitrate membrane, φ = 25 mm). The heavy metals concentrations in the supernatant were analyzed by using the ICP-OES.
The SM&T procedure consists of three steps of extractions such as acetic acid extraction (exchangeable phase, step 1), hydroxylamine hydrochloride extraction (reducible phase, step 2), and ammonium acetate extraction (oxidizable phase, step 3). After finishing each step, the mixtures were centrifuged for 10 min at 1,500 rpm, and the residues were washed with distilled water before using them for the next steps. The supernatants from each step were analyzed by the ICP-OES.
2.8. Quality Assurance and Quality Control (QA/QC)
Compliance with quality assurance and quality control (QA/QC) guidelines was achieved by performing two consecutive measurements to determine average values and relative standard deviations (RSDs). The above RSDs for Pb, Cd, Cr, Cu, Zn, and Ni equaled 1.05, 0.83, 0.70, 1.47, 0.86 and 0.40, respectively, and the standard calibration slope was verified by performing standard solution measurements every 20 samples [24].
3. Results and Discussion
3.1. Effect of Pressure, Temperature and L/S Ratio on CO2 Carbonation Rate
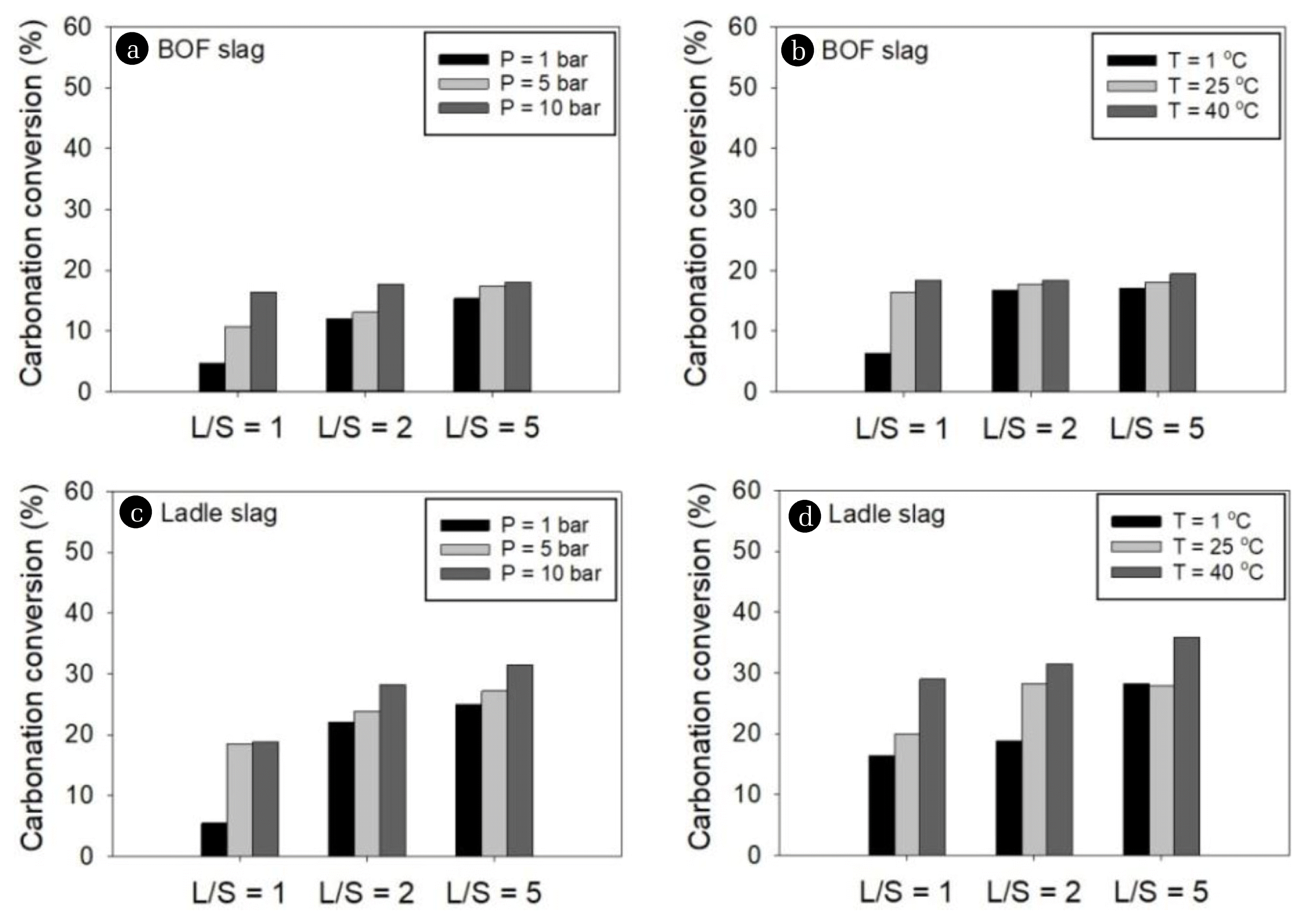
The effects of pressure, temperature, and L/S ratio on aqueous carbonation kinetics were investigated. Several operating conditions such as 1, 5, and 10 bar for pressure, 1, 25, and 40°C to investigate effect of temperature and 1, 2, and 5 mL/g-dry of L/S ratios were tested to draw optimum conditions.
As shown in Fig. S1, the carbonated CO2 concentration, qCO2(t) increased with time and then remained steady at equilibrium. The carbonation data were fitted to the PFOKM and PSOKM as the results were summarized in Table 1, respectively. In terms of R2 values, the PSOKM (R2 = 0.866–0.998) fitted the CO2 carbonation data better than PFOKM (R2 = 0.709–0.990) for all slags.
