Abstract3D printing has recently influenced membrane science. As a green alternative to current membrane fabrication methods, 3D printing prevents the mixing of highly toxic chemicals into water through its sustainable production. Furthermore, the risk of exposure to these toxic materials and of mechanical accidents during the fabrication is also attenuated. This type of in-situ fabrication eliminates logistic-based problems caused by transportation and packaging. Eliminating packaging and reducing transportation and precision-based waste also reduces CO2 emissions. The advantages of 3D-printed membranes are correlated with each other and promote a greener environment. In this article, we collect their contributions under the sub-titles of sustainability, risk reduction, cost-effectiveness, precision and mobility.
1. IntroductionAs the water-energy nexus has become the primary concern related to the outcomes of global warming, a need for energy-efficient methods for water purification has emerged [1–3]. Membrane technologies have already been widely adopted for real-life applications. However, compared to other purification methods, membrane treatments have crucial drawbacks that must be solved. Fabrication techniques are important in addressing these drawbacks.
Over the last few decades, phase inversion, interfacial polymerization, stretching, track-etching and electrospinning have been the most widely used conventional techniques. Phase inversion was introduced in 1960 and is the current basis for most commercially available membranes [4, 5]. Most porous polymeric membranes are fabricated via non-solvent induced phase separation (NIPS) and thermally induced phase separation (TIPS) methods [6]. For NIPS, the polymer solution is immersed in a non-solvent coagulation bath resulting in the exchange of solvent and non-solvent to form an asymmetric, dense surfaced membrane that can be used for reverse osmosis (RO) and nanofiltration (NF) systems. Demixing and precipitation occur during the NIPS type fabrications. TIPS is applied with semi-crystalline polymers and utilizes the thermal energy of a dope solution as the main driving force. They provide highly porous symmetric structures, which are suitable for microfiltration (MF) and membrane contactor applications [7–9]. It is based on the decrease in solvent quality as a result of decreased temperature. After the demixing, the solvent is removed by extraction, evaporation or freeze drying [10]. The main problem of these methods is the waste of huge amounts of solvents. Furthermore, with these methods, it is difficult to control the precision and the uniformity. One of the most promising methods for the future of membrane fabrication is interfacial polymerization. Conventional interfacial polymerization has been conducted by immersing the support into monomer solutions followed by heat treatment. Although this method has the same waste and uniformity problems as NIPS and TIPS, it has recently been developed further through the use of 3D printing.
In 2018, Ma et al. [11] introduced the first 3D printing–assisted thin film composite (TFC) membrane fabrication by electro-spraying two monomer solutions to fabricate a polyamide layer through interfacial polymerization on a polymer substrate. This approach was further developed to fabricate the first self-standing 3D-printed ultrafiltration (UF) membrane [12]. Rather than getting the advantages of interfacial polymerization such as high perm-selectivity properties, electro-spraying based these works are also the initials of future 3D-printed membranes, which can utilize further approaches that can even design the pore structures through computer modeling. In addition, 3D printing offers more in terms of sustainability, risk reduction, cost-effectiveness, precision and mobility (Fig. 1). Here, we present these major points that designate 3D printing as a promising, green revolution for the fabrication of polymeric membranes.
2. Five Breakthroughs of 3D Printed Membranes2.1. SustainabilityManufacturing is responsible for 33% of the total carbon footprint [13]. This ratio will increase as the energy required for manufacturing increases. According to the International Energy Outlook 2017, prepared by U.S. Energy Information Administration, energy consumption by manufacturing will increase steeply by 2040 [14] (Fig. 2). However, some recent projections show that these consumptions could be reduced substantially by 3D printing. As a derivation of 18 projections, Delphi projections show that the carbon footprint of manufacturing and transportation will be greatly reduced by 3D printing [15]. When considering membrane fabrications, these ratios increase.
Although membrane processes are known as green processes, the same cannot be said about fabrication. Membrane fabrication requires highly toxic and dipolar aprotic solvents like N-methyl-2-pyrrolidone (NMP), N,N-dimethylformamide (DMF) and N,N-dimethylacetamide (DMAc) [16, 17]. Fifty billion liters of solvent-containing wastewater are generated every year when these solvents are mixed with water during the membrane fabrication [18] (Fig. 3). Further disposal of these solvent results in a huge amount of CO2 emissions. For example, the fabrication of TFC membranes with phase inversion methods creates a large amount of liquid and gas waste during solution casting, phase inversion (very high), solvent swapping, crosslinking, washing, conditioning, coating and active layer formation processes. Different from the conventional methods, 3D printing does not use membrane materials more than required amount, and it also eliminates coagulation bath. Therefore, the huge amount of wastes production highly decreases, so does resulting CO2 emissions.
2.2. Risk ReductionSynthesis of a polymeric membrane requires toxic materials, exposure to which can pose a health risk. Even when precautions are taken, there are still risks of harm by those materials. Current phase separation processes for membrane fabrications require a homogeneous polymer solution to transform into a two-phase system. The solid, polymer-rich phase forms the membrane structure, while a liquid polymer-poor phase forms the porous structure [19]. During the process, the polymer is dissolved in a mixture of a volatile good solvent and an involatile poor solvent. This is followed by the evaporation of the good solvent, which enriches the cast film in non-solvent, causing precipitation. The preparation process of the solvent and the evaporation of the volatile solvent creates a high risk of exposure. Exposure to these toxic solvents can result in various health problems, ranging from light skin problems, headaches or irritation to life-threatening illnesses including cancer and permanent neural problems (Table 1). Detailed risks of these solvents are provided in Table 1 together with the most common solvents.
Through 3D printing–based membrane fabrications, the risk of exposure will be reduced to a minimum level as 3D printing is a process which is controlled by remote commands through a computer.
When industrial fabrications are considered, there is an additional risk of accidents occurring during the mechanical processes. Such accidents will also be reduced to some extent with the assistance of 3D printing. In addition, because of the in-situ nature of 3D-printing fabrication, transportation accidents will be eliminated.
2.3. Cost-EffectivenessSince its inception, 3D printing has become integrated into almost every area of science; one of the main reason for this is the reduced fabrication cost that is associated with 3D printing. However, when we consider membrane science, the situation is more relative to the scale. Considering the current situation of 3D-printing technology, it would be controversial to say low-cost fabrication when it comes to the ones requiring nano-precision. Long printing times and complex machinery make even sub-micron pored membrane fabrication very costly [50]. However, the research of Chowdhury et al. [12] demonstrates the promising future of low-cost 3D-printed membranes.
The main cost advantage of 3D printing for membrane fabrication is the ability to fabricate required amount without wasting materials. Unlike the materials wasted during the phase inversion of conventional fabrication methods, 3D printing does not use more materials than required. Comparing immersing type and electro-spraying type interfacial polymerizations clearly demonstrates this difference. Another important benefit of 3D printing is the reduction in labor cost for mass production. Machines will eventually replace some human laborers through the application of 3D printing. When in-situ fabrication of membranes become common, shipping and packing costs will also no longer be a concern [51]. In addition, bespoke fabrication for the membrane sizes required for specific needs will reduce the cost associated with sizing the membrane.
2.4. Precision3D printing is a precise fabrication method that produces complex structures in macroscale [52, 53]. Although was previously unheard of to say the same thing about micro and nanoscales, recent developments in 2-photon polymerization by spatiotemporal focusing of the femtosecond laser pulses made it possible to have ≈100 nm resolution [54]. It is currently possible to do surface printing around 60 nm resolution through super-resolution laser direct writing [55]. This can go down up to 20 nm when lithographic methods are considered, which was previously applied successfully at Caltech in the creation of nano-architected metals [56]. It is not a huge leap to go below 10 nm resolution, which will allow us to adjust the pore sizes of the polymeric membranes more precisely.
The developed precision of 3D printing will also let researches apply biomimetic structures successfully. Indeed, it has been successfully applied in micro-scale by mimicking surfaces from nature. Immersed surface accumulation 3D printing of micro-scale artificial hairs with eggbeater heads, which mimic the Salvinia molesta leaf, is an example of this [57]. As 3D-printing technology is further developed, we will likely see this research approach in nano-scale as well.