The pseudo-first-order rate constant (k1, min−1) for aqueous carbonation of BOF and ladle slags increased with pressure, temperature, and L/S ratio except for ladle slag at P = 10 bar and L/S = 5 mL/g-dry. The maximum capacity of CO2 carbonation (qCO2,m1 ) in PFOKM at equilibrium increased with pressure, temperature, and L/S ratio for both BOF and ladle slags, except for BOF slag at L/S = 2 mL/g-dry, P = 10 bar, and T = 40°C. The qCO2,m1 values of ladle slag were consistently higher than those of BOF slag.
The maximum capacity of CO2 carbonation (qCO2,m2 ) in PSOKM also increased with pressure, temperature, and L/S ratio for both BOF and ladle slags (Table 1). The highest qCO2,m2 value of ladle slag (1.0380 mmol/g) obtained at 10 bar, 40°C and L/S = 5 mL/g-dry was higher than that of BOF slag (0.5835 mmol/g). On the contrary, the carbonation rates of CO2 into BOF slag was faster than those into ladle slag. The highest pseudo-second-order rate constants (k2, mol−1 min−1) for BOF and ladle slags were 0.3694 and 0.1203 g/mmol/min obtained at T = 40°C, P = 10 bar and L/S = 2 mL/g-dry and at T = 1°C, P = 10 bar and L/S = 5 mL/g-dry, respectively. The k2 value was not correlated with pressure, temperature, and L/S ratio, however, the k2 and initial carbonation rate (r0) of BOF slag were higher than those of ladle slag at all conditions.
Fig. 1 shows that the carbonation conversion ratio (%), a ratio of the carbonated CO2 into slags from the injected CO2 gas, increases with pressure, temperature, and L/S ratio for both BOF and ladle slags, which was in agreement with other researches [8, 13, 32]. Polettini et al. [8] and Ukwattage et al. [13] reported that the rate of carbonation is enhanced up to 50°C due to Ca leaching that was probably the rate-determining reaction step at below 50°C. In this study, the L/S ratio played more import role than the pressure and temperature in determining carbonation efficiency of BOF and ladle slags. This is because the high L/S ratio can increase hydration and dissolution of Ca and CO2 in aqueous phase [33], as indicated by Eq. (1)-(3), thereby increasing CO2 sequestration.