Furthermore, precise fabrication also provides uniformity for 3D-printed membranes. Current fabrication methods cannot produce uniform membranes with uniform pore distribution and thickness. The size control properties of 3D printing would bring uniformity to this area of membrane science, helping to clear away any possible conflict between producers and customers.
2.5. Mobility3D printing is a convenient fabrication method that is versatile, rapid and in-situ (making it ready to use for any case in place). Furthermore, it is possible to fabricate complex structures to order without having to account for transportation time. In this way, on-demand fabrication carries great importance. One 3D printer launched by the National Aeronautics and Space Administration (NASA) at the International Space Station in 2014 is an example of the mobility of additive manufacturing technologies. With this 3D printer, astronauts are able to produce various types of objects according to their requirements, such as replacements for broken parts or innovative tools under space conditions [58].
The mobility of 3D printing applies to membrane science in several ways. In-situ fabrication of membranes is highly beneficial for desalination membranes in particular as these types of membranes are mostly required in dry regions as in Gulf countries. The high temperature in such arid areas can result in the loss of membrane properties. Therefore, high-cost protections are needed until the membrane starts its filtration life. In-situ fabrication will reduce the length of the waiting period before use. Furthermore, in the case of urgent requirement, any broken or damaged module part of a membrane system can also be rapidly produced and replaced through 3D printing. This type of fabrication can also be incorporated with 3D scanning.
3. Current State and Limitations of 3D Printed MembranesMost of the limitations for 3D printed membranes come from the current status of 3D printing technology. 3D printing technology has already reached nano-scale production level. However, for this level, the precision that we mentioned in previous paragraphs becomes a critical problem. Even though, 3D printing is advantageous for the precision of micro or macro level fabrications, further developments are required for the nano-scale. Nevertheless, this applies to current fabrication methods as well, since they are not precise in nano-level [59]. Therefore, as the technology develops, 3D printing is expected to be the leading technology for nano-scale fabrication.
Huge sizes of 3D printers are another barrier to the current 3D printing technology in terms of footprint. Even for the fabrication of small sized objects, massive 3D printers are required. Consequently, mass production of the products becomes difficult as well.
Another issue that serves as a limitation to the use of 3D printers is price. Even though the machine printing in macroscales is affordable, micro or nano-scale printing ones are difficult to afford to the most regions of the world [60]. The high prices are also related to large sizes of current printers. That is, when printing scale is reduced to micro and nano-sizes, more complex machines with larger sizes are required. The greater material and assembly costs of these machines result in the higher prices.
Lastly, most of the 3D printers, especially the ultra-precise ones, are not able to fabricate with common materials used in simple 3D printers. They are designated to print with specific materials, such as Accura, Duraform, FabPro or Visijet series: polypropylene-like printing materials of 3D Systems [61].
These limitations of 3D printing technology are eliminated day by day as result of on-going researches. Therefore, it is not very far to see convenient size and ultra-precise 3D printers that is able to print with any selected material.
4. ConclusionsAs a greener alternative to current high-waste membrane fabrication methods, 3D printing minimizes the waste created during the production, packaging and transportation processes. Furthermore, the risks are also reduced by remote fabrication.
Currently, research related to 3D-printed membranes is still in its infancy. However, as 3D printing itself continues to develop, membrane fabrication utilizing the technology will also develop and will be commonly applied in water treatment and desalination plants. Researchers should pay further attention to 3D-printed membranes by considering their major contributions as a sustainable, risk-reducing, cost-effective, precise and mobile fabrication method for a greener environment.
AcknowledgmentThis work was supported by GIST Research Institute(GRI) grant funded by the GIST in 2019.
NotesAuthor Contributions This work has been held under the coordination and supervision of H.C.(Professor). N.Y.(Ph.D student) has done the scientific literature review and written the manuscript by also preparing the figures as first author. M.S.(Postdoctoral fellow) and H.P.(Research Professor) have revised the manuscript for several times and added critical parts to the manuscript for the final version. References1. Alayande AB, Akinlolu-Raphael SJ. Nigeria Water Crisis: A Function of Failed Governmental Planning and Policies. IGLUS Quarterly. 2019;5(1)27–31.