3.2. Comparison of Physicochemical Characteristics of Raw and Carbonated Slags
The BET surface area, pore volume, pore size and the results of XRF analysis for raw and carbonated slags were listed in Table 2. In comparison of Table 1 and 2, the maximum capacities of CO2 carbonation (qCO2,m1 and qCO2,m2 ) in ladle slag were higher than those in BOF slag, it can be attributed to the specific surface area, pore volume, pore size, and Ca and Mg contents of slags. Ukwattage et al. [13] and Salman et al. [34] also reported the same conclusion that CO2 sequestration capacity is directly dependent on the proportions of binary oxides (CaO and MgO) and/or hydroxides (Ca(OH)2 and Mg(OH)2) in the matrix. Pan et al. [7] and Ko et al. [35] emphasized that the hydration of Ca2+ and Mg2+ in steel slags during carbonation is important to for accomplish high CO2 reactivity and capture capacity. Baciocchi et al. [36] and Boone et al. [37] reported that the higher specific surface enhances the reactivity of slags with CO2, and the greater pore volume and pore size increase the capacity for carbonated mineral precipitates.
Fig. 2 shows the XRD analyses for raw and carbonated slags. The main crystal phases of BOF and ladle slags were quartz (SiO2, Q) and dicalcium silicate (Ca2SiO4, D), respectively. The BOF slag also contains calcite (CaCO3, C) and calcium hydroxide (Ca(OH)2, H), and the ladle slag has calcite (C), brucite (Mg(OH)2, B), and katoite (Ca3Al2(OH)12, K). However, no Mg was detected in BOF slag as examined by XRD as well as XRF analysis (Table 2). After carbonation, the peak of calcium hydroxide (Ca(OH)2, H) in BOF slag disappeared and the peak area of the dicalcium silicate (Ca2SiO4, D) in ladle slag decreased, whereas the intensity of the calcite (CaCO3, C) peaks in BOF and ladle slags remarkably increased.
The decomposition of hydrates and carbonates in slags was investigated by the thermogravimetric analysis (TGA). Fig. 3 shows that decomposition of hydrates can be divided into three weight loss fractions, assigned to (i) moisture loss at 25–105°C, (ii) organic carbon oxidation and MgCO3 decomposition at 105–500°C, and (iii) CaCO3 decomposition at 500–780°C [38]. The mass loss of CaCO3 in raw and carbonated BOF slags at 500–780°C was about 7% and 16%, while that of ladle slag was 3.7% and 13%, respectively. The content of CaCO3 in carbonated BOF and ladle slags was approximately 5% and 9.3%, respectively, as indicated by TGA, confirming that ladle slag exhibited higher CO2 uptake than BOF slag.
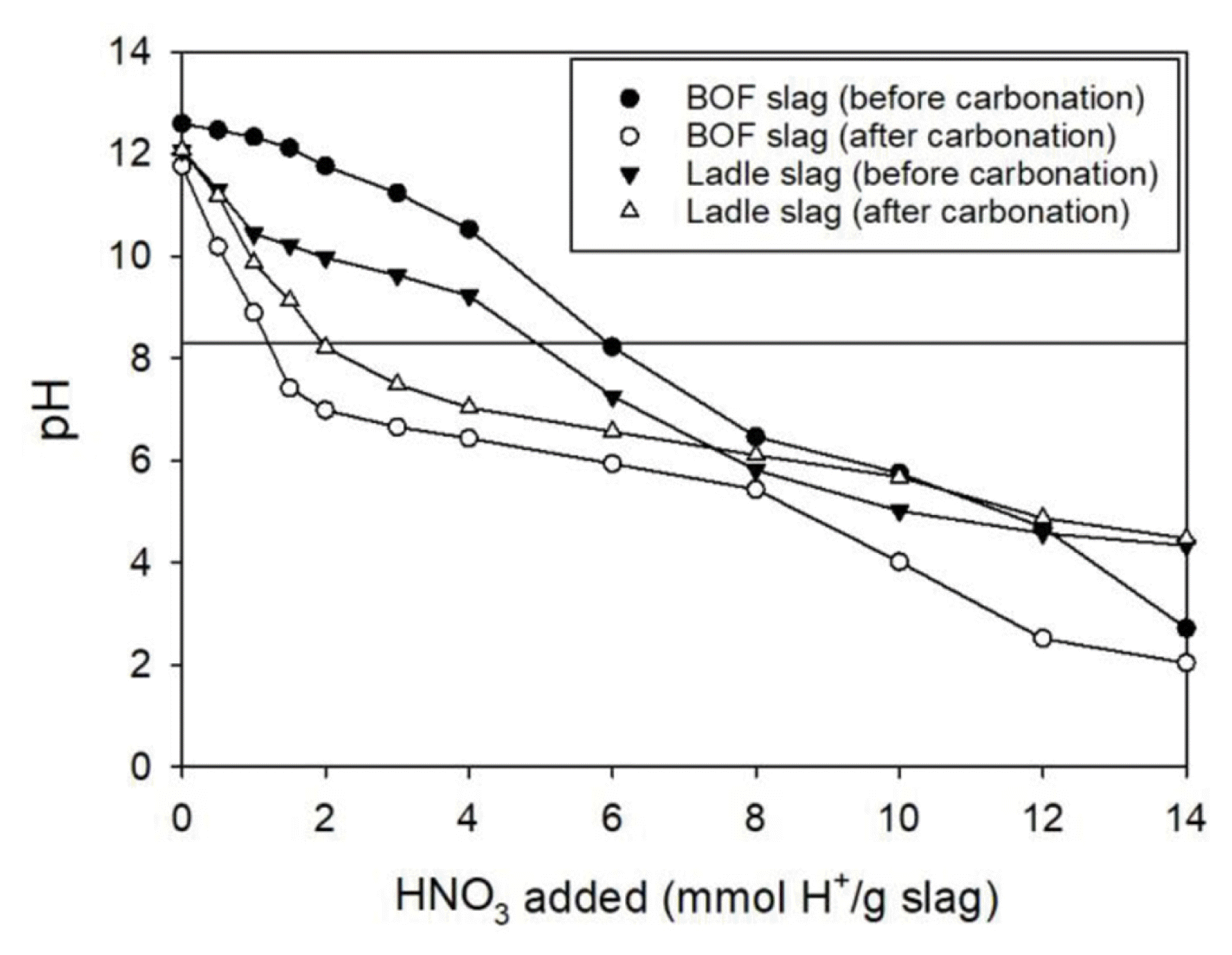
The ANC values of raw and carbonated slags were compared. As shown in Fig. 4, ANC of BOF and ladle slags decreased after carbonation. At pH 8.2, the ANC values of raw and carbonated BOF slags were 6.0 and 1.2 mmol H+/g slag, respectively, while those of raw and carbonated ladle slags were 5.0 and 2.0 mmol H+/g slag, respectively. The change of ANC values for BOF slag was higher than that for ladle slag, indicating that carbonated ladle slag has higher pH buffering capability than carbonated BOF slag because of higher CaCO3 content of ladle slag [26].
Consequently, ladle slag has higher potential of CO2 sequestration with producing CaCO3, because ladle slag has higher Ca content, BET surface area, pore volume, pore size and pH buffering capacity than BOF slag as supported by several analytic methods such as BET, XRD, SEM, TGA and ANC tests.
3.3. Effect of Carbonated Slag Amendment on the Immobilization of the Heavy Metals
Effect of carbonated slag amendment on the immobilization of heavy metals in the field-contaminated soils was estimated by using TCLP and SM&T extraction methods. The results of the extraction methods are summarized in Table 3 and 4, respectively.
According to TCLP test, Pb, Cu and Zn were the major contaminants in soils A and B. The highest TCLP-extractable heavy metals concentration observed was 75.2 mg/L of Cu in soil B. Pb concentrations in soils A and B (19.5 and 38.1 mg/L, respectively) also exceeded USEPA-regulated limits (5 mg/L), whereas Cd and Cr concentrations did not exceed the limits (see Table 3). TCLP extraction data for Cu, Ni and Zn was also used to compare the immobilization efficiencies of these heavy metals in soils although they are not included in TCLP extraction regulation.