2. Yanar N, Choi H. Energy Perspectives of Korea (Republic of) with a General Outlook on Renewable Energy. IGLUS Quarterly. 2019;4(4)4–8.
3. Yanar N, Choi H. Urban Water Management and Quality-Based Water Use. IGLUS Quarterly. 2019;5(1)4–6.
4. Hołda AK, Vankelecom IFJ. Understanding and guiding the phase inversion process for synthesis of solvent resistant nanofiltration membranes. J Appl Polym. 2015;132(27)
5. Loeb S. The Loeb-Sourirajan Membrane: How It Came About. Synthetic Membranes. ACS Symposium Series. 153AMERICAN CHEMICAL SOCIETY; 1981. p. 1–9.
6. Arahman N, Maimun T, Mukramah , Syawaliah . The study of membrane formation via phase inversion method by cloud point and light scattering experiment. AIP Conference Proceedings. 2017;1788(1)030018
7. Gu M, Zhang J, Wang X, Tao H, Ge L. Formation of poly(vinylidene fluoride) (PVDF) membranes via thermally induced phase separation. Desalination. 2006;192(1)160–167.
8. Jung JT, Kim JF, Wang HH, di Nicolo E, Drioli E, Lee YM. Understanding the non-solvent induced phase separation (NIPS) effect during the fabrication of microporous PVDF membranes via thermally induced phase separation (TIPS). J Membr Sci. 2016;514:250–263.
9. Yeow ML, Liu YT, Li K. Morphological study of poly(vinylidene fluoride) asymmetric membranes: Effects of the solvent, additive, and dope temperature. J Appl Polym. 2004;92(3)1782–1789.
10. Lalia BS, Kochkodan V, Hashaikeh R, Hilal N. A review on membrane fabrication: Structure, properties and performance relationship. Desalination. 2013;326:77–95.
11. Ma X-H, Yang Z, Yao Z-K, Guo H, Xu Z-L, Tang CY. Interfacial Polymerization with Electrosprayed Microdroplets: Toward Controllable and Ultrathin Polyamide Membranes. Environ Sci Technol Lett. 2018;5(2)117–22.
12. Chowdhury MR, Steffes J, Huey BD, McCutcheon JR. 3D printed polyamide membranes for desalination. Science. 2018;361( 6403)682.
13. Liu L, Shamir A, Wang CCL, Whiting E, editors3D printing oriented design: geometry and optimization. 2014;
14. U.S. Energy Information Administration. International Energy Outlook 2017. 2017.
15. Jiang R, Kleer R, Piller FT. Predicting the future of additive manufacturing: A Delphi study on economic and societal implications of 3D printing for 2030. Technol Forecast Soc. 2017;117:84–97.
16. Guillen GR, Pan Y, Li M, Hoek EMV. Preparation and Characterization of Membranes Formed by Nonsolvent Induced Phase Separation: A Review. Ind Eng Chem Res. 2011;50(7)3798–817.
17. Alayande AB, Obaid M, Yu H, Kim IS. High-flux ultrafiltration membrane with open porous hydrophilic structure using dual pore formers. Chemosphere. 2019;227:662–669.
18. Razali M, Kim JF, Attfield M, Budd PM, Drioli E, Lee YM, et al. Sustainable wastewater treatment and recycling in membrane manufacturing. Green Chem. 2015;17(12)5196–5205.
19. Strathmann H, Kock K. The formation mechanism of phase inversion membranes. Desalination. 1977;21(3)241–255.
20. Satoh T, Omae K, Nakashima H, et al. Relationship between acetone exposure concentration and health effects in acetate fiber plant workers. Int Arch Occ Env Hea. 1996;68(3)147–153.
21. Smith MT. Advances in Understanding Benzene Health Effects and Susceptibility. Annu Rev Publ Health. 2010;31(1)133–148.
22. Iregren A, Löf A, Toomingas A, Wang Z. Irritation effects from experimental exposure to n-butyl acetate. Am J Ind Med. 1993;24(6)727–742.
23. Malley LA, Bamberger JR, Stadler JC, et al. Subchronic toxicity of cyclohexane in rats and mice by inhalation exposure. Drug Chem Toxicol. 2000;23(4)513–537.