All immobilization treatments by using both raw and carbonated slags successfully led to decrease TCLP-extractable heavy metals concentrations for all soils (Table 3). For soil A, the TCLP-extractable Pb concentration was 19 mg/L, surpassing the USEPA-regulated level of 5 mg/L, whereas it decreased to less than 0.28 mg/L after immobilization with raw and carbonated slags (i.e., immobilization efficiency > 98%). TCLP-extractable Zn also decreased from 29.4 mg/L in raw soil to less than 0.52 mg/L after immobilization with raw and carbonated slags except for carbonated BOF slag (1.19 to 6.67 mg/L). Cd, Cr, and Ni were not detected both before and after immobilization.
TCLP-extractable Pb concentration in soil B decreased from 38 mg/L in raw soil to 0.31 and 0.77 mg/L after immobilization using raw and carbonated ladle slags, respectively, while the use of raw and carbonated BOF slag resulted in the decrease to 4.58 and 5.20 mg/L, respectively. Cd, Cr, Cu, Ni, and Zn were well retained after immobilization with their TCLP-extractable concentrations decreased to below 0.55 mg/L.
Table 4 presents SM&T results in the exchangeable, reducible, oxidizable, and residual heavy metals fractions in soils A and B, after immobilizing using raw and carbonated BOF and ladle slags, respectively. The exchangeable fraction (step 1) generally decreased whereas the residual fraction (step 4) increased after immobilization with raw and carbonated slags.
The difference between using raw and carbonated slags was not noticeable for soil A while the fractionation of soil B contaminated by higher concentrations of heavy metals showed that raw slags performed better than carbonated ones. This observation is explained by the fact that the immobilization is affected by soil pH, with the use of raw slag resulting in a higher pH compared to the use of carbonated slag. This is because of the decrease in pH buffer capacity as OH− concentrations decreased during carbonation. The decrease in TCLP-extractable concentrations was due to the pozzolanic reaction where Ca in the slag was reacted with silica (SiO2) present in the soils to form C-S-H (Ca-Si-OH) gel that is responsible for the development of binding force [39] and the formation of additional calcite [40]. Calcite formed by carbonation and addition of slag seals the soil pores, resulting in preventing heavy metals leaching [40, 41]. Makhloufi et al. [42] also reported that calcium present in slags can form C-S-H gel during hydration due to the pozzolanic reaction. Carbonated slag generally has less Ca available for the pozzolanic reaction due to consumption of calcium hydroxide by CO2 binding in the preceding carbonation.
4. Conclusions
The applicability of BOF and ladle slags for CO2 carbonation and the immobilization efficiencies of heavy metals between raw and the carbonated slags were investigated. For higher capacity of aqueous carbonation, high pressure (5 atm), high temperature (40°C) and high L/S ratio (L/S = 5 mL/g-dry) were required. The maximum carbonated concentrations of CO2 in slags were 0.584 and 1.038 mmol/g of BOF and ladle slags, respectively. The physicochemical characteristics of slags including BET surface area pore volume and size, Ca contents, and pH buffering capacity affected the efficiency of CO2 carbonation.
The immobilization efficiencies of heavy metals with raw and carbonated slags were also evaluated by TCLP and SM&T procedures. The results of the leaching tests showed no effect of carbonation on the immobilization because of high calcium content (as CaO and Ca(OH)2) in slags. According to TLCP results, the extractable concentrations of heavy metals were varied irregularly with characteristics of soils, before and after carbonation, and dosage ratio of slag to soil. It should be noted that extractable concentrations of several heavy metals such as Zn and Pb allowed increasing after carbonation whereas other heavy metals did not have the same results. SM&T analyses also showed the unsystematic correlation in exchangeable and residual fractions between the raw and carbonated slags. In summary, although a clear correlation between TCLP and SM&T results and the physicochemical properties of soils and slags were not found, the results implicates that carbonated slag have benefits of heavy metals immobilization as well as CO2 storage.