24. Hotchkiss JA, Andrus AK, Johnson KA, Krieger SM, Woolhiser MR, Maurissen JP. Acute toxicologic and neurotoxic effects of inhaled 1,2-dichloroethane in adult Fischer 344 rats. Food Chem Toxicol. 2010;48(2)470–481.
25. Perbellini L, Princivalle A, Caivano M, Montagnani R. Biological monitoring of occupational exposure to N,N-dimethylacetamide with identification of a new metabolite. Ann Occup Environ Med. 2003;60(10)746.
26. Okuda H, Takeuchi T, Senoh H, et al. Developmental Toxicity Induced by Inhalation Exposure of Pregnant Rats to N,N-Dimethylacetamide. J Occup Health. 2006;48(3)154–160.
27. Cai S-X, Huang M-Y, Xi L-Q, et al. Occupational dimethylformamide exposure. Int Arch Occ Env Hea. 1992;63(7)461–468.
28. Fleming LE, Shalat SL, Redlich CA. Liver injury in workers exposed to dimethylformamide. Scand J Work Environ Health. 1990;16(4)289–292.
29. Redlich CA, Beckett WS, Sparer J, et al. LIver disease associated with occupational exposure to the solvent dimethylformamide. Ann Intern Med. 1988;108(5)680–686.
30. Wrbitzky R. Liver function in workers exposed to N,N-dimethylformamide during the production of synthetic textiles. Int Arch Occ Env Hea. 1999;72(1)19–25.
31. Syme R, Bewick M, Stewart D, Porter K, Chadderton T, Glück S. The role of depletion of dimethyl sulfoxide before autografting: on hematologic recovery, side effects, and toxicity. Biol Blood Marrow Tr. 2004;10(2)135–41.
32. Glowa JR. Behavioral and neuroendocrine effects of diethyl ether exposure in the mouse. Neurotoxicol Teratol. 1993;15(4)215–21.
33. Takeuchi Y, Ono Y, Hisanaga N, Kitoh J, Sugiura Y. A comparative study on the neurotoxicity of n-pentane, n-hexane, and n-heptane in the rat. Br J Ind Med. 1980;37(3)241.
34. Perbellini L, Brugnone F, Cocheo V, De Rosa E, Bartolucci GB. Identification of the n-heptane metabolites in rat and human urine. Arch Toxicol. 1986;58(4)229–234.
35. Herskowitz A, Ishii N, Schaumburg H. N-Hexane Neuropathy. A syndrome occurring as a result of industrial exposure. New Engl J Med. 1971;285(2)82–85.
36. Iwata M, Takeuchi Y, Hisanaga N, Ono Y. A study on biological monitoring of n-Hexane exposure. Int Arch Occ Env Hea. 1983;51(3)253–60.
37. Sanagi S, Seki Y, Sugimoto K, Hirata M. Peripheral nervous system functions of workers exposed to n-hexane at a low level. Int Arch Occ Env Hea. 1980;47(1)69–79.
38. Wada Y. Intoxication polyneuropathy following exposure to n-hexane. Clin Neurol. 1965;5:591–598.
39. Eells JT, Salzman MM, Lewandowski MF, Murray TG. Formate-Induced Alterations in Retinal Function in Methanol-Intoxicated Rats. Toxicol Appl Pharm. 1996;140(1)58–69.
40. Prah JD, Goldstein GM, Devlin R, et al. Sensory, Symptomatic, Inflammatory, and Ocular Responses to and the Metabolism of Methyl Tertiary Butyl Ether in a Controlled Human Exposure Experiment. Inhal Toxicol. 1994;6(6)521–38.
41. Saida K, Mendell JR, Weiss HS. Peripheral nerve changes induced by methyl n-butyl ketone and potentiation by methyl ethyl ketone. J Neuropathol Exp Neurol. 1976;35(3)207–225.
42. Lee KP, Chromey NC, Culik R, Barnes JR, Schneider PW. Toxicity of N-Methyl-2-pyrrolidone (NMP): Teratogenic, Subchronic, and Two-Year Inhalation Studies. Toxicol Sci. 1987;9(2)222–235.
43. Bader M, Rosenberger W, Rebe T, et al. Ambient monitoring and biomonitoring of workers exposed to N-methyl-2-pyrrolidone in an industrial facility. Int Arch Occ Env Hea. 2006;79(5)357–364.
44. Lübbe J, Ruffieux C, Van Melle G, Perrenoud D. Irritancy of the skin disinfectant n-propanol. Contact Derm. 2001;45(4)226–231.
45. Rajabally YA, Mortimer NJ. Acute neuropathy and erythromelalgia following topical exposure to isopropanol. Vet Hum Toxicol. 2004;46(1)24–25.
46. Elovaara E, Pfäffli P, Savolainen H. Burden and Biochemical Effects of Extended Tetrahydrofuran Vapour Inhalation of Three Concentration Levels. Acta Pharmacol Toxicol. 1984;54(3)221–226.
47. Abbate C, Giorgianni C, Munaò F, Brecciaroli R. Neurotoxicity induced by exposure to toluene. Int Arch Occ Env Hea. 1993;64(6)389–392.
48. von Euler G, Ögren SO, Li XM, Fuxe K, Gustafsson JÅ. Persistent effects of subchronic toluene exposure on spatial learning and memory, dopamine-mediated locomotor activity and dopamine D2 agonist binding in the rat. Toxicology. 1993;77(3)223–232.
49. Langman JM. Xylene: Its toxicity, measurement of exposure levels, absorption, metabolism and clearance. Pathology. 1994;26(3)301–309.
50. Low Z-X, Chua YT, Ray BM, Mattia D, Metcalfe IS, Patterson DA. Perspective on 3D printing of separation membranes and comparison to related unconventional fabrication techniques. J Membrane Sci. 2017;523:596–613.
51. Jasiuk I, Abueidda DW, Kozuch C, Pang S, Su FY, McKittrick J. An Overview on Additive Manufacturing of Polymers. JOM. 2018;70(3)275–283.
52. Yanar N, Son M, Park H, Choi H. Bio-mimetically inspired 3D-printed honeycombed support (spacer) for the reduction of reverse solute flux and fouling of osmotic energy driven membranes. J Ind Eng Chem. 2019;83:343–350.
53. Yanar N, Son M, Yang E, et al. Investigation of the performance behavior of a forward osmosis membrane system using various feed spacer materials fabricated by 3D printing technique. Chemosphere. 2018;202:708–715.
54. Chu W, Tan Y, Wang P, et al. Centimeter-Height 3D Printing with Femtosecond Laser Two-Photon Polymerization. Adv Mater Technol. 2018;3(5)1700396.
55. You S, Li J, Zhu W, Yu C, Mei D, Chen S. Nanoscale 3D printing of hydrogels for cellular tissue engineering. J Mater Chem B. 2018;6(15)2187–2197.
56. Vyatskikh A, Delalande S, Kudo A, Zhang X, Portela CM, Greer JR. Additive manufacturing of 3D nano-architected metals. Nat Commun. 2018;9(1)593.
57. Yang Y, Li X, Zheng X, Chen Z, Zhou Q, Chen Y. 3D-Printed Biomimetic Super-Hydrophobic Structure for Microdroplet Manipulation and Oil/Water Separation. Adv Mater. 2018;30(9)1704912.
58. Witze A. NASA to send 3D printer into space. Macmillan Publishers Ltd; London, England: 2014.
60. Jasveer S, Jianbin X. Comparison of different types of 3D printing technologies. IJSRP. 2018;8(4)1–9.
61. 3D Systems. Polypropylene-like Materials [Internet]. c2020;[cited 18 April 2020]. Available from: https://www.3dsystems.com/materials/plastic
Fig. 2Effect of manufacturing on energy consumption. The image is reproduced from a previous study [14] of U.S. Energy Information Administration. 
Fig. 3Calculated solvent concentration in the wastewater during conventional membrane fabrication processes with the threshold level for requiring an appropriate treatment prior to disposal or reuse. This figure shows the outcome of a survey conducted by Razali et al. about how membrane manufacturing companies dispose of coagulation bath wastewater. The image is reproduced from Ref. [18] – Published by The Royal Society of Chemistry. 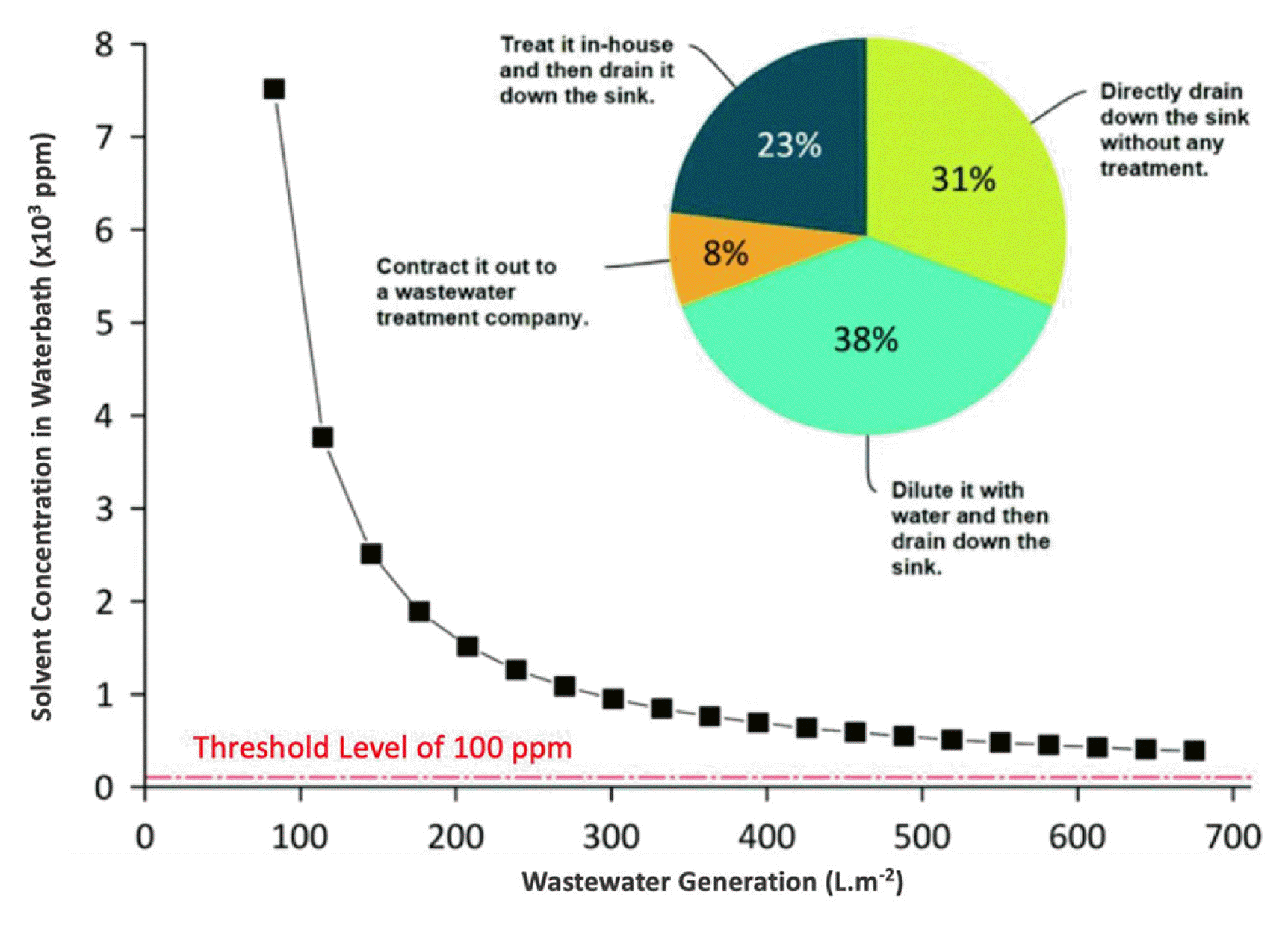
Table 1Toxic solvents used during membrane fabrication and their related health effects (* indicates results obtained from animal experiments).
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